| This is the sandbox page where you will draft your initial Wikipedia contribution.
If you're starting a new article, you can develop it here until it's ready to go live. If you're working on improvements to an existing article, copy only one section at a time of the article to this sandbox to work on, and be sure to use an edit summary linking to the article you copied from. Do not copy over the entire article. You can find additional instructions here. Remember to save your work regularly using the "Publish page" button. (It just means 'save'; it will still be in the sandbox.) You can add bold formatting to your additions to differentiate them from existing content. |
Role in disease
RyR1 mutations are associated with malignant hyperthermia and central core disease. Mutant-type RyR1 receptors exposed to volatile anesthetics or other triggering agents can display an increased affinity for cytoplasmic Ca2+ at activating sites as well as a decreased cytoplasmic Ca2+ affinity at inhibitory sites. [1] The breakdown of this feedback mechanism leads to uncontrolled release of Ca2+ into the cytoplasm, and increased ATP hydrolysis resulting from ATPase enzymes shuttling Ca2+ back into the sarcoplasmic reticulum leads to excessive heat release. [2]
RyR2 mutations play a role in stress-induced polymorphic ventricular tachycardia (a form of cardiac arrhythmia) and ARVD. It has also been shown that levels of type RyR3 are greatly increased in PC12 cells overexpressing mutant human Presenilin 1, and in brain tissue in knockin mice that express mutant Presenilin 1 at normal levels, and thus may play a role in the pathogenesis of neurodegenerative diseases, like Alzheimer's disease.
The presence of antibodies against ryanodine receptors in blood serum has also been associated with myasthenia gravis.
Sudden cardiac death in several young individuals in the Amish community (four of which were from the same family) was traced to homozygous duplication of a mutant RyR2 (Ryanodine Receptor) gene. Normal (wild type) ryanodine receptors are involved in CICR in heart and other muscles, and RyR2 functions primarily in the myocardium (heart muscle).
Structure
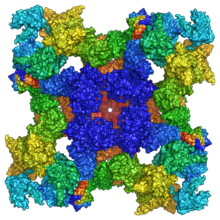
Ryanodine receptors are multidomain homotetramers which regulate intracellular calcium ion release from the sarcoplasmic and endoplasmic reticula. [3] They are the largest known ion channels, with weights exceeding 2 megadaltons, and their structureal complexity enables a wide variety of allosteric regulation mechanisms. [4] [5] RyR1 cryo-EM structure revealed a large cytosolic assembly built on an extended α-solenoid scaffold connecting key regulatory domains to the pore. The RyR1 pore architecture shares the general structure of the six-transmembrane ion channel superfamily. A unique domain inserted between the second and third transmembrane helices interacts intimately with paired EF-hands originating from the α-solenoid scaffold, suggesting a mechanism for channel gating by Ca2+. [6] [7]
-
^ Yang, Tianzhong; Ta, Tram Anh; Pessah, Isaac N.; Allen, Paul D. (2003-07).
"Functional Defects in Six Ryanodine Receptor Isoform-1 (RyR1) Mutations Associated with Malignant Hyperthermia and Their Impact on Skeletal Excitation-Contraction Coupling". Journal of Biological Chemistry. 278 (28): 25722–25730.
doi:
10.1074/jbc.m302165200.
ISSN
0021-9258.
{{ cite journal}}: Check date values in:|date=( help)CS1 maint: unflagged free DOI ( link) - ^ Reis, Marcelo; Farage, Mariana; Meis, Leopoldo de (2002-01-01). "Thermogenesis and energy expenditure: control of heat production by the Ca 2+ -ATPase of fast and slow muscle". Molecular Membrane Biology. 19 (4): 301–310. doi: 10.1080/09687680210166217. ISSN 0968-7688.
-
^ Santulli, Gaetano; Lewis, Daniel; des Georges, Amedee; Marks, Andrew R.; Frank, Joachim (2018), Harris, J. Robin; Boekema, Egbert J. (eds.),
"Ryanodine Receptor Structure and Function in Health and Disease", Membrane Protein Complexes: Structure and Function, vol. 87, Singapore: Springer Singapore, pp. 329–352,
doi:
10.1007/978-981-10-7757-9_11,
ISBN
978-981-10-7756-2,
PMC
5936639,
PMID
29464565, retrieved 2023-05-14
{{ citation}}: CS1 maint: PMC format ( link) -
^ Lanner, J. T.; Georgiou, D. K.; Joshi, A. D.; Hamilton, S. L. (2010-11-01).
"Ryanodine Receptors: Structure, Expression, Molecular Details, and Function in Calcium Release". Cold Spring Harbor Perspectives in Biology. 2 (11): a003996–a003996.
doi:
10.1101/cshperspect.a003996.
ISSN
1943-0264.
PMC
2964179.
PMID
20961976.
{{ cite journal}}: CS1 maint: PMC format ( link) -
^ Van Petegem, Filip (2015-01).
"Ryanodine Receptors: Allosteric Ion Channel Giants". Journal of Molecular Biology. 427 (1): 31–53.
doi:
10.1016/j.jmb.2014.08.004.
{{ cite journal}}: Check date values in:|date=( help) - ^ Santulli G, Marks AR (2015). "Essential Roles of Intracellular Calcium Release Channels in Muscle, Brain, Metabolism, and Aging". Current Molecular Pharmacology. 8 (2): 206–22. doi: 10.2174/1874467208666150507105105. PMID 25966694.
- ^ Zalk R, Clarke OB, des Georges A, Grassucci RA, Reiken S, Mancia F, et al. (January 2015). "Structure of a mammalian ryanodine receptor". Nature. 517 (7532): 44–9. Bibcode: 2015Natur.517...44Z. doi: 10.1038/nature13950. PMC 4300236. PMID 25470061.