| This is an archive of past discussions. Do not edit the contents of this page. If you wish to start a new discussion or revive an old one, please do so on the current talk page. |
| Archive 1 | Archive 2 | Archive 3 | Archive 4 | Archive 5 |
Picture of an atom
In your picture of the helium atom would it be possible to label the nucleons conseculatively 1,2 3, and 4, and then draw an arrow through each of the nucleons to show the spin orientation relationship?WFPM WFPM ( talk) 18:30, 27 August 2008 (UTC) It would be really nice to have a real image of an atom. I know it is done, I even think I saw it in O'Hanian.
- Are you referring to having an accurate diagram of an atom, or an image of an atom made from a Scanning tunneling microscope (or equivalent)? Iotha 02:53, 26 January 2007 (UTC)
"If an apple was magnified to the size of the Earth, then the atoms in the apple would be approximately the size of the original apple." - If satilites can view people from outer space, then surely we will soon have the technology to see the atoms in an apple? (Seb-Gibbs) —Preceding unsigned comment added by Seb-Gibbs ( talk • contribs) 21:22, 23 January 2008 (UTC)
Atoms are smaller than the wavelength of visible light, so even if you had an unbelievably powerful microscope, it will always be impossible to see without special imaging techniques (that's why we use electron microscopes). 24.131.183.162 ( talk) 17:39, 10 February 2008 (UTC) darkstaruav, 10 February, 2008
- You can see a picture of a concept of the atomic nuclei on Talk:Nuclear model. WFPM WFPM ( talk) 16:22, 17 May 2008 (UTC).WFPM WFPM ( talk) 21:24, 27 May 2008 (UTC).WFPM WFPM ( talk) 21:31, 27 May 2008 (UTC)
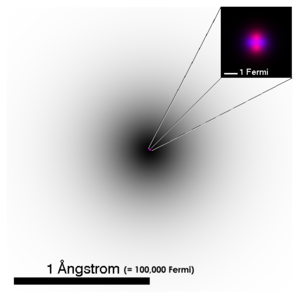
hey, i dont get why you would write 100k fm in the picture - it's meant to be accessible to anyone and a femtometer doesn't mean much to most people.. would be better to express it in millimeters, or even in nanometers, which most people have heard of i think (UTC) —Preceding unsigned comment added by Popffabrik ( talk • contribs)
Question about image of atoms
In the article there is an image that is said to be a picture showing individual atoms, is this true? Does this picture actually show the atoms that make up that sheet of gold?
Sorry if this isnt the propper way of asking this, but I'm just dumbfounded by this, as far as my limited knowledge went, they hadnt been seen nor pictured in any form yet. 200.109.43.50 ( talk) 01:09, 4 January 2008 (UTC)
- Yes, images which represent atoms have been generated for several years. I think the probe is actually detecting the electric field of the atom's electrons at very close range, but I'd have to go read up on the technique to confirm that. The image is resolving things which are the size of atoms, whatever the technology is actually doing; it's equally amazing whether electrons, photons, or X-rays were being used to create the image. -- SEWilco ( talk) 02:04, 4 January 2008 (UTC)
- You're actually looking at a map of electron density (the probability that an electron exists in that location), not a 'snapshot' image of a physical surface (which, after all, doesn't really exist in the way a surface of a macroscopic ball exists)
Furmanj (
talk) 11:48, 4 January 2008 (UTC)
- Nobody has an "Image" of an atom. What is available are images of concepts of atoms from which you are supposed to make your preference. We also have a set of "combined facts and/or opinions" which are the suggested criteria for making such a selection. While doing so, I suggest that you learn as much as you can about the physical events occurring in the volumes of space where atoms are being created. The most notable one I can find is the Hubble images of the center of the Whirlpool Galaxy (M51). If you want an image of a concept, I recommend that you look at Talk:Nuclear model WFPM WFPM ( talk) 23:48, 14 May 2008 (UTC)WFPM WFPM ( talk) 14:06, 15 May 2008 (UTC)WFPM WFPM ( talk) 21:42, 27 May 2008 (UTC)
- You're actually looking at a map of electron density (the probability that an electron exists in that location), not a 'snapshot' image of a physical surface (which, after all, doesn't really exist in the way a surface of a macroscopic ball exists)
Furmanj (
talk) 11:48, 4 January 2008 (UTC)
I'd like to talk here about the article's paragragh about Electron orbit energy levels. These are supposed to be occurring to the orbiting electrons in the electrostatic attractive field areas and involve a concept of the existence of a zero or free electron level from which the involved electron can fall into discrete levels of lost free energy or minus incremental energy values. The incremental units of these values are usually given in Electron volts, which is both an energy times time (erg-second) and an equivalent angular momentum (MVR) unit. In Bohr's original orbit theories the orbits were considered to have actual angular momentum magnitude values, but in the orbital theories no values of angular momentum are considered. And the radiation emissions are usually indicated as being emitted from the center of the atom. This obscures and damages the ability of a person to conceptualize as to how the atom is able to get rid of angular momentum and wuld indicate that some constituent of the atom, hopefully the electron should be retheorized in some manner to allow it to emit radiation at the end of a lever arm, vis a vis the whip principal or whatever. If you'll look at my pictures of real physical nuclear models((Talk:Nuclear model)) you'll see that I had gotten as far as that question in the models, but not far enough to answer the question. But I think it's a worthy subject for discussion in your discussion about Orbital electron energy levels in your evolving artical about the atom.WFPM WFPM ( talk) 21:49, 20 August 2008 (UTC)WFPM WFPM ( talk) 14:59, 21 August 2008 (UTC)
- If I may attempt to jump to the crux of your discussion, you seem to be saying that there is a concern about radiation emissions being perceived as coming from the nucleus, rather than from the decaying electron. The article states that, "In order for an electron to transition between two different states, it must absorb or emit a photon at an energy matching the difference in the potential energy of those levels." This would not seem to agree with your assertion.— RJH ( talk) 18:57, 21 August 2008 (UTC)
I Just guess that I cant concieve of a kind of physical motion of the electron that isnt some kind of radial motion involving a conic radial orbit with constant angular momentum and a constant value of lost potential energy. And if the electron was causing the emission of some of the mass of the atom in the form of radiation particles or radiation waves as goes the theory then a reasonabally correct description of the electron's motion would be desirable for conceptual purposes. And I think it would involve an orbit with a conserved amount of angular momentum. WFPM WFPM ( talk) 21:08, 21 August 2008 (UTC)
Another aspect of this radiation concept problem has to do with what might be called compartmentalism of concepts. So when discussing radiation from the atom it may me adequate to just discuss energy difference values. But when you want to discuss radiation from the center of the ((Whirlpool Galaxy)) you get involved with whether or not the emitted radiation is carrying off some of the observed excess angular momentum in the system as some scientists think it does. And so the process of radiation from matter in that system in terms of MVR change values rather than MVsquared values would have to be conceptually discussed.WFPM 66.139.107.38 ( talk) 16:52, 22 August 2008 (UTC)WFPM WFPM ( talk) 16:59, 22 August 2008 (UTC)
I might even add to that the problem in or own solar system where the sun has managed to acquire 99+ percent of the matter of the original solar gas system but now has only a small portion of of the system's angular momentum.WFPM WFPM ( talk) 16:18, 26 August 2008 (UTC)
- Again, this is off topic. I would probably be best to discuss this matter at the Wikipedia:Reference_desk/Science. Thank you.— RJH ( talk) 22:50, 1 September 2008 (UTC)
Number of atoms in the Universe
I'm having a little difficulty trying to solidify this value, so I moved it here for the moment. The estimates fluctuate depending on the author and I can't find a really solid scientific paper on the topic. Here are some examples:
| Value | Reference |
|---|---|
| 1079 |
"The Universe". National Solar Observatory.
May 21,
2001. Retrieved 2008-02-15. {{
cite web}}: Check date values in: |date= (
help)
|
| 1080 | Champion, Matthew (
September 11,
1998).
"How many atoms make up the universe?". MadSci Network. Retrieved 2007-01-02. {{
cite web}}: Check date values in: |date= (
help)
|
| 1081 |
deGrasse Tyson, Neil (1994). Universe Down to Earth. Columbia University Press. pp. p. 10.
ISBN
023107560X. {{
cite book}}: |pages= has extra text (
help)
|
Any suggestions? Thanks.— RJH ( talk) 17:29, 15 February 2008 (UTC)
- Expansion of the universe? Seriously, it's a problem. First, we should qualify that we're talking only about the visible universe, because the part over the horizon is probably a lot larger, and Travelocity rates to get there are steep. But once you get the mass of the visible part down (which should be derivable from the expansion rate), you subtract dark matter (since not made of atoms), and them divide the rest by the average molecular weight of post big bang "crap", which at one He per 12 H is about 2e27 atoms per kg or so, right?
