This article needs additional citations for
verification. (December 2014) |
| |

| |
| Names | |
|---|---|
|
IUPAC name
Diphosphono hydrogenphosphate
| |
|
Systematic IUPAC name
Triphosphoric acid Tripolyphosphoric acid | |
| Other names
Diphosphonophosphoric acid
Phosphono trihydrogenpyrophosphate Phosphonopyrophosphoric acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.030.752 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
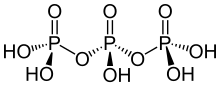
| H5P3O10 | |
| Molar mass | 257.95 g/mol |
| Acidity (pKa) | See body |
| Conjugate base | Triphosphate |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Corrosive (C) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Triphosphoric acid (also tripolyphosphoric acid), with formula H5P3O10, is a condensed form of phosphoric acid. In the family of phosphoric acids, it is the next polyphosphoric acid after pyrophosphoric acid, H4P2O7, also called diphosphoric acid.
Compounds such as ATP ( adenosine triphosphate) are esters of triphosphoric acid.
Triphosphoric acid has not been obtained in crystalline form. The equilibrium mixture with an overall composition corresponding to H5P3O10 contains about 20% of triphosphoric acid. A solution of the pure species can be obtained by ion exchange of the sodium salt, sodium triphosphate, at 0 °C. [1]
Triphosporic acid is a pentaprotic acid, meaning that it can release five protons in basic enough conditions. Sources differ on the corresponding pKa values:
- 1.0; 2.2; 2.3; 5.7; 8.5 [1]
- 1.0; 2.2; 2.3; 3.7; 8.5 [2]
- small; small; 2.30; 6.50; 9.24[ citation needed]
References
- ^ a b Corbridge, D. (1995). "Chapter 3: Phosphates". Studies in inorganic Chemistry vol. 20. Elsevier Science B.V. pp. 169–305. ISBN 0-444-89307-5.
- ^ Holleman, Arnold Frederik; Wiberg, Egon (2001), Wiberg, Nils (ed.), Inorganic Chemistry, translated by Eagleson, Mary; Brewer, William, San Diego/Berlin: Academic Press/De Gruyter, p. 729, ISBN 0-12-352651-5