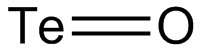
| |
| Names | |
|---|---|
| Other names
Tellurium(II) oxide
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| TeO | |
| Molar mass | 143.60 g/mol |
| Related compounds | |
Other
cations
|
Sulfur monoxide Polonium monoxide |
|
Tellurium dioxide Tellurium trioxide | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
The diatomic molecule tellurium monoxide has been found as a transient species. [2] Previous work that claimed the existence of TeO solid has not been substantiated. [3] The coating on DVDs called tellurium suboxide may be a mixture of tellurium dioxide and tellurium metal. [4]
History
Tellurium monoxide was first reported in 1883 by E. Divers and M. Shimose. [5] It was supposedly created by the thermal decomposition of tellurium sulfoxide in a vacuum,[ citation needed] and was shown to react with hydrogen chloride in a 1913 report. [6] Later work has not substantiated the claim that this was a pure solid compound. [2] By 1984, the company Panasonic was working on an erasable optical disk drive containing "tellurium monoxide" (really a mixture of Te and TeO2). [7]
See also
- Tellurium dioxide
- Tellurium trioxide
- Lead carbide – originally thought to be a pure compound, but now considered more likely to be a mixture of carbon and lead
- Iodine pentabromide – originally thought to be a pure compound, but now considered to probably be a mixture of iodine monobromide and excess unreacted bromine
References
- ^ "Tellurium monoxide". NIST (National Institute of Standards and Technology). Retrieved 2013-12-16.
- ^ a b Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8..
- ^ Viktor Guttman, Main Group Elements: Group VI and Group VII - p. 141.
- ^ Tyan, Y.-S.; Preuss, D. R.; Vazan, F.; Marino, S. J. (1986). "Laser recording in tellurium suboxide thin films". Journal of Applied Physics. 59 (3): 716. Bibcode: 1986JAP....59..716T. doi: 10.1063/1.336588. ISSN 0021-8979.
- ^ Sir William Crookes, Chemical News and Journal of Industrial Science, vol. 49, página 93. Chemical news office, 1884 (digitalized 15 Dec. 2008). Visited 2013-12-03.
- ^ The Analyst, vol. 37, Royal Society of Chemistry, Society of Public Analysts and Other Analytical Chemists, Society for Analytical Chemistry, Chemical Society, Royal Society of Chemistry, 1913 (digitalized 31 mar. 2010).
- ^ Electronic Design, vol. 32, nr. 24-26, p. 11, Hayden Publishing Company, 1984. Visited 2013-12-03.