S
B
H
arris 18:07, 15 February 2008 (UTC)
- I understand how it is derived and what is meant by the visible universe. What is needed is a bullet-proof reference.—
RJH (
talk) 19:12, 15 February 2008 (UTC)
- The size and density of the observable universe were only really pinned down in this century, with the current best data from WMAP. So all these references are out of date. I could derive the number from the data given in the WMAP papers, but it would be WP:original research ;) 140.105.79.118 ( talk) 15:54, 15 May 2008 (UTC)
- I understand how it is derived and what is meant by the visible universe. What is needed is a bullet-proof reference.—
RJH (
talk) 19:12, 15 February 2008 (UTC)
Given the sun's mass at 10E33.3 grams times 10E23.8 nucleons/gram we get 10E57.1 nucleons/sun. Then if we buy the 10E11suns/galaxy and 10E11galaxies/universe theories, we get 10E79.1 nucleons/universe; plus whatever amount of unknown matter you want to add. But I think we ought to start out with a large amount of neutrons. Like maybe 90%?WFPM WFPM ( talk) 02:00, 17 June 2008 (UTC)WFPM WFPM ( talk) 19:11, 17 June 2008 (UTC)
- Don't forget interstellar and intergalactic matter, which are significant contributors.— RJH ( talk) 15:48, 22 June 2008 (UTC)
Kind of depends on which physical problem you're worried about? Galactic rotation? or Red Shift? or what? WFPM WFPM ( talk) 02:29, 24 June 2008 (UTC)
- Nicastro et al (2008) has some interesting observations on the distribution of baryonic matter under the standard cosmological model. At least half of the baryonic matter is currently thought to be in the intergalactic medium.— RJH ( talk) 16:33, 24 June 2008 (UTC)
Is that matter what we're looking at in the center of the whirlpool galaxy? What I see is matter with a lot of excess angular momentum. And how are we supposed to gid rid of that?WFPM WFPM ( talk) 13:12, 27 June 2008 (UTC)
The topic of the article is atoms, and the picture of the center of M51 appears to me to be one of the best in showing the environment in which the process of accumulating matter into atoms is occurring, and I really would like to understand the process, like the matter recipe and how the system is getting rid of what appears to be excess angular momentum. And after all the universe is theorized to be made up of approximately 10E11 of these or similar galaxies. and they just recently discovered a supernova in M51. So if we could explain the recipe and process in it we would be well on our way to understanding the processes of the universe at large. WFPM WFPM ( talk) 23:02, 27 June 2008 (UTC)
- M51 is a planar galaxy and the constituent components are moving primarily under the influence of gravity, including density waves. To claim that it bears a close likeness to an atom is to ignore the fundamental differences between electricity and gravity, and to toss out the well established phenomenon of quantum mechanics. I'll have to disagree.— RJH ( talk) 19:01, 21 August 2008 (UTC)
- ==It is interesting to note that the article starts of about atoms as if they were either the or among the most important constituent parts of the group of entities making up the "Real Physical Universe" It is only deep into a discussion of their "origin" and conjectural details that it estimated that their mass percentage is around 4 percent of the total. Thus the concept that the "atoms" are a "minor?" part of a matter accumulation process being carried out by nature is overlooked. And their place in the hierarchy of the entities of matter is described but not organized until you get to "Universe Origination" theories where controversial details are discussed in more detail but not with much organizational clarity. For example in this section, Universe quantities of Atoms are talked about, when the question is evidently about nucleons. An explanation of the relative importance of "atoms" to the rest of the hierarchy might be helpful even in this "Atom" article. WFPM WFPM ( talk) 23:46, 18 October 2008 (UTC)
completely stable
What do you mean by completely stable in "The heaviest completely stable atom is that of lead-208"? Don't all atoms have a limited lifetime (although some are indeed quite long)? Randomblue ( talk) 18:46, 15 February 2008 (UTC)
- A stable element has at least one stable isotope. I added a wikilink.— RJH ( talk) 19:05, 15 February 2008 (UTC)
edit to intro
I removed the statement that the wave-particle duality was used to model the atom. Though it takes no stretch of the imagination that the two are related, the statement is vague. The sentence that remains is clear and sufficient. Also I removed the reference Harrison (2003). Without a title or a publisher this could be any of a hundred articles os books. -- V. ( talk) 05:03, 13 May 2008 (UTC)
- The Harrison reference refers to the book cited at the bottom of the article, in the "Book references" section: Harrison, Edward Robert (2003). Masks of the Universe: Changing Ideas on the Nature of the Cosmos. Cambridge University Press. ISBN 0521773512. -- Itub ( talk) 08:53, 13 May 2008 (UTC)
- I didn't do it, but my guess is that this method is used because it allows one to have several references to different pages of the same book without excessive duplication and without using "ibid" and such. For example, ref 1 might say "Harrison (2003), p 42" and ref. 2 might say "Harrison (2003), p 89". But I don't see any book citation in this article with page number, so the separation is not really justified IMO. -- Itub ( talk) 18:51, 13 May 2008 (UTC)
- The separation of the books into a list was requested as part of the FA candidate edits. A reviewer wanted a list that they could easily read through. (See Nigel, Wikipedia:Featured article candidates/Atom.) The reference is to the book list on this page and is unambiguous, so I restored it. If the issue is with the lack of page numbers, I can work on adding them in where appropriate.— RJH ( talk) 22:26, 13 May 2008 (UTC)
- There are other strange and vauge statements here. To describe the atom as the smallest particle in a chemical element is simply wrong. You might seek to say that you don't need to go any smaller to talk about chemistry, but the oxidation states of atoms depend on electron transfer, and this underpins much of chemistry. Really, an atom is not the smallest particle in anything. I dislkie the use of term electron cloud, also. I know it's due to Feynman, and I don't discourage its use in the correct circumstances, but it's an analogy to the behaviour of electrons. To say that an atom is a nucleus surrounded by an electron cloud is to assert something false about the fundamental nature of electrons.— Che Gannarelli ( talk) 09:58, 9 July 2008 (UTC)
- You raise some interesting points. The chemical element describes it as a type of atom; if it is false here, then that article would also seem to be in error. The term "electron cloud" seems like a useful and colorful descriptor for readers that are unfamiliar with quantum mechanics. The behavior is clarified down in the text and on the electron cloud page. I'm not sure I see the harm.— RJH ( talk) 17:54, 11 July 2008 (UTC)
- I agree that describing the atoms as, "the smallest particle that constitutes a chemical element", is incorrect, or at least misleading. All atoms are chemical elements and all chemical elements are atoms (they are not, of course, synonyms as different isotopes are different atoms but are still the same chemical element). I think there needs to be a balance between rigorous correctness and good intuitive explanation, but I don't think this sentence can be justified either way (someone not familiar with the concept of an atom is probably even less familiar with chemical elements). I propose something along the lines of:
| “ | The atom is the smallest building block of ordinary matter, that is the gases, liquids and solids that comprise the macroscopic world in which we live. | ” |
- The specific point this sentence is making is that although atoms are composed of smaller parts, these components cannot themselves form large-scale structures. In addition this sentence immediately makes the connection between atoms and the familiar materials around us. I realize a lot of work has gone into this article and I agree that it is worthy of featured article status, but I think this point needs to be addressed. I would appreciate any comments, alternative suggestions, or counter arguments.-- DJIndica ( talk) 13:07, 15 July 2008 (UTC)
- I wasn't necessarily agreeing that the statement was false; only that if it is false then it would have to be false in both locations. The statement about atoms forming the smallest particle of a chemical element seems fine to me. Your proposed revision, on the other hand, is a little more debatable. You are introducing the vague notion of "ordinary" matter. Unfortunately, much of matter in the Universe is plasma, so one might consider that ordinary matter.— RJH ( talk) 14:47, 15 July 2008 (UTC)
- I think DJ's proposed revision is excellent. It is short, snappy, to the point and accurate. The 'vagueness' of 'ordinary matter' is not the problem. At the start of any Wikipedia article, generalities are stated first. Of course, scientific articles in particular must be accurate, but using 'ordinary matter' in the sentence is clear enough. If need be, 'non-ordinary matter' can be mentioned later on to clarify the difference. Also, I don't think plasma counts as 'ordinary matter' because the Universe contains a lot of it (cf. dark matter is the most abundant type of matter in the universe, but one clearly cannot describe it as 'ordinary matter' !!!). DJ's revision is a huge improvement over the status quo and therefore I believe it should be incorporated into the article. Thanks. :) MP ( talk• contribs) 19:32, 15 July 2008 (UTC)
- It's a fair point that much of the matter in the Universe is not the "ordinary matter" this sentence refers to, but I think it is important, in the introduction at least, to make the point the much of the material we are familiar with from everyday life is made of atoms (and the rest is made of molecules which are themselves made of atoms). The first sentence needs to give an intuitive working definition of the atom for people not familiar with the concept; people knowledgable enough to debate whether baryonic matter inc. plasma or dark matter is more "ordinary" are not those the first sentence is aimed at. For a definition to be useful, it has to relate the new concept to something with which the reader is already familiar.-- DJIndica ( talk) 18:07, 16 July 2008 (UTC)
I had another go at clarifying this, as discribed at the bottom of the page, but it has been rather curtly reverted by User:Rracecarr, who declined to describe why he didn't "think this ... an improvement". The issue of inaccuracy is something I discuss at length at the bottom. Essentially, there are no independet atoms in covalent matter. The Britannica's description of an atom as the smallest uncharged unit of ordinary matter is about as simple as one can get while still being true. We need some intellegent discussion about this, rather than curt reverts. Che Gannarelli ( talk) 15:52, 17 July 2008 (UTC)
Encyclopaedia Britannica
The 9th edition of the EB has two very informative articles about the Atom and the theory of the force of universal gravitational attraction by Prof. G Clerk Maxwell (Atom & Attraction) that explain the development of the classical concepts related to this subject matter. They are very much worthy of review and consideration. WFPM WFPM ( talk) 23:18, 14 May 2008 (UTC)
- (O/T NOTE: I fixed the format of your additions by placing it at the end, removing the blank from the start of the lines and moving the comments to a separate line from the section title. Please take a look at Help:Contents/Editing Wikipedia for more information on how to edit wikipedia. Thank you.— RJH ( talk) 15:04, 15 May 2008 (UTC))
Proposed Czech image
An anonymous poster made the suggestion that we use the image at right as it uses a radial decay proportional to e-r for the electron cloud. Is there interest in using this image in place of the current helium atom diagram? I'm not sure whether the listed units are best for this purpose.— RJH ( talk) 17:15, 16 May 2008 (UTC)
Unsourced additions
The following additions, while probably correct, are also unsourced. To avoid a FAR, I would like to make sure that this page continues to satisfy the FA criteria.
- Of the chemical elements, 80 have one or more stable isotopes (elements 43, 61, and all elements numbered 83 or higher have no stable isotopes). As a rule, there is, for each atomic number (each element) only a handful of stable isotopes, the average being 3.4 stable isotopes per element which has any stable isotopes. Sixteen elements have only a single stable isotope, while the largest number of stable isotopes observed for any element is ten (for the element tin).
- Stability of isotopes is affected by the ratio of protons to neutrons, and also by presence of certain "magic numbers" of neutrons or protons which represent closed and filled quantum shells. Of the 269 known stable nuclides, only four have both an odd number of protons and odd number of neutrons are known: 2H, 6Li, 10B, 14N. Also, a very very long-lived radioactive odd-odd nuclides (40K, 50V, 138La, 180mTa) occur naturally. Most odd-odd nuclei are highly unstable with respect to beta decay, because the decay products are even-even, and are therefore more strongly bound, due to nuclear pairing effects.
It is often far easier to remove unsourced entries than it is to try and source somebody else's input, so I am hoping that citations are readily available for this material. Otherwise I think this should be pruned back. Thoughts?— RJH ( talk) 15:30, 23 May 2008 (UTC)
- The first paragraph can be sourced to the chart of the nuclides as it contains just trivial counting. The second one is more analytic but I'm sure can be based on any nuclear chemistry/physics book. -- Itub ( talk) 16:06, 23 May 2008 (UTC)
- Perhaps then the CRC Handbook of Chemistry & Physics will cover both? It looks like the Table of Isotopes includes suitable information, so I'll just use that unless somebody squawks. Thanks. The only problem remaining is the use of "very very long-lived", which conflicts with Wikipedia:MoS#Unnecessary_vagueness. (Very very long-lived is 100 years to me...)— RJH ( talk)
First paragraph
There's some kind of glitch in the last sentence of the first paragraph: the grammatical construction's wrong, the content seems redundant given the previous content, and there are a couple of stray characters at the end.
Paul Magnussen ( talk) 15:38, 9 July 2008 (UTC)
African?
| Smallest recognized division of a chemical element |
"Classification Smallest recognized division of a 'african'"
I'm not sure of what this refers to...'african' links to Africa, and although I am sure that quarks are a smaller division of Africa than an atom, I don't think this is what was intended to be discussed.
Buteo lineatus ( talk) 19:59, 9 July 2008 (UTC)
Issue has been resolved. Previously, "chemical element" was "african," but this has been remedied.
Buteo lineatus ( talk) 20:00, 9 July 2008 (UTC)
Problem in intro
- The sentence
taken from the intro, is totally wrong. The only thing that determine the type of an atom is the number of protons. Turiacus ( talk) 22:34, 9 July 2008 (UTC)A chemical element is determined entirely by the type of atom it contains, which in turn is determined by the number of protons, electrons and neutrons that constitute the atom.
