Test.

BoPET (biaxially-oriented polyethylene terephthalate) is a polyester film made from stretched polyethylene terephthalate (PET) and is used for its high tensile strength, chemical and dimensional stability, transparency, reflectivity, gas and aroma barrier properties, and electrical insulation.
A variety of companies manufacture boPET and other polyester films under different brand names. In the UK and US, the most well-known trade names are Mylar, Melinex, and Hostaphan. [1]
BoPET is used to create flexible types of packaging and lids used by many different industries such as the food, comic and archival storage industries. It is also commonly used as electrical and thermal insulation. [2]
History
Polyester (PET), a commonly recognized synthetic fiber made up of chemicals found in petroleum, uses Polyethylene Terephthalate as a basis of production. Polyester's large history of research led to the production of an extremely durable type of polyester, named BoPET (commonly known as Mylar), in the 1950's.
Original Patent
In 1941, an advancement upon the research of Wallace Carothers, an Organic Chemist, led to the creation and patent of the substance known as Polyethylene Terephthalate. This advancement is accredited to John Rex Whinfield and James Tennant Jackson, two chemists of the Calico's Printer Association of Manchester. This invention led to the creation of the first polyester fiber, laying down the stepping stones to the invention of BoPET, soon to be known as the strongest synthetic polyester film of it's time.
DuPont's Expansion
During the 1920's, DuPont, now known for the creation BoPET (Mylar), was heavily focused on nylon research, which at the time, was considered a very highly regarded up and coming synthetic fiber. It wasn't until 1945, DuPont's purchasing of Imperial Chemical Industries' (ICI) patent for Terylene polyester in hopes of further development of the invention, that DuPont began to re-initiate their research efforts towards the polyester industry. This new research initiative brought forth many trademarked polyester products, including the infamous Mylar, also known as BoPET. [3]
BoPET film was developed in the mid-1950s, originally by DuPont, Imperial Chemical Industries (ICI), and Hoechst. [4]
Historical Use
In 1955 Eastman Kodak used Mylar as a support for photographic film and called it "ESTAR Base". [5] The very thin and tough film allowed 6,000-foot (1,800 m) reels to be exposed on long-range U-2 reconnaissance flights. [6]
In 1964, NASA launched Echo II, a 40-metre (131 ft) diameter balloon constructed from a 9-micrometre (0.00035 in) thick mylar film sandwiched between two layers of 4.5-micrometre (0.00018 in) thick aluminum foil bonded together. [7]
Manufacture
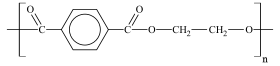
Biaxial or Monaxial orientation methods are used in the production of BoPET since it is a thinner material, so that it can have properties such as tensile strength, toughness, heat stability, tear initiation resistance, and barrier.
Biaxial Orientation
The manufacturing process begins with a film of molten polyethylene terephthalate (PET) being extruded onto a chill roll, which quenches it into the amorphous state. [8] It is then biaxially oriented by drawing. The most common way of doing this is the sequential process, in which the film is first drawn in the machine direction using heated rollers and subsequently drawn in the transverse direction, i.e. orthogonally to the direction of travel, in a heated oven. It is also possible to draw the film in both directions simultaneously, although the equipment required for this is somewhat more elaborate. Draw ratios are typically around 3 to 4 in each direction.
Once the drawing is completed, the film is " heat set" or crystallized under tension in the oven at temperatures typically above 200 °C (392 °F). The heat setting step prevents the film from shrinking back to its original unstretched shape and locks in the molecular orientation in the film plane. The orientation of the polymer chains is responsible for the high strength and stiffness of biaxially oriented PET film, which has a typical Young's modulus of about 4 GPa (0.58×106 psi). Another important consequence of the molecular orientation is that it induces the formation of many crystal nuclei. The crystallites that grow rapidly reach the boundary of the neighboring crystallite and remain smaller than the wavelength of visible light. As a result, biaxially oriented PET film has excellent clarity, despite its semicrystalline structure.
If it were produced without any additives, the surface of the film would be so smooth that layers would adhere strongly to one another when the film is wound up, similar to the sticking of clean glass plates when stacked. To make handling possible, microscopic inert inorganic particles are usually embedded in the PET to roughen the surface of the film such as silicon dioxide. [9]
Biaxially oriented PET film can be metallized by vapor deposition of a thin film of evaporated aluminium, gold, or other metal onto it. The result is much less permeable to gases (important in food packaging) and reflects up to 99% of light, including much of the infrared spectrum. For some applications like food packaging, the aluminized boPET film can be laminated with a layer of polyethylene, which provides sealability and improves puncture resistance. The polyethylene side of such a laminate appears dull and the PET side shiny.