- The new intro fixes that. It is much better than the old one. Great Job ! Turiacus ( talk) 20:08, 14 July 2008 (UTC)
- You're right. The text was incorrect. I'd added it myself, and it was sloppy. It was in fact my attempt at a re-wording to remove the following line, now restored: "The atom is the smallest particle that constitutes a chemical element". I still feel that statement to be simply incorrect (as was my own). A chemical element is no less constituted of smaller particles simply because they are arranged into atoms. Furthermore, as electrons are transferred and shared in bonding, it is inaccurate in an important way, and not merely a strict sense, to say that atoms are the smallest constituents of an element; they're not even the smallest important constituents.
Lastly, please note the above section on "First paragraph". This has all been discussed. Che Gannarelli ( talk) 00:57, 15 July 2008 (UTC)
- You're right. The text was incorrect. I'd added it myself, and it was sloppy. It was in fact my attempt at a re-wording to remove the following line, now restored: "The atom is the smallest particle that constitutes a chemical element". I still feel that statement to be simply incorrect (as was my own). A chemical element is no less constituted of smaller particles simply because they are arranged into atoms. Furthermore, as electrons are transferred and shared in bonding, it is inaccurate in an important way, and not merely a strict sense, to say that atoms are the smallest constituents of an element; they're not even the smallest important constituents.
- Sorry, but your logic seems flawed. The fact that both atoms and chemical elements are composed of still smaller particles is essentially irrelevant to the assertion. The exchange of electrons are important in chemical reactions, true, but this is not a statement about chemical reactions. Since you can not form a chemical element solely out of independent subatomic particles, it is a true statement that the atom is the smallest "particle" that constitutes a chemical element.— RJH ( talk) 14:41, 15 July 2008 (UTC)
- Ordinary matter is made of atoms, ions, or molecules. The second of these differs from the definition of an atom by the presence or absence of an electron, and the third, in the strong covalency regime, contains nothing that can be described as an individual atom. The statement is therefore palpably false; an atom is simply not the smallest particle of anything.
- Britannica has a nice definition in the opening of their article "atom (matter)", by the way, which may indicate that they were agonizing over the difficulties of this, also. They state that an atom is the smallest unit into which matter may be divided without the release of charged particles. It's a more complex statement than the first, certainly, but it is at least true. I can't readily come up with a simpler, and accurate, introduction to the idea, I'm afraid. Che Gannarelli ( talk) —Preceding comment was added at 15:20, 17 July 2008 (UTC)
- Consider: An atom is the smallest unit of an element that retains the chemical properties of that element. However, I do see an issue with ions -- they too might also qualify as "retaining chemical properties". Britannica's def also has an issue with ions. However, "The atom is the smallest building block of ordinary matter" just will not do - it would depend upon what the supposed "builder" chooses to start with. As far as I know, nobody has ever "built" an atom. Additionally, according to Big Bang theory, elements were "built up" out of smaller particles than atoms -- JimWae ( talk) 16:01, 17 July 2008 (UTC)
- Let's not over-complicate things. No reason to bring in cosmology. Just need a statement that's as simple as possible without being untrue. This really is a minefield! By the way, I chopped out the "solids, liquids and gases" bit because colloids, liquid crystals and so forth all fall within the pervue of this discussion. A common term for the type of "ordinary" matter we're referring to (excluding plasmas, and weirder things) is "atomic matter", but that's getting circular, and makes me want to put a cold towel on my head! —Preceding unsigned comment added by Cmsg ( talk • contribs) 16:09, 17 July 2008 (UTC)
- If we need cosmology, we need it - at least here on the talk pages. "The atom is the smallest possible uncharged unit of ordinary matter" also does not do the job. Neutrons would qualify as uncharged - so unless you stipulate what ordinary matter is - which gets back to elements - there's a problem. Stipulative definitions are the bane of scientific writing-- JimWae ( talk) 16:14, 17 July 2008 (UTC)
- If you want to go with the simplest definition and most accurate definition, an atom is a group of one or more protons, with zero or more bounded neutrons, and with zero or more bounded electrons.
A bunch of 7 protons and a neutron together is 8
7N7+
(Nitrogen-8 ionized 7 times, which would be unstable as hell). A bunch of 1 proton and 245 neutrons with a bounded electron would be 246
1H0
(again insanely unstable). Saner examples would be ionized tritium 3
1H1+
and unionized carbon-13 13
6H0
.
So for recap:
Z Number of protons Decides the "element" N Number of neutrons Decides the "isotope" of the element ne Number of electrons Decides the "ion" of the isotope of the element
Headbomb { ταλκ – WP Physics: PotW} 16:36, 17 July 2008 (UTC)
- Okay, let me have a go at stating what we're trying to convey. Then we'll find a form of words.
- 1) Ordinary matter, such as we all understand what we mean by that, is made up of either atoms, ions, or covalent molecules (possibly macroscopic), in which atoms have no individual identity (although ionic cores, comprising a nucleus and those electrons not involved in covalency, do retain their identity).
- 2) We want some kind of idea of smallest, because that's important to the definition of an atom, and yet getting a decent form of words for that is hard.
- 3) Whatever form of words is employed should seek to render your point about neutrons moot. "Ordinary matter" may not be divisible into atoms in the simplest sense of that description, but nuclei at least retain their identity. The real bonding process involve entities such as nuclei + electrons, or tightly-bound core ions + bonding-involved valence electrons.
- So. The idea of "smallest significant constituent" (in the sense that protons, neutrons and core electrons don't do anything independent of atomic cores) is important. Yet the idea of neutral charge is also significant, for an atom is an important entity for exactly the reason that while it may not be the fundamental unit of ordinary matter, if you take electrons off one of them, it becomes charged.
- I know how I'd describe this to a class: electrons are involved in bonding. An atom is defined in the non-chemical bonding limit. Start with the idea of an elemental gas, and state that it's not as simple as that when atoms coalesce. However, that's not a nice brief statement of what an atom is, suitable for this context.
- This is going to wind me up no end. Off to find some books on solid state physics and atomic physics. Hope somebody has a flash of genius. Che Gannarelli ( talk) 16:43, 17 July 2008 (UTC)
- Headbomb: you beat me to the chase. I think that the difficulty with simply listing the constituants of an atom, is that you're really saying nothing about physics. That's point 4), I suppose, and really the hardest of all: encapsulate at least a hint of why an atom is something physically important, and not just a way of counting subatomic particles. Che Gannarelli ( talk) 16:43, 17 July 2008 (UTC)
- I don't like the introductory sentence "The atom is the smallest possible uncharged unit of ordinary matter." I see several problems with this. For starters, what about neutrons? Are they not ordinary matter? Rracecarr ( talk) 17:46, 17 July 2008 (UTC)
- Nor do I. Consider: An atom is the smallest uncharged unit of an element that retains the chemical properties of that element -- possibly add and can participate in a chemical reaction. This eliminates ions. No need for "possible" which can easily be misread as meaning "possibly"-- JimWae ( talk) 17:55, 17 July 2008 (UTC)
- That seems good. I don't understand all the fuss about charge though. I always thought an ion is a charged atom--different number of electrons, but it's still an atom. Am I wrong about this definition? If so, is there a source showing that? If I'm not wrong, why not eliminate "uncharged", and go with An atom is the smallest unit of an element that retains the chemical properties of that element? Rracecarr ( talk) 17:59, 17 July 2008 (UTC)
- I like that very much, Rracecarr. Encapsulates nicely the idea that it's no longer that element, once you go below the level of the atom. Glad to not worry about this one any more. Che Gannarelli ( talk) 18:13, 17 July 2008 (UTC)
Proposed lead top-down re-write
- While it's better than what used to be there, I feel the prose is kinda weak and things seems to be introduced from the top-down rather than in a bottom-up approach. How about this version instead?
- While the atom was first conceptualized as an indivisible component of matter by early Indian and Greek philosophers, the modern understanding of the atom is that it is a small agglomeration of protons, neutrons, and electrons, ranging from a tenth of a nanometer to a few nanometers wide. A definition often encountered in introduction-level chemistry textbooks is that an atom is the smallest unit of a chemical element that retains the properties of that element. In the 17th and 18th centuries, chemists provided a physical basis for this idea by showing that certain substances could not be further broken down by chemical methods. During the late 19th and early 20th centuries, physicists discovered subatomic components and structure inside the atom (protons, neutrons, and electrons), thereby demonstrating that the 'atom' was not indivisible.
- Atoms have very small nuclei (about a few femtometers wide) made of protons and neutrons (also called nucleons), containing most of their mass (over 99.9% [1]), with electron clouds surrounding them. Atoms are classified into elements (depending on the number of protons), isotopes (depending on the number of neutrons), and ions (depending on the number of electrons). Atoms with too many or too few neutrons relative to the number of protons are unstable and subject to radioactive decay. [2]
- Electrons are responsible for most of the chemical and physical properties of atoms. They occupy sets of stable energy levels, or orbitals, and can move between them by absorbing or emitting photons that match the energy differences between the levels. Classical models of electron behaviour in atoms include the plum-pudding model, the Bohr model, and the Rutherford model (amongst others), while quantum models includes the Sommerfeld model, and the Schrodinger model (amongst others).
Headbomb { ταλκ – WP Physics: PotW} 02:06, 18 July 2008 (UTC)
- I think that's possibly a much better approach. A top down introduction is great, and the historical approach avoids the obvious danger of making simple but untrue statements. Che Gannarelli ( talk) 09:50, 18 July 2008 (UTC)
- Well bringing history after definition breaks the flow, which is also important. Headbomb { ταλκ – WP Physics: PotW} 19:44, 18 July 2008 (UTC)
- Per Wikipedia:Lead section, "The first paragraph needs to unambiguously define the topic for the reader." The proposed re-ordering moves that down to the second paragraph, leaving the reader dangling. Sorry, but I remain opposed, and I do not at all believe the flow is being broken.— RJH ( talk) 19:49, 18 July 2008 (UTC)
Okay I reworded things. How about now?
- I don't like starting with history. Leads should start with a definition, like RJHall says. Your proposed lead would work better for the article on History of atomic theory. -- Itub ( talk) 07:22, 19 July 2008 (UTC)
- But it does start with the definition... Headbomb { ταλκ – WP Physics: PotW} 11:38, 19 July 2008 (UTC)
The thing that fascinates me about encyclopaedic approaches to studies about physical entities is that our egocentric bias makes us miss the basic question, which what is the logic controlling the entities and the processes in the first place. And the logic of atoms is that nature has created a process of reaccumulating difuse matter that it have previously difused for some purpose in the first place, and what are the significant details of that accumulating process. So we start with difuse matter (or energy if you like), and then after phase 1 have a lot of nucleons (or energy packages], and these are accumuleted into atom package, and behind that we have a large package of matter (energy) waiting to gobble the whole thing all up (phase 3} and the question is, What are the controling factors in that process? And when are we going to get around to discussing that? WFPM WFPM ( talk) 13:34, 1 September 2008 (UTC) And so, if we examine the logic of the process of accumulation of matter (energy) we see that nature first created matter (energy), then created a reaccumulation process, which incidently increases factorially as a function of the number of units of mass (energy) versus the direct one to one relationship of mass-energy creation. Then how could nature fail to be able to reaccumulate the difuse matter (energy) and defeat the original purpose? And do you have an article on this? WFPM WFPM ( talk) 14:11, 1 September 2008 (UTC)
Oct 1 2006 version still has merit
http://en.wikipedia.org/?title=Atom&oldid=78921569 still has info that needs to be included in current version -- JimWae ( talk) 19:33, 17 July 2008 (UTC)
Comparison & contrast with molecules needs to come much sooner in current version(is it even included?) -- JimWae ( talk) 19:49, 17 July 2008 (UTC)
"In chemistry and physics, an atom ( Greek ἄτομος or átomos meaning "indivisible") is the smallest possible particle of a chemical element that retains its chemical properties. The word atom originally meant the smallest possible indivisible particle, but after the term came to have a specific meaning in science, atoms were found to be divisible and composed of smaller subatomic particles.
Most atoms are composed of three types of subatomic particles which govern their external properties:
- electrons, which have a negative charge and are the least massive of the three;
- protons, which have a positive charge and are about 1836 times more massive than electrons; and
- neutrons, which have no charge and are about 1839 times more massive than electrons.
Protons and neutrons make up a dense, massive atomic nucleus, and are collectively called nucleons. The electrons form the much larger electron cloud surrounding the nucleus.
Atoms can differ in the number of each of the subatomic particles they contain. Atoms of the same element have the same number of protons (called the atomic number). Within a single element, the number of neutrons may vary, determining the isotope of that element. The number of electrons associated with an atom is most easily changed, due to the lower energy of binding of electrons. The number of protons (and neutrons) in the atomic nucleus may also change, via nuclear fusion, nuclear fission or radioactive decay, in which case the atom is no longer the same element it was.
Atoms are electrically neutral if they have an equal number of protons and electrons. Atoms which have either a deficit or a surplus of electrons are called ions. Electrons that are furthest from the nucleus may be transferred to other nearby atoms or shared between atoms. By this mechanism atoms are able to bond into molecules and other types of chemical compounds like ionic and covalent network crystals.
Atoms ... are conserved in chemical reactions.
Atoms and molecules
For gases and certain molecular liquids and solids (such as water and sugar), molecules are the smallest division of matter which retains chemical properties; however, there are also many solids and liquids which are made of atoms, but do not contain discrete molecules (such as salts, rocks, and liquid and solid metals). Thus, while molecules are common on Earth (making up all of the atmosphere and most of the oceans), most of the mass of the Earth (much of the crust, and all of the mantle and core) is not made of identifiable molecules, but rather represents atomic matter in other arrangments, all of which which lack the particular type of small-scale order that is associated with molecules.
Most molecules are made up of multiple atoms; for example, a molecule of water is a combination of two hydrogen atoms and one oxygen atom. The term "molecule" in gases has been used as a synonym for the fundamental particles of the gas, whatever their structure. This definition results in a few types of gases (for example inert elements that do not form compounds, such as helium), having "molecules" consisting of only a single atom. "
Because understanding the difference between atoms & molecules it key to understanding both concepts. Understanding how atoms can combine is part of understanding what atoms are -- JimWae ( talk) 21:42, 17 July 2008 (UTC)
- This article is about atoms and that should be the primary focus. By earlier consensus during the review process, molecules and chemistry were de-emphasized in this article.— RJH ( talk) 21:59, 17 July 2008 (UTC)
Disagree about the "halting & clunky". That's your POV. I'd say pointed & specific - it takes time to talk about each topic rather than trying to cover too may topics at once (sweeping & vague) - and thereby even needing to repeat itself -- JimWae ( talk) 21:42, 17 July 2008 (UTC)
- Article has been through a Featured Article review where the lead was deemed acceptible by multiple reviewers. I reverted to the old form as your revisions are personal preference. Please discuss the change to gain consensus first. Thank you.— RJH ( talk) 21:55, 17 July 2008 (UTC)
AcceptAble or not, does not mean it does not need improvement. Correcting suggestion that proton numbers are key to formation of ions, and fixing omission of mention of nuclear reaction and molecules are not simply matters of personal preference. I also object to your suggestion that I have not been using tak page -- JimWae ( talk) 22:03, 17 July 2008 (UTC)
It also adds information about how atoms can vary & does NOT suggest that proton removal forms ions -- JimWae ( talk) 21:48, 17 July 2008 (UTC)
- Well thank you for bringing this issue to the talk page. The lead section has undergone many reviews and several re-writes, but improvement is always possible. However, I would much prefer that we first reach a consensus on whether large-scale changes are necessary. Note that the lead is intended to be a short summary of the main article. It is beneficial therefore to keep it brief, rather than extending it to cover every possible fact. It is also expected that the "prose is engaging, even brilliant, and of a professional standard". Certainly you could agree that this can be a challenging task.
- As for suggesting that proton removal forms ions, well the text only states that an ion is formed when the number of protons differs from the number of electrons.— RJH ( talk) 22:15, 17 July 2008 (UTC)
-- a vaugeness that can easily be fixed -- JimWae ( talk) 22:17, 17 July 2008 (UTC) Saying the number of protons determines the chemical element and the number of neutrons determines the isotope of that element is epistemologically backwards. We do not count the number of protons & neutrons to determine the element or the isotope. We have determined that atoms of the same element have the same number of protons AND determined they sometimes differ in the number of neutrons. -- JimWae ( talk) 22:17, 17 July 2008 (UTC)
- I just want to point out that the following definition was good enough for IUPAC for its glossary of chemical terminology, also known as the Gold book: "Smallest particle still characterizing a chemical element. It consists of a nucleus of a positive charge (Z is the proton number and e the elementary charge) carrying almost all its mass (more than 99.9%) and Z electrons determining its size." [1] -- Itub ( talk) 22:30, 17 July 2008 (UTC)
- Reply:
- An atom is classified according to its number of protons and neutrons: the number of protons determines the chemical element and the number of neutrons determines the isotope of that element.
- versus
- Atoms of the same chemical element have the same number of protons; atoms of the same element that vary in the number of neutrons are called isotopes of that element.
- To me the second sentence has lost the context of what it means to be an isotope; it is ambiguous. The statement implies the number of neutrons are varying, whereas they are not. A fixed number of neutrons for a given element corresponds to a particular isotope. The first sentence makes that clear.—
RJH (
talk) 22:39, 17 July 2008 (UTC)
- Atoms can lose or gain protons in nuclear reactions, thereby becoming a different element with a different atomic number. Nuclear reactions also involve a change in the number of neutrons.
- Discussion of radioactivity in the lead should be consolidated. Placing this at the end of the first scatters the discussion across separate paragraphs. Thanks.— RJH ( talk) 22:39, 17 July 2008 (UTC)
edit conflict--
- For a group of scientists "characterizing" might be fine, but for the general public it needs to be "unpacked". This is a general public encyclopedia
- If this is a general issue with wikipedia, then it's probably off topic for this talk page. Wikipedia pages aren't written for a specific audience education level. (See
photon for example.) Generally my impression has been that a page only needs to be internally consistent regarding education level, while striving to
explain jargon particular to the speciality.—
RJH (
talk) 18:54, 18 July 2008 (UTC)
- Characterizing may sound like a scary word, but at least is not misleading like saying that an atom "has the chemical properties" of an element. Elements have many chemical properties that are not displayed by single atoms. If a simple verb is desired, one could say that "an atom is the smallest particle that can be said to be a chemical element". -- Itub ( talk) 07:17, 19 July 2008 (UTC)
- If this is a general issue with wikipedia, then it's probably off topic for this talk page. Wikipedia pages aren't written for a specific audience education level. (See
photon for example.) Generally my impression has been that a page only needs to be internally consistent regarding education level, while striving to
explain jargon particular to the speciality.—
RJH (
talk) 18:54, 18 July 2008 (UTC)
- If the article does not give at least some brief, detailed explanation of how atoms combine, it has not told the story of atoms. The only current explanaton is buried almost half-way down in the article as "By this mechanism, atoms are able to bond into molecules and other types of chemical compounds like ionic and covalent network crystals". This is assuming too much about what the general reader already knows on the topic.
- As molecules depend on the element involved, the consensus was to rely on the chemical element page for most of that information. For this reason, various details were pared back to just focus on the atom as an issolated entity. Yes, perhaps it may make sense to remove (or alter) the sentence you mention as well.— RJH ( talk)
- In general, I find scientific writing sometimes suffers from the delusion that the author is witnessing the "creation" of the universe & is involved in the "planning" of the next step. Thus we get terms like "building block" & phrases like "the number of protons determines the element" - rather than acknowleding that science is a discovery, not a building project. --
JimWae (
talk) 22:42, 17 July 2008 (UTC)
- I'll assume you're writing in good faith and not really trying to insult us here. It's important to keep in mind that this is an encyclopedia, not an article in a general public science magazine. We don't need to popularize science here; just present the facts in a transparent and readable manner.— RJH ( talk)
- OK, VARY might be ambiguous too. Instead of atoms of the same element that vary in the number of neutrons are called isotopes of that element we could say atoms of the same element which differ in the number of neutrons are called isotopes OR atoms with the same number of protons but a different number of neutrons are called isotopes.
Proposed change to lead: discussion of radioactivity
Per the discussion with JimWae above, I think it makes sense to expand upon the following sentence in the lead:
- In atoms with too many or too few neutrons relative to the number of protons, the nucleus is unstable and subject to radioactive decay.
JimWae's wording could be adopted by inserting the following text after the above sentence:
- As a result, the nucleus can gain or lose or gain protons, thereby becoming a different element with a different atomic number. Nuclear reactions can also involve a change in the number of neutrons.
Does anybody object to this idea, or want to present some better wording? Thank you.— RJH ( talk) 14:41, 23 July 2008 (UTC)
- "Too many or too few" suggests there is a "Goldilocks number" that is "just right". Many elements are unstable no matter how many neutrons they have --
JimWae (
talk) 18:26, 23 July 2008 (UTC)
- True. The first sentence of the 'Radioactive decay' section would probably work better as a summary.—
RJH (
talk) 15:29, 24 July 2008 (UTC)
- Then again, it shouldn't be necessary to repeat information presented earlier in the lead. This may be sufficient:
- Each element has at least one isotope with unstable nuclei that can undergo radioactive decay. This can result in a transmutation that changes the number of protons or neutrons in a nucleus.