Other coatings, such as conductive indium tin oxide (ITO), can be applied to boPET film by sputter deposition.
Monaxial Orientation
Monaxial Orientation is most commonly used when the BoPET material is being manufactured for the use of shrink-sleeve label films. This is due to the fact that this process allows for a high amount of shrinkage to occur when the film is exposed to relatively low temperatures, as well as increased stiffness, optical properties, barrier properties and increased tear strength, which is desired for this type of product. The process is extremely similar to that of Biaxial Orientation, but has differences towards the end of the production process. Once the thicker film of molten polyethylene terephthalate (PET) is laid onto a cooled surface, it is immediately temperature conditioned, and then is stretched between two rollers of differentiating speeds. These rollers are held extremely close to one another to lower the amount of unwanted gap where orientation normally takes place. [10]
Properties
BoPET has many properties that make it very useful in the commercial world. These properties include;
- Transparent
- Electric Insulator
- High Tensile Strength
- Chemical Stability
- Reflective
- Gas Barrier
- Odor Barrier
- Heat Stability
- Tear Initiation Resistance [2]
Applications
BoPET (Mylar), originally created in the 1950's, has since been used in many types of industrial areas, such as; electronics, magnetic media, imaging, graphics, industrial, and packaging fields. [11]
Uses for boPET polyester films include, but are not limited to:
Packaging
- Laminates containing metallized boPET foil (In technical language called printin or laminate web substrate) protect food against oxidation and aroma loss due to its gas barrier properties, as well as its high durability., achieving long shelf life. Examples are coffee "foil" packaging and pouches for convenience foods.
- BoPET is used in packaging that contains products which are irritable to moisture changes, such as pharmaceutical products.
- White boPET web substrate is used as lidding for dairy goods such as yogurt.
- Clear boPET web substrate is used as lidding for fresh or frozen ready meals. Due to its excellent heat resistance, it can remain on the package during microwave or oven heating.
- Roasting bags
- Metallised films
- Laminated sheet metal (aluminum or steel) used in the manufacture of cans ( bisphenol A-free alternative to lacquers)
Covering over paper
- A clear overlay on a map, on which notations, additional data, or copied data, can be drawn without damaging the map
- Metallized boPET is used as a mirror-like decorative surface on some book covers, T-shirts, and other flexible cloths.
- Protective covering over buttons/pins/ badges
- The glossy top layer of a Polaroid SX-70 photographic print
- As a backing for very fine sandpaper
- boPET film is used in bagging comic books, in order to best protect them during storage from environmental conditions (moisture, heat, and cold) that would otherwise cause paper to slowly deteriorate over time. This material is used for archival quality storage of documents by the Library of Congress [12] (specifically Mylar® type D) [13] and several major library comic book research collections, including the Comic Art Collection at Michigan State University. [14] While boPET is widely (and effectively) used in this archival sense, it is not immune to the effects of fire and heat and could potentially melt, depending on the intensity of the heat source, causing further damage to the encased item. [15]
- Similarly, trading card decks (such as Pokémon, Magic: The Gathering, and Yu-Gi-Oh!) are packaged in pouches or sleeves made of metallized boPET. It can also be used to make the holographic artwork featured on some cards, typically known as "holos", "foils", or "holofoils".
- For protecting the spine of important documents, such as medical records.
Insulating material
- An electrical insulating material
- Insulation for houses and tents, reflecting thermal radiation
- One sided reflective BoPET is often used as insulation because of its metallic properties, allowing it to reflect 99% of light
- When being used to increased warmth on the interior, the reflective side is placed facing inwards so that it will reflect the heat that would normally escape
- When being used to keep cool, the reflective side is placed facing outward so that it reflects heat from the outside, which would otherwise penetrate into the interior
- Five layers of metallized boPET film in NASA's spacesuits make them radiation resistant and help regulate temperature.
- Metallized boPET film emergency blankets conserve a shock victim's body heat.
- As a thin strip to form an airtight seal between the control surfaces and adjacent structure of aircraft, especially gliders.
- Light insulation for indoor gardening.
- Fire shelters, used by wildland firefighters.
- Aluminized proximity suits used by fire fighters for protection from the high amount of heat release from fuel fires.
- Used in sock and glove liners to lock in warmth
Solar, marine and aviation applications
- Metallized boPET is intended to be used for solar sails as an alternative means of propulsion for spacecraft such as Cosmos 1
- Metallized boPET solar curtains reflect sunlight and heat away from windows.
- Aluminized, as an inexpensive solar eclipse viewer, although care must be taken, because invisible fissures can form in the metal film, reducing its effectiveness.