- Will this work?— RJH ( talk) 14:54, 27 July 2008 (UTC)
- Then again, it shouldn't be necessary to repeat information presented earlier in the lead. This may be sufficient:
- True. The first sentence of the 'Radioactive decay' section would probably work better as a summary.—
RJH (
talk) 15:29, 24 July 2008 (UTC)
g vs. kg
Why are the particles masses given in "g" instead of "kg" as in the International System of Units? I think that particles masses are given in kg in practically all textbooks. Eklipse ( talk) 15:24, 24 July 2008 (UTC)
- Good question. I'm not really sure why. They needed to be made consistent at some point and they ended up as grams. The CRC Handbook uses kg, so that would seem like the logical choice.— RJH ( talk) 15:35, 24 July 2008 (UTC)
- Kilograms should be used. Headbomb { ταλκ – WP Physics: PotW} 15:52, 24 July 2008 (UTC)
I created this template so people could quickly-browse atomic models. The skeleton is there, but little meat. Help is appreciated. Headbomb { ταλκ – WP Physics: PotW} 01:29, 1 September 2008 (UTC)
- I'd like to suggest using the {{ navbox}} format instead. It can include the same information, but you only have to expand the table rather than each row.— RJH ( talk) 17:06, 8 September 2008 (UTC)
How about looking at the picture of a real physical nuclear model, light in Talk:Nuclear model.WFPM WFPM ( talk) 20:12, 8 September 2008 (UTC)
I changed it into a navbox and expanded things a bit. Headbomb { ταλκ – WP Physics: PotW} 22:10, 8 September 2008 (UTC)
"Physicists Create Millimeter-sized 'Bohr Atom'"
The article formerly in the "Size" section formerly included the text "The largest atoms observed, as defined by the distance of the outermost electron from the nucleus, have a diameter approaching one millimeter for several orbits of the outermost electron. [3] " This was deleted 16:48 on 14 August by BenRG with the comment "The article says they manually put an electron in a large semiclassical orbit from which it almost immediately decayed." The extremes of physics are not something to delete and ignore. The article, in Science Daily, from July 1, 2008 [2] is titled "Physicists Create Millimeter-sized 'Bohr Atom'." It says they intentionally created a Bohr-like atom with an electron in a defined orbit, (not "manually" since their hands are large and electrons are small) but by adding energy to the outermost electron. It is still an atom even if the outermost electron is at an unusually high energy level. Many substances in nuclear physics are "artificially" created and last for very brief periods before changing or decaying. The article said "Using laser beams and electric fields, Rice physicists coaxed a point-like, "localized" electron to orbit far from the nucleus of a potassium atom." Scientists from Oak Ridge National Laboratory and Vienna University of Technology collaborated with a team from Rice University led by Barry Dunning, and it was published in Physical Review Letters. It is said to have applications "in next-generation computers and in the study of classical and quantum chaos." I feel that this demonstration should be included in the article, and welcome any suggestions for how to make it as clear as possible. An atom with a diameter of about a millimeter and a localized electron in a well defined orbit is atypical but worth noting. Edison ( talk) 15:37, 5 September 2008 (UTC)
- The abstract at Physical Review Letters can be seen at [3] for "Realization of Localized Bohr-Like Wave Packets," by J. J. Mestayer, B. Wyker, J. C. Lancaster, F. B. Dunning, C. O. Reinhold, S. Yoshida, and J. Burgdörfer, Phys. Rev. Lett. 100, 243004 (2008) . It says "Although these wave packets slowly dephase and eventually lose their localization, their motion can be monitored over several orbital periods. These wave packets represent the closest analog yet achieved to the original Bohr model of the hydrogen atom, i.e., an electron in a circular classical orbit around the nucleus. The possible extension of the approach to create “planetary atoms” in highly correlated stable multiply excited states is discussed." Edison ( talk) 16:29, 5 September 2008 (UTC)
- I've just taken a look at the paper itself, and it says that the radius of the orbits they created is circa 5 µm, not "approaching one millimeter" as the Science Daily article said. I assume this is a mistake by Science Daily or by whoever wrote the Rice University press release. Also, the paper makes it clear that the goal of the work is to create semiclassical orbits and not large-radius orbits as such. So I'm not sure this is actually the largest orbital radius ever achieved, since maximizing the radius wasn't their goal.
- My edit summary also said that the sentence I deleted "seems more misleading than informative", and I still feel that way. Discussions of atomic radii normally refer to ground-state atoms and ions. Once you admit excited states, I think the only real limit to atomic size is what one considers reasonable to call an "atom". I could place a proton and an electron a centimeter apart in an evacuated chamber and declare them to be an excited state of hydrogen provided they eventually collapsed into ground-state hydrogen. But I'd be more likely to describe that as a proton and an electron merging into an atom or as an H+ ion capturing an electron. I guess I wouldn't object to something along the lines of "If excited states are considered there is no upper limit to the size of an atom in principle. Electrons have been placed into semiclassical orbits with a radius of 5 µm in the lab, over 10,000 times larger than a typical atomic radius.[cite paper]" -- BenRG ( talk) 18:26, 5 September 2008 (UTC)
I thought that the Bohr orbits were supposed to be stable and not involve energy loss radiation. The energy loss radiation was supposed to occur "per saltum" as the orbit fell from one level to another. WFPM WFPM ( talk) 16:57, 6 September 2008 (UTC)
The man made black hole?
Tomorrow they will do some experiments on the subatomic level, is that worth it, is that on the level, why waste money, what kind of black hole can be created or r they overdoing things big time? I dont thing force is great enough to produce such effects?! Who cares what happened in first second, i think there were many big bangs! —Preceding unsigned comment added by Ornamentalone ( talk • contribs) 22:22, 9 September 2008 (UTC)
- Atoms are not black holes nor are they relevant to the LHC. You are off topic.
Eeekster (
talk) 22:05, 16 September 2008 (UTC)
- May be you should have more respect and learn how to read, i wrote on black hole experiments, subatomic level, as all matter must consist of atoms, i only concentrated on black hole experiment! —Preceding
unsigned comment added by
Ornamentalone (
talk •
contribs) 22:23, 16 September 2008 (UTC)
- Not all matter consists of atoms. Protons (what will be used in the LHC) are matter and don't consist of atoms. Nor do black holes consist of atoms. Take this discussion to the LHC pages, not here. S B H arris 00:24, 17 September 2008 (UTC)
- If you really want to discuss the possibilty of black holes from the LHC, the best place would be Safety of the Large Hadron Collider. Eeekster ( talk) 00:34, 17 September 2008 (UTC)
- May be you should have more respect and learn how to read, i wrote on black hole experiments, subatomic level, as all matter must consist of atoms, i only concentrated on black hole experiment! —Preceding
unsigned comment added by
Ornamentalone (
talk •
contribs) 22:23, 16 September 2008 (UTC)
WHAT AN ATOM IS.....in a 6th graders mind :)
An atom is everytihg in the world, air,liquid and,gas. —Preceding unsigned comment added by 70.179.87.249 ( talk) 22:10, 8 October 2008 (UTC)
- ==That's the way kids think. We've got 5 different things, all similar, and it's an accomplishment that he came up with 5 different things. And so he is learning. So he has the right attitude and how can we help him with that? I once heard an interview on the radio with a kid who had taken an interest in the planets and knew more about them than I did (dubious criteria). I think that helping organize the information is important. I have memorized the periodic table and it has helped a lot. WFPM
WFPM (
talk) 22:06, 19 October 2008 (UTC)
- Well, unfortunately, there is a space limit on these articles, so information on chemical element and state of matter ends up on other pages; otherwise the article would need to be as long as a book to cover everything. We do have articles on air, liquid and gas, so probably those would be good starting points for somebody seeking information. We also have a simple english article on the atom that could be expanded. Thanks.— RJH ( talk) 15:28, 20 October 2008 (UTC)
- ==How about a simple article about the periodic table, and how it could be better understood if people didn't insist on making it so complicated, Asimov did it. WFPM WFPM ( talk) 17:53, 20 October 2008 (UTC) But he's not a man of few words. WFPM WFPM ( talk) 17:56, 20 October 2008 (UTC)
- ==Okay so there's a simple periodic table article. But it didn't recognize me as WFPM when I tried to log in and I made 2 contributions as ?. And I dont dare suggest how many complications are buried in the standard periodic table. WFPM WFPM ( talk) 18:27, 20 October 2008 (UTC)
- == I'll try something and see what happens. And what's —? WFPM WFPM ( talk) 03:12, 21 October 2008 (UTC)
Like charges repel
Like charges repel. What are the forces that keep the protons with similar charges are so close to each other in nucleus of an atom. The distance among electrons and protons are greater as compared to the distance among protons in nucleus. 96.52.178.55 ( talk) 16:46, 24 October 2008 (UTC) Khattak
- Talk pages of articles are for suggesting improvement to the article, for other questions ask WP:Reference desk. BTW, see nuclear force. -- Army 1987 (t — c) 16:57, 24 October 2008 (UTC)
Comment from an outsider
I have just now made some minor changes and have noticed other points that need expert attention. For example, there is a difference between electrons and orbitals that isn't obvious in places in this article. Also, the level of sophistication of the text varies quite a bit. Finally, chemists would argue about the article's first sentence since the stable macroscopic manifestations of many elements are not carried over to the atomic level. [Example: Elemental nitrogen is N2, so an atom (N) of this element doesn't behave like a mole of nitrogen in the room where you are now reading this sentence.] I'm not a regular contributor to this long article, so take these comments for what they are worth. - Astrochemist ( talk) 16:44, 25 October 2008 (UTC)
- Thank you. Yes the page has undergone some revisions since it became FA, and it might need some editing for use by a general-audience. Some of the contributors may assume too much knowledge on the part of the typical reader, for example. I also see your point about the first sentence. It has been rehashed several times, and it looks like it still isn't satisfactory. That definitely needs to be debated some more in order to get it just right.— RJH ( talk) 17:11, 25 October 2008 (UTC)
Definition of atom
Here are some of the definitions I found on the net:
- The smallest particle of an element which can enter into a chemical combination.
- Half baked definition. The "smallest" particle of an element would be electrons/protons/neutrons since elements are made of them. I get what they are trying to argue, but the wording is very poor.
- The smallest particle of matter that can exist in combination with other atoms, building up or constituting molecules.
- Better definition, but not quite there.Relies on the concept of matter, which is often (though not always) ill-defined. It is also a circular definition.
- An atom is the smallest unit of chemical structure, and no chemical change can alter it.
- Flat out wrong, as you can ionize or exite atoms and that is certainly a change.
- An atom is the smallest irreducible constituent of a chemical system.
- Best "layman definition", but not one that is true. Atoms can be reduced to electrons, proton and neutrons, the later two can be broken into up and down quarks.
- The smallest unit into which matter can be divided without the release of charged particles. (Encyc. Brit.)
- Horribly worded and the fact that atoms regularly undergo beta decay means this definition is not true.
- The smallest particle of an element that can not be broken down without losing its properties.
Other definitions define the atom in terms of the element. However, the element is in turn defined in terms of the atom, which seems to result in a circular definition.
- Ions because problematic under this definition, as removing an electron changes the properties of that element (say Iron), even thought it remains the same element (Iron, albeit charged).
- Examples:
- An atom is the smallest particle differentiable as a certain chemical element.
- The smallest unit of a chemical element that can exist.
- The atom is the smallest particle that constitutes a chemical element.
Here is the current definition:
- The atom is the smallest unit of an element that retains the chemical properties of that element.
Here is my first attempt at a definition to address Astrochemist's concern:
- The atom is the smallest component of matter that can not be further broken down by chemical means, and it forms the basic unit of a chemical element.
although I'm not sure if that would include the hydrogen ion. Does anybody have a better definition?— RJH ( talk) 20:56, 27 October 2008 (UTC)
- The real definition an atom is "any bound state of protons and electrons", with a possible extension to include ionized hydrogen. You can generalize it to any bound state of baryons and leptons ( exotic atoms are those not made of protons/electrons). This is a rather complicated definition for the reader. Simply saying an atom is a pack of bunched up protons/neutrons(optional)/electrons would be both technically accurate and accessible. Headbomb { ταλκ – WP Physics: PotW} 04:34, 29 October 2008 (UTC)
- Yes that would be satisfactory. Then the concept of molecules could be introduced as a pack of bunched atoms. Headbomb { ταλκ – WP Physics: PotW} 18:15, 29 October 2008 (UTC)
Okay I reworked the lead paragraph it a little:
- An atom is a basic unit of matter consisting of a central nucleus surrounded by a cloud of negatively charged electrons. The dense nucleus contains positively charged protons and electrically neutral neutrons. The electrons in an atom are bound to the nucleus by the electromagnetic force. Likewise, a group of atoms can remain held together by the electromagnetic force, forming a molecule. When the number of protons in the nucleus equals the number of electrons, the atom is electrically neutral; otherwise it is an ion and has a net positive or negative charge. An atom is classified according to its number of protons and neutrons: the number of protons determines the chemical element and the number of neutrons determines the isotope of that element.
Does anybody see any further issues with this? Thanks.— RJH ( talk) 18:49, 29 October 2008 (UTC)
- I would write "often contains electrically neutral neutrons" as neutrons are not a requirement (or at least something to that effect). I have minor reservations about "basic unit of matter", but to correct that would make the lead waaaaaaaaaay too technical (matter = stuff made of elementary fermions, to the basic units of matter are quarks and leptons, rather than atoms). Wikilinking matter would be sufficient IMO.
- How about
An atom is a basic unit of matter consisting of a dense, central nucleus surrounded by a cloud of negatively charged electrons. The atomic nucleus contains positively charged protons and often, but not always, electrically neutral neutrons. The electrons of an atom are bound to the nucleus by the electromagnetic force. Likewise, a group of atoms can remain bound to each other, forming a molecule. An atom containing an equal number of protons and electrons is electrically neutral, otherwise it has a positive or negative charge and is an ion.
An atom is classified according to the number of protons and neutrons in its nucleus: the number of protons determines the chemical element, and the number of neutrons determine the isotope of the element. -
Headbomb {
ταλκ
κοντριβς –
WP Physics} 19:45, 29 October 2008 (UTC)
- Given that no nucleus contains several protons but no neutrons, I'd rather say "The atomic nucleus contains positively charged protons and electrically neutral neutrons (except for the hydrogen-1 nucleus, which is a single proton)." Except for this minor point, your proposal sounds OK to me. -- Army 1987 (t — c) 10:24, 30 October 2008 (UTC)
- How about
- Well, unfortunately, when you go there you also have to correct for the plural form of protons. My preference is to go with the least distracting form and then clarify it in the body of the article. How about, "The atomic nucleus contains a mix of positively charged protons and electrically neutral neutrons." I.e. leave it vague, as it is covered by the end of the paragraph. We could add a sentence at the end saying that hydrogen-1 is the only nucleus with no neutrons.
- I'm not real fond of the paragraph break; the article needs to follow Wikipedia:LEAD to avoid FAR, so I think it should be kept to four paragraphs.— RJH ( talk) 17:19, 30 October 2008 (UTC)
- Well since we're describing what a nucleus is rather than defining it, sure, as long as hydrogen-1 is mentionned as the only stable atom/isotope without neutrons. Remove the paragraph break if you want. So something like
Headbomb { ταλκ κοντριβς – WP Physics} 19:47, 30 October 2008 (UTC)An atom is a basic unit of matter consisting of a dense, central nucleus surrounded by a cloud of negatively charged electrons. The atomic nucleus contains a mix of positively charged protons and electrically neutral neutrons (except in the case of Hydrogen-1, which is the only stable isotope with zero neutrons). The electrons of an atom are bound to the nucleus by the electromagnetic force. Likewise, a group of atoms can remain bound to each other, forming a molecule. An atom containing an equal number of protons and electrons is electrically neutral, otherwise it has a positive or negative charge and is an ion. An atom is classified according to the number of protons and neutrons in its nucleus: the number of protons determines the chemical element, and the number of neutrons determine the isotope of the element.
- Well since we're describing what a nucleus is rather than defining it, sure, as long as hydrogen-1 is mentionned as the only stable atom/isotope without neutrons. Remove the paragraph break if you want. So something like
Rare Forms
"For unknown reasons, antimatter particles are rare in the universe, hence, no antimatter atoms have been discovered in nature.[130][131]"
- I would suggest re-wording this sentence (or removing it entirely?). The main reason for the rarity of antimatter particles in nature is their instant annihilation when they come into contact with matter. There are two referenced articles after the sentence that say this. 86.131.69.5 ( talk) 22:45, 28 November 2008 (UTC)
- Unfortunately, using the same logic I can say that matter particles are relatively rare in the universe because 99% were wiped out by antimatter. The sentence is a reference to baryogenesis, which remains incompletely explained and is therefore unknown. Perhaps it can be re-worded into a more satisfactory form?— RJH ( talk) 22:59, 28 November 2008 (UTC)
- However theoretical I can't accept that the reason for the rarity of antimatter is unknown. How about re-wording it as follows, or similar? "No naturally occuring antimatter particles have been discovered as each annihilates when it comes into contact with its corresponding particle.[130][131] The baryogenesis theories offer incomplete explanations." It maintains the theoretical aspects, gives a reason for antimatters rarity and provides the reader with links to related topics. Any ideas? Kae1is ( talk) 17:41, 29 November 2008 (UTC)
- That's a hypothesis and we shouldn't declare things to be settled just because the idea makes sense. The reason for the matter-antimatter asymmetry is unknown, or at the very least, not firmly established. If you can't accept that, then you have a problem with the state of science in general, because there's a lot we don't know. Headbomb { ταλκ κοντριβς – WP Physics} 19:41, 29 November 2008 (UTC)
- I would simply mention that relative to matter, antimatter particles a relatively rare, and that the reason for this rarity is "not yet fully understood" and point to the baryogenesis article. I don't have a specific wording to propose however. Headbomb { ταλκ κοντριβς – WP Physics} 20:44, 29 November 2008 (UTC)
- The initial reason for the rarity is not fully understood, but the current reason, annihilation, is fairly self evident, which was my query in the first place. There was no distinction made. How about:
- "No naturally occuring antimatter particles have been discovered as each annihilates when it comes into contact with its corresponding particle.[130][131] The first causes of this rarity is not yet fully understood, however, the baryogenesis theories may offer an explanation." Kae1is ( talk) 14:17, 30 November 2008 (UTC)
- In retrospect your statement is not correct: naturally occuring antimatter particles are created continually from radioactive decay. Also see proton-proton chain reaction, which relies on the production of positrons.— RJH ( talk) 19:47, 30 November 2008 (UTC)
- In that case the current sentence is also incorrect and should be changed. If it's worded, "Naturally occuring antimatter particles are rare as each..." 86.131.69.5 ( talk) 21:21, 30 November 2008 (UTC)
- de-indent: No, the current statement is correct. It asserts scarcity, rather than lack of existence.— RJH ( talk) 19:54, 1 December 2008 (UTC)
atoms and molecules
Atoms are the basics of life. Everything we know is made of atoms. Do you know what an atom is? I don't. I do you know what a molecule is? It is a bunch of atoms put together just like our bodies. —Preceding unsigned comment added by Hayden014 ( talk • contribs) 13:55, 18 December 2008 (UTC)
- Didn't they warn you to stay away from the purple mushroom? ;-) — RJH ( talk) 19:30, 18 December 2008 (UTC)
See also
a link to Intel Atom requested. —Preceding unsigned comment added by 71.206.65.120 ( talk) 00:34, 17 January 2009 (UTC)
- It's on Atom (disambiguation). Vsmith ( talk) 19:08, 20 January 2009 (UTC)
Alternative Atom Model
To date, although the theories on atom model are said quite established, there are still few unsolved questionable problems, such as the non-linearity in Moseley X-ray plot for higher atomic mass number, the 'V' shape of H2O molecule, the revolution of electrons in atom and etcetera. An alternative atom model is proposed for alternative understanding. More detail explanation is presented in an alternative atom model Kongkokhaw ( talk) 16:30, 20 January 2009 (UTC)
- Has this been published in a peer reviewed journal? If not, see WP:FRINGE. Vsmith ( talk) 19:04, 20 January 2009 (UTC)
Spaces Between Atoms
If everything is made of matter, of which the building blocks are atoms, of which the building blocks are fermions ( quarks and leptons) and gauge bosons and maybe Faddeev-Popov ghosts, of which the building blocks are what? ( Strings?). Still, we do not tackle what is in-between the fundamental particles? How is "empty space" accounted for? Are we currently just defining this into being (or some kind of non-being) and not talking about it? andersole ( talk) 00:10, 21 January 2009 (UTC)
- If you think about matter as waves (per the wave-particle duality), then it is not clear there actually is empty space between particles. An interesting book that tackles this question is Nothingness, The Science of Empty Space by Henning Genz (2001). See also the vacuum state, virtual particles and vacuum energy articles. But yes there does seem to be a level below which the fundamental nature of space remain unclear. I've seen some articles that talk about "atoms of space" for example.— RJH ( talk) 23:05, 21 January 2009 (UTC)
Atoms
Element consists of atoms and atom consists of protons, neutrons, electrons depend upon the element. What are their masses made up of? Means what kind of material (matter) is in their masses? 96.52.178.55 ( talk) 02:33, 5 March 2009 (UTC) Khattak
- I think that's the big 6 billion Euro question. See for example: Higgs boson and Large Hadron Collider.— RJH ( talk) 19:25, 5 March 2009 (UTC)
Working of STM on this page
In the Identification section, there is a line "Electrons tunnel through the vacuum between two planar metal electrodes, on each of which is an adsorbed atom, providing a tunneling-current density that can be measured.". I wonder why it is explicitly mentioned that there should be atoms adsorbed to the metal electrodes. What kind of atoms are they referring to? By the way, in STM, one measures a tunneling current, not a tunneling current density. And the metal electrodes are not planar, one of them is a tip and certainly not a plane. Erwin ( talk) 11:03, 12 March 2009 (UTC)
- I have no idea. The nobel lecture cited only mentions using a gold tip to measure a gold surface, and that only a limited set of materials are suitable for the tip. The STM article mentions tungsten, platinum-iridium and gold as materials used for tips.— RJH ( talk) 21:15, 12 March 2009 (UTC)
Reference to dark matter
In the "Origin and current state" section, there is a line "Atoms form about 4% of the total mass density of the observable universe." This is not true, atoms form 4% of the total energy density of the observable universe, also according to the website which is given as reference. Erwin ( talk) 11:05, 12 March 2009 (UTC)
- Fair enough. I wonder if it will turn out that mass-energy equivalence doesn't hold for dark matter or dark energy?— RJH ( talk) 20:58, 12 March 2009 (UTC)
- "Mass density" and "energy density" have the same meaning in this context. I don't object to the change, but it wasn't wrong before. -- BenRG ( talk) 23:02, 14 March 2009 (UTC)
Etymology of "Atom"
We need an etymology of the word "Atom" Faro0485 ( talk) 21:35, 9 April 2009 (UTC)
- Did you read the article before posting? The origin of the word is already mentioned in the lead, and is covered in the history. That seems more than sufficient. Thanks.— RJH ( talk) 15:57, 10 April 2009 (UTC)
Link to Plutonium
Towards the end of the article there are references to Plutonium and Neptunium. To the Neptunium article a link is present, but not to the Plutonium article. Unfortunately I cannot edit it myself due to the semi-protected status. Arjan Mels ( talk) 18:21, 16 April 2009 (UTC)
Smallest Atom?
Why does the article under size state, "Consequently, the smallest atom is helium with a radius of 32 pm, while one of the largest is caesium at 225 pm."