- High performance sails for sailboats, hang gliders, paragliders and kites
- Use boPET films as the back face of the PV modules in solar panels
- Metallized boPET as a reflector material for solar cooking stoves
- To bridge control surface gaps on sailplanes (gliders), reducing profile drag
Science
- Amateur and professional visual and telescopic solar filters. BoPET films are often annealed to a glass element to improve thermal conductivity, and guarantee the necessary flat surface needed for even telescopic solar observation. Manufacturers will typically use films with thicknesses of 280–500 micrometres (0.011–0.020 in), in order to give the films better resilience. 250-micrometre (0.0098 in) thickness films with a heavy aluminium coating are generally preferred for naked-eye Solar observation during eclipses.
- Films in annular ring mounts on gas-tight cells, will readily deform into spherical mirrors. Photomultiplier cosmic-ray observatories often make use of these mirrors for inexpensive large (1.0 m and above), lightweight mirror surfaces for sky-sector low and medium energy cosmic ray research.
- As a light diaphragm material separating gases in hypersonic shock and expansion tube facilities.
- As a beamsplitter in Fourier transform infrared spectroscopy, typically with laser applications. Film thicknesses are often in the 500 micrometre range.
- Coating around hematocrit tubes.
- Insulating material for a cryocooler radiation shield.
- As a window material to confine gas in detectors and targets in nuclear physics.
- In CT scanners it acts as a physical barrier between the xray-tube, detector ring and the patient allowing negligible attenuation of the xray beam when active.
- Spacecraft are insulated with a metallized BoPET film.
- The material for solar sails of spacecraft.
Biomedical
- BoPET is one of the commonly employed polymers clinically for orthopedic applications, vascular grafts, ocular application, dentistry, tissue engineering, and drug delivery systems
- BoPET's attributes allow for specific level of durability, stiffness, bio compatibility, bio stability, chemical stability, and dimensional stability, making it the best suited material
- Hydrophobic aromatic groups with high levels of crystalline structures present that can restrict hydrolytic breakdown are introduced to a Polyethylene Terephthalate matrix
- Surface functionalization has been used to improve polymer-cell interaction
- Holds advantages over non-degradable materials such as metals or ceramics [16]
Electronic and acoustic applications
- Carrier for flexible printed circuits.
- boPET film is often used as the diaphragm material in headphones, electrostatic loudspeakers and microphones.
- boPET film has been used in the production of banjo & drumheads since 1958 due to its durability and acoustical properties when stretched over the bearing edge of the drum. They are made in single- and double-ply versions, with each ply being 2–10 mils (0.051–0.254 mm) in thickness, with a transparent or opaque surface, originally used by the company Evans.
- The advantage's of BoPET film allow for a consistency in pitch and tone that is otherwise impossible
- boPET film is used as the substrate in practically all magnetic recording tapes and floppy disks.
- Metallized boPET film, along with other plastic films, is used as a dielectric in foil capacitors.
- Clear boPET bags are used as packaging for audio media such as compact discs and vinyl records.
- Clear and white boPET films are used as core layers and overlays in Smart Cards.
Hydroponic Gardening
- BoPET can be used to reflect wasted light towards a desired target
- Improves the usability of light within the plant when practicing Hydroponic Gardening
- Advantages from BoPET use cause an increase in the speed of growth for the plant itself
- Can increase light efficiency by 40%, or even more if the patterns of reflection overlap across multiple fixtures and reflectors
- BoPET is popular for use in grow tents, where it is bonded to the interior walls, ceiling and floor, while a thick canvas fabric is used for the outer shell
- Using patterned Mylar reduces instances of hot spots and broadcasts light the best [11]
Printing Mediums
- Before the widespread adoption of CAD, engineering drawings or architectural drawings were plotted onto sheets of boPET film, known as drafting film. The boPET sheets become legal documents from which copies or blueprints are made. boPET sheets are more durable and can withstand more handling than bond paper. Although " blueprint" duplication has fallen out of use, mylar is still used for its archival properties, typically as a record set of plans for building departments to keep on file.
- Overhead transparency film for photocopiers or laser printers (boPET film withstands the high heat).
Other
- Balloons, metallic balloons.
- Route information signs, called rollsigns or destination blinds, displayed by public transport vehicles
- For materials in kites
- Covering glass to decrease probability of shattering
- In theatre effects as confetti.
- As the adhesive strip to attach the string to a teabag
- One of the many materials used as windsavers or valves for valved harmonicas
- On farmland and domestic gardens, highly reflective aluminized PET film ribbons are used to keep birds away from plants.
- Measuring tape
- Protecting pinball machine playfields from wear
- Used in dentistry when restoring teeth with composite.
- In nail polish, as a coloured and finely shredded additive to create a glitter effect.
- Numismatics - The product is used instead of PVC for safe coin storage. Chemical composition is more conducive to the metals coins are made of i.e Silver & Copper (Coins used to be stored in PVC sleeves which damage coins when exposed for longer periods due to chlorine being released.)