Yet the page on Hydrogen says it has a radius of 25 pm. —Preceding unsigned comment added by 74.197.113.33 ( talk) 05:51, 4 May 2009 (UTC)
- It may be because there are multiple different atomic radii, depending on how it is computed. This is discussed on the atomic radius article— RJH ( talk) 15:04, 4 May 2009 (UTC)
Question about antimatter
"The atom is a basic unit of matter that consists of a dense central nucleus surrounded by a cloud of negatively charged electrons." Since an atom is matter, may a basic unit of antimatter also be called an atom, albeit with a qualifier? If so, the atom would not necessarily have a nucleus surrounded by negatively charged electrons, it would be surrounded by positively charged positrons. SpoilingRoot ( talk) 07:21, 24 June 2010 (UTC)
- Talk pages are not for general discussion of the topic. See antiparticle generally, and antihydrogen for the simplest explanation and example. If you need more information, try the Wikipedia: Reference Desk/Science Reference Desk. Somnambulent ( talk) 20:27, 3 September 2010 (UTC)
Start re the subject matter
Suggest that starting out the article by jumping to the subject of antimatter is immediately leading the discussion away from the subject. Which should start by the concept that the Atom is made up of an accumulation of one or more nucleons and then go on from there. And there are people who have the periodic table memorized. WFPM ( talk) 14:12, 25 September 2010 (UTC)
Assumed knowledge
The following edited text:
- Of the 250 known stable nuclides, only four have both an odd number of protons and odd number of neutrons: 2H, 6Li, 10B and 14N. Also, only four naturally occurring, radioactive odd-odd nuclides have a half-life over a billion years: 40K, 50V, 138La and 180mTa.
assumes familiarity with the periodic table on the part of the the reader. My suspicion is that most readers will not have the table memorized, and so this fails the WP:JARGON policy. Is there anybody who objects to me spelling these out, as is done in the remainder of the article?— RJH ( talk) 21:09, 26 May 2009 (UTC)
- Go for it. I'm certain it was done out of laziness and wanting to minimize the length. S B H arris 21:40, 2 June 2009 (UTC)
History section
My sense on the History section is that it should be a brief, WP:Summary style description of the key concepts in the Atomic theory article. It has show a tendency to grow over time, sometimes adding in what appear to be secondary details. For this reason I did some trimming back. Hopefully this didn't offend anybody. The details can always be expanded upon in the main article. I do think that the atomic theory article is in need of improvement and better referencing. Thank you.— RJH ( talk) 17:30, 2 August 2009 (UTC)
If you want to know about the history of knowledge about atoms, you should read Maxwell's 14 page article about "atom" in the 9th edition of the Encyclopedia Britannica. WFPM ( talk) 22:15, 27 April 2010 (UTC)
not to split atoms
Basically agree with RJH's comment, but some key concepts require quite obvious names and dates, presented in quite obvious manners. for example, the originator of modern atomic theory (Dalton, 1803), needs to be boldly credited in this basic article introducing atoms and atomic theory. in this spirit, section 1.2 is entitled as obviously as possible:
1.2 John Dalton, originator of modern atomic theory (1803)
similarly, otto hahn and lise meitner's mentions in section 1.4 should be slightly expanded to include dates and details (e.g., german anti-semitism decreased german physicists):
"In 1938, the German chemist Otto Hahn (a student of Rutherford's) directed neutrons onto uranium atoms expecting to create transuranic elements. Instead, his chemical tests showed barium, a lighter element, as a product. In 1939, his physics colleague Lise Meitner (living in Sweden to avoid anti-Jewish persecution in Germany) and her nephew Otto Frisch verified Hahn's result were the first experimental nuclear fission. In 1944, Hahn received the Nobel prize in chemistry in which, despite the efforts of Hahn, Meitner and Frisch's contributions were not recognized."
ciao--
diremarc
(talk) 21:34, 9 August 2009 (UTC)
- A the section is about more than John Dalton, your title change was misleading. In addition this article is about atoms, rather than the history of anti-semitism, so I think it should be focused on the former. The details should definitely be covered in the main history article on the subject, but here I think it is wandering off topic.— RJH ( talk) 16:49, 10 August 2009 (UTC)
A significant thing to note in this matter is how the lead and follow situation in science concepts wandered around all over Europe, with occasional inputs from other areas. See Gustave Le Bon's "The evolution of matter". WFPM ( talk) 15:11, 28 April 2010 (UTC)
masses are a bit off-
Electron - 1 e .00911 e-28 kg Protons - 1836 e 16.72596 e-28 kg + ----- 16.73507 e-28 kg Nuetrons - 1839 e 16.75329 e-28 kg - ----- -.01822 e-28 kg 1n(0) -> 1p(0) + 1e(0) + E(due to changes in momentum [KE] and position [PE]of the system) (0) is the ground charge state. For protons it is a + charge, for electrons -. a p+ is a proton with 1 less charge, a p- is a proton with one addicional charge. a n+ is a nuetron with 1 less charge, a n- is a neutron with one addicional charge. a p- ≠ n(0) and a n+ ≠ p(0) due to differences in KE and PE (spin, size, mass, etc). a proton is 3 electron masses less heavy than an nuetron (used to be different). 3 electron masses in energy Ee = KE(spin, orbit) + PE(gravitic well). The base to which the measurement of charges are made is the nuetron, no charge. If the base for charging was made from the perspecitve of the proton, then the neutron would be 1 electron charge above the proton and the electron, 2 electron charges above the proton. Obviously, neither the neutron nor the proton are the true zero line for charge measurement. If you use the base energy of an electron Ebe= KEe + PEe within a gravitic well (a standard 0 level mass that exerts gravitic energy on all three), then the electron would have a base of 1, the proton at 1836 and the nuetron at 1839 on a mass energy basis due to the influence of gravity. fractalhints: ( 190.38.108.98 ( talk) 22:44, 29 December 2009 (UTC))
PS: Sorry about the formatting, shows up correctly in edit. —Preceding unsigned comment added by 190.38.108.98 ( talk) 22:48, 29 December 2009 (UTC)
- Taking your first remark:
Electron 1 e .00911 e-28 kg Protons 1836 e 16.72596 e-28 kg + ------------------------ 16.73507 e-28 kg Neutrons 1839 e 16.75329 e-28 kg - ------------------------- -.01822 e-28 kg
- Is your assertion that 1 neutron = 1 electron + 1 proton with no net energy change?— RJH ( talk) 22:17, 30 December 2009 (UTC)
The question is also as to how many times can this charge change take place in a single nucleon. The quark theory only allows two states of charge, + or -, (I think?) And for no reason. So a beta- change happens to neutrons and a beta+ happens to protons. And an ec doesn't involve a change in mass?. And the rest of the mass change is due to a mysterious change in the electrostatic/kinetic energy content. So it's kind of hard to explain it when you don't know what it is. WFPM ( talk) 15:28, 28 April 2010 (UTC)
Ångström
Do we have to use this dated non-SI unit? Can we not use picometres? JIMp talk· cont 21:27, 21 January 2010 (UTC)
- The convention in the WP:MoS is: "In scientific articles, use the units employed in the current scientific literature on that topic". If Angstroms are no longer being used in the scientific literature (physics? chemistry?), then a revision would make sense.— RJH ( talk) 15:40, 22 January 2010 (UTC)
The Anbstrom happens to be 10e-8cm which is about the size of the organized area around an atom. So you get a convenient reference of other sizes related to the atomic function size. WFPM ( talk) 15:37, 28 April 2010 (UTC)
Automate archiving?
Does anyone object to me setting up automatic archiving for this page using MiszaBot? Unless otherwise agreed, I would set it to archive threads that have been inactive for 30 days and keep ten threads.-- Oneiros ( talk) 14:08, 24 January 2010 (UTC)
 Done--
Oneiros (
talk) 19:25, 6 February 2010 (UTC)
Done--
Oneiros (
talk) 19:25, 6 February 2010 (UTC)
- Thirty days seems a little short.—22:52, 6 February 2010 (UTC)
Deleted parts of history
Around 1985, Steven Chu and co-workers at Bell Labs developed a technique for lowering the temperatures of atoms using lasers. In the same year, a team led by William D. Phillips managed to contain atoms of sodium in a magnetic trap. The combination of these two techniques and a method based on the Doppler effect, developed by Claude Cohen-Tannoudji and his group, allows small numbers of atoms to be cooled to several microkelvin. This allows the atoms to be studied with great precision, and later led to the Nobel prize-winning discovery of Bose-Einstein condensation.
Yes, but what great insight did they achieve? This paragraph is inconclusive.
Historically, single atoms have been prohibitively small for scientific applications. Recently, devices have been constructed that use a single metal atom connected through organic ligands to construct a single electron transistor.[44] Experiments have been carried out by trapping and slowing single atoms using laser cooling in a cavity to gain a better physical understanding of matter.
Another inconclusive paragraph. So they made a super-small transistor - what relevance is this to the article? Kurzon ( talk) 17:52, 24 April 2010 (UTC)
- The article on atoms suffers terribly from trying to define what we should put in "practical applications." Yike, this includes all of chemistry and engineering, if you look at it one way. Everything that uses normal matter materials. The "fix" is to include only applications that use "SINGLE" atoms, and that cuts it down to size. S B H arris 00:31, 26 April 2010 (UTC)
Boy! you guys certainly have diverging opinions! One only about atoms and subcontents, and the other about any type of use of the atom. But we certainly need to keep our eye on the ball and concentrate somewhere on the properties of the atom. And as Dr. Pauling said, if you want good ideas you first get a lot of ideas and then cull them out. WFPM ( talk) 15:47, 28 April 2010 (UTC) Would also like to see this supercooling tried on atoms of OO9F18 to see if atom is still unstable at that low a temperature.