Technological Advancements
The plastic resin known as Polyethylene Terephthalate is the most commercially common polyester that is used today. Soon after being created, Polyethylene Terephthalate became the most wide spread synthetic fiber produced around the world. This technology today has advanced so greatly that it is now seen in almost every application imaginable.
Carbon Reinforcement
Looking to increase Polyethylene Terephthalate's already powerful electrical, chemical and physical-chemical properties, scientists nowadays have found that adding carbon based materials such as graphene, carbon fiber, and carbon nanotubes to the PET Polymer is the answer to creating a more powerful synthetic fiber. This is due to carbon based materials having extremely prominent thermal conductivity properties, as well as strong chemical and physical-chemical properties.
Nano-Composite Technology
Polyethylene Terephthalate's wide variety of applications and field of use made it an automatically assumed prospect for use within newly emerging technological advances now and in the future as well. Using brand new developments surrounding the synthesis of PET Nano-composites, Polyethylene Terephthalate is now able to see huge increases in highly desired properties such as mechanical strength, permeability, electrical conductivity, and thermal stability. This is accomplished by incorporating nano-fillers such as carbon nanotubes, nano-clays, and other inorganic nanoparticles. These new advancements suggest a very promising future of nano-composite PET technologies. [16]
References
- ^ Mark T. DeMeuse (2011). Biaxial Stretching of Film: Principles And Applications. Elsevier. p. 48.
- ^ a b Helmenstine, Anne Marie, Ph D. Dr Helmenstine holds a Ph D. in biomedical sciences, Is a Science Writer, educator, consultant She has taught science courses at the high school, college, and Graduate Levels. “What You Should Know About Mylar.” ThoughtCo. Accessed March 10, 2019. https://www.thoughtco.com/mylar-polyester-film-608929
- ^ “The History of Polyester.” Accessed March 10, 2019. http://theinventors.org/library/inventors/blpolyester.htm.
-
^ See, for example:
- Izard, Emmette Farr, "Production of polyethylene terephthalate", U.S. patent no. 2,534,028 (filed: 1948 May 13; issued: 1950 December 12).
- Adams, John Francis Edward; Gerber, Kenneth George; Holmes-Walker, William Anthony, "Process for the production of biaxially oriented polyethylene terephthalate film", U.S. patent no. 3,177,277 (filed: 1957 May 10 ; issued: 1965 April 6).
- ^ "Kodak HCF Film/ESTAR Base" (PDF). www.kodak.com. Eastman Kodak Company. April 2015. Retrieved 2018-08-24.
- ^ Eyes in the Sky, Dino A. Brugioni 2010, Naval Institute Press, ISBN 978 1 59114 082 5, pp. 102, 115.
- ^ Staugaitis, C. & Kobren, L. "Mechanical And Physical Properties of the Echo II Metal-Polymer Laminate (NASA TN D-3409)", NASA Goddard Space Flight Center (1966).
- ^ "Process Flow". Ampef.com. Retrieved 2018-08-24.
- ^ Thiel, Ulrich. "Polyester Additives" (PDF). Dr. Thiele Polyester Technology. Retrieved 4 January 2019.
- ^ “Understanding Biaxially and Monaxially Oriented Films | Packaging World.” Accessed March 10, 2019. https://www.packworld.com/article/material-type/polymers/understanding-biaxially-and-monaxially-oriented-films.
- ^ a b “Uses of Mylar | Techwalla.Com.” Techwalla. Accessed March 10, 2019. https://www.techwalla.com/articles/uses-of-mylar
- ^ [1] Archived June 4, 2004, at the Wayback Machine
- ^ "What is Mylar Paper - More Than Just Decoration". Jampaper.com. Retrieved 2015-07-02.
- ^ Scott, Randall W. (1998). "A Practicing Comic-Book Librarian Surveys His Collection and Craft". Serials Review. 24 (1): 49–56.
- ^ "Albany library's entire collection exposed to smoke | Athens County Public Libraries". Myacpl.org. 2008-06-17. Retrieved 2015-07-02.
- ^ a b "ScienceDirect". www.sciencedirect.com. Retrieved 2019-03-23.
External links
- History of Polymers & Plastics for Teachers. by The American Chemistry Council (HTML format) or (PDF format) - 1.9MB, which includes the " chasing arrow" recycling symbols (PET is #1) and a description of plastics.
- An interesting toy has been developed using boPET and a stick-shaped Van de Graaff generator.
- The Biaxial Orientation Production process is displayed.
- Patent for the Biaxial Orientation Production process. Filed by EI duPont de Nemours and Co
Category:Dielectrics Category:Plastics Category:Polyesters Category:Reflective building components Category:Packaging materials