- Please allow me to mention that the template at the top of this page says, "This is not a forum for general discussion of the article's subject."— RJH ( talk) 22:35, 18 August 2010 (UTC)
Electrons and rings
Someone wrote that Thomson thought the electrons rotated in rings within the atom. I've read elsewhere that Thomson believed electrons were vortex rings in the aether - they didn't move in rings, they were rings. Perhaps there was a confusion here (this happens easily with obsolete scientific theories). I welcome any clarifications. Kurzon ( talk) 18:15, 24 April 2010 (UTC)
- No, the vortex/nebular atom was Thomson's idea in the 1880's before he found the electron (what he called the "negative corpuscle," literally "negative body") experimentally. So this was a particle in his mind. His 1904 plum pudding model included his negative bodies, although the positive part of his atom is still very hazy and well... nebulous. "Cloud model with little bodies" is a better name than plum pudding model, which name of course Thomson never used. S B H arris 19:35, 24 April 2010 (UTC)
The explanation for the Bohr orbit model's being shot down was that it had to emit energy to stay in orbit. But in a conical orbit no energy is lost, and all conical orbits with the same major diameter have the same period of time and constant amount of lost free energy. Plus they all have a varying amount of angular momentum, except the circle which has a constant maximum amount for the concerned period. So the elimination of the Bohr orbit concept and the adoption of the Non angular momentum but energy containing "orbital" concept must have been for spectroscopic data or other purposes. WFPM ( talk) 22:31, 25 April 2010 (UTC) I don't like this much because I think that one of the functions of the electron is to allow the atom to get rid of excess angular momentum. WFPM ( talk) 22:43, 25 April 2010 (UTC) Like when a pitcher throws a fastball he may be emitting a particle with kinetic energy, but he's also gotten rid of a considerable amount of angular momentum. WFPM ( talk) 23:03, 25 April 2010 (UTC) This, of course, involves the "whip" principle which might indicate the necessity for some kind of structure of the emitting system. WFPM ( talk) 23:08, 25 April 2010 (UTC)
- Conical orbits? You mean conic section as in ellipses? The ellipse and circle are the only closed orbits. They both radiate energy classically, as the electron accelerates all the time in both. If you work it out the hydrogen electron should totally collapse and fall onto the proton (at which time they'd just be sitting there in contact) in about 1/100th of second, no matter what orbit it started in. But of course, quantum mechanically that can't happen, since it localizes the electron too well. The electron in ground-state hydrogen HAS no angular momentum. It's like a planet in an elliptical orbit with infinite thinness or eccentricity. As thought it only goes back and forth. S B H arris 00:28, 26 April 2010 (UTC)
Ah yes! But I'm noting that the conical orbit concept doesn't involve a loss in orbital energy until you get involved in the concept of electromagnetic activity, and that presumably conical physical orbits don't result in loss in energy. Now in the concept of electromagnetic energy, a charged particle is in a state of electrostatic potential in relation to it's location within an electrostatic field. And the question becomes as to whether a motion within that field results in a change in the electrostatic energy level. And I can't see how the circular orbit change of location of a particle around a centrally located electrostatic charge field would do it. And in an elliptical orbit the motion might involve an oscillatory change of position in the field, which might involve energy emissions, but that's not what the explanation says or explains. But maybe I'm nit picking. WFPM ( talk) 02:03, 26 April 2010 (UTC) And if you're willing to allow an attracted particle to move around without energy loss in an iso potential energy level in both the gravitational and electrmagnetic fields, why wouldn't you be willing to do so in an iso electrostatic energy level field. WFPM ( talk) 04:34, 26 April 2010 (UTC) How about a "reeled in" tethered electron in hydrogen? WFPM ( talk) 12:54, 27 April 2010 (UTC)
- You're not nitpicking, you're just wrong. Charged masses radiate EM waves when they are accelerated, and that includes in circular orbits like a cyclotron (see Larmor formula). Even uncharged gravitational objects radiate gravity waves (see Gravitational wave in circular orbits, although for solar system-scale masses, the energy loss is so small (about a 1000 watts for Jupiter orbiting the Sun) that it makes no difference and is not measurable. It has been measured in binary systems of neutron stars, however, and was Nobel Prize winnng work (see Hulse-Taylor binary). S B H arris 16:33, 30 April 2010 (UTC)
I see that in the Larmor formula that particles in a circular constant velocity orbit theoretically aren't supposed to radiate. WFPM ( talk) 00:06, 4 May 2010 (UTC) And in the Gravitational wave I read with interest the controversy about the effect of wave properties of a nonexistent medium upon real matter and hopeful detection methods.
- WFPM: If you are looking for a place to discuss your ideas, I recommend http://www.physicsforums.com/ — RJH ( talk) 15:35, 28 April 2010 (UTC)
So you think that the statement that a circularly moving electron in an electrostatic field where it doesn't lose energy of either position or motion has to be radiating energy due to a "classical" concept is not challengable, but must be accepted as part of any discussion. I bet you don't do that about the stability of gravitational orbits. WFPM ( talk) 16:06, 28 April 2010 (UTC)
Well I'm in favor of maximum understanding. So when I'm in your area please keep me in rein. WFPM ( talk) 20:47, 28 April 2010 (UTC)
Unsourced entries
The following content has been tagged as unsourced for several months. In order to remain an FA quality page, FA criteria 1c needs to be satisfied so that the "Claims are verifiable against high-quality reliable sources and are supported by inline citations". I'm moving the entries here until suitable citations can be found.
- In 1918, Rutherford discovered that the positive charge within every atom was always equal to an integer multiple of hydrogen nuclei, and deduced that all nuclei contained positively-charged particles called protons. The mass of the nucleus often exceeded this multiple, however, and speculated that the excess mass to be composed of neutrally-charged particles ( neutrons). citation needed
- The structure of the cloud varies with the number of electrons present in the cloud. There exist a number of different methods of electron counting, such as the octet rule and eighteen electron rule. These tend to be rules of thumb and are not valid across all atoms. Beginning chemistry students are often told the shell structure is simply 2, 8, 8, 8, 8, 8, 8, [...] to make the teaching process easier. The actual numbers of electrons per shell in the larger atoms can be considerably different, such as 2, 8, 18, 32, 50, 72, but this complexity is reserved for the more advanced student. citation needed
- Other nuclides have atomic masses and molar masses very close to whole numbers in their usual units, such as hydrogen-1. However, except for carbon-12, they cannot be exactly integer numbers, because the masses of different nuclides are not exact integer ratios of each other, although they do not differ from whole number ratios by more than 1%, and often much less. citation needed
If this is objectionable, please clarify. Thanks.— RJH ( talk) 17:26, 14 May 2010 (UTC)
Components
"Though the word atom originally denoted a particle that cannot be cut into smaller particles, in modern scientific usage the atom is composed of various subatomic particles." However, earlier in the article it said part of the definition was that it couldn't be divided by chemical means. So if it is divided by something other than chemical means, an atom is technically true to its definition, yes? 96.25.31.89 ( talk) 17:50, 24 September 2011 (UTC)
- I don't believe the article says anywhere that an atom is defined to be indivisible (with or without a restriction on what means are allowed for dividing the atom); it does say that Antoine Lavoisier defined it to be "a basic substance that could not be further broken down by the methods of chemistry". I don't think it's necessarily fruitful to argue whether the modern use of the word "atom" is "technically" true to this definition or another. I could argue, for example, that electrons are in fact atoms since they too cannot be "broken down by the methods of chemistry". What we mean when we say "atom" is, of course, "a basic unit of matter that consists of a dense central nucleus surrounded by a cloud of negatively charged electrons". Leonxlin ( talk) 04:49, 13 October 2011 (UTC)
Fields of power in the atom. Can someone answer this.
As far as I know, when we are speaking about the electromagnetic forces inside the atom of any element, is at rest. In the nucleus, the protons and neutrons are held together by the strong interaction, which as far as I know is only attractive. That is, the nucleotides are attracting each other by the strong nuclear force, even though the electromagnetic force is trying to hold the protons apart . This is because the protons have a positive sign and positive signs repel each other. Nevertheless, at very short distances, the nuclear force is much stronger than the electromagnetic force and keeps the protons in the compound that identifies the nucleus. The cloud of electrons outside the nucleus which by the way are not influenced by the nuclear force, have a negative sign and they repel each other. The relative distances between the particles are as far as I know in this context not very important, nevertheless it will be worth to mention that the nucleons are much more massive than the electrons and the radius of the nucleus is relatively much smaller than the apparent diameter that make the outskirts of the swarm of electrons. Can someone explain to me: Why are the electrons not attracted to the protons? They have opposite signs and they should be attracted by the electromagnetic force. And if they are attracted, why don't they collide with the protons? What is holding the equilibrium? I will be grateful if someone can explain this. —Preceding unsigned comment added by Enciclopedikt ( talk • contribs) 12:53, 19 June 2010 (UTC)
- The best place to ask this type of question is on the
Wikipedia:Reference desk/Science. Per the message box at the top, this page isn't really a discussion forum on the topic.—
RJH (
talk) 14:37, 19 June 2010 (UTC)
- However, the short answer is that the electrons ARE attracted to the protons and positive nucleus. And they get as close as they can to it (sometimes going right through it), but they can't stop, because stopping would localized them, and the uncertainty principle (that is, their wave-nature) prevents them from going into a smaller volume to be nearer the protons. The smaller the volume an electron is confined in, the more kinetic energy it has, and so the larger its orbital must be. This causes a balance in size. This minimum volume for confining electrons is what determines the size of the electron cloud of an atom, and thus the size of atoms themselves. S B H arris 18:46, 19 June 2010 (UTC)
- Thank you for your swift answer. I'll check the Reference desk/Science for more innput. —Preceding unsigned comment added by Enciclopedikt ( talk • contribs) 21:38, 19 June 2010 (UTC)
Catoms
Suggest a link be added to see also section for catoms, an attempt to create "synthethic" atoms using claytronics. 70.247.168.63 ( talk) 03:15, 19 November 2010 (UTC)
- I wonder if perhaps instead we should have a dedicated "Atom" navbox? We can then include links such as this.— RJH ( talk) 16:06, 17 December 2010 (UTC)
Atoms being photographed
A hydrogen atom has now been successfully photographed, and the same technique can apparently be applied to all other atoms (since hydrogen atoms are the smallest). I think this is worthy of mentioning in the article, but do others agree? 203.45.33.202 ( talk) 07:11, 28 November 2010 (UTC)
- Perhaps "atom photography" (I used the term loosely) in general deserves a mention, but this is hardly the first picture of single atoms. The atom is not the limit of photography, Canadian researchers like
Paul Corkum even took movies of electronic clouds moving around.
Headbomb {
talk /
contribs /
physics /
books} 07:16, 28 November 2010 (UTC)
- It is a good news for electron microscopy (though I don't think it is really new), but not here. In any case, proper reference is needed, as there are dozens of possible artifacts. To clarify - observing single atoms is very, very old result, but the lightest observable atom was lithium, AFAIK, but I think some groups were close to hydrogen. Materialscientist ( talk) 07:26, 28 November 2010 (UTC)
Indian nationalist POV
The same sentence with superlative “nationalist” adjectives exists in other articles of wikipedia. This sentence is partially indian nationalist POV.
“....The earliest references to the concept of atoms date back to ancient India in the 6th century BCE,[8] appearing first in Jainism.[9] The Nyaya and Vaisheshika schools developed elaborate theories of how atoms combined into more complex objects.[10] In the West, the references to atoms emerged a century later from Leucippus,....”
The Nyaya school is relatively recent. See Nyaya school and Nyaya sutras.. The existence of Kanada founder of Vaisheshika is very controversial, sources ranging from the sixth century BCE to second century CE. The fact that he might have been born and lived in western part of India probably during Mauryan empire or during an Indo-greek kingdom could suggest a hellenistic influence. If he is lived in VI century BCE the question is different but at moment unfortunately we don’t know exactly when he was alived. I am agree that a theistic atomism might be born in India indipendently from Mediterranean world but there are not evidences to affirm “earliest” and “before”. If the atom tradition is present in Jainism before Kanada. I would like to know the name of composition explaining atoms before Kanada. I think that grammatical and verbal word compositions in Vedas is an another thing ( Wikipedia:No original research ) but no atomism as in Democritus book fragments or in Kanada sutras (that are written in advanced CE era).
And about the book of Gangopadhyaya, Mrinalkanti. Indian Atomism: history and sources. Atlantic Highlands, New Jersey: Humanities Press, 1981. ISBN 0-391-02177-X. If this book has been subjected to peer review, and if this book follows the scientific method. I do not think that it uses these superlative and comparative adjectives.
I think is necessary to remove the adjectives "earliest" and "first" and the “comparative sentence” with western atomism because there are no evidences. My proposed amendment is as follows (obviously the english can be better)
“References to the concept of atoms date back to ancient India probably in the 6th century BCE,[8] appearing in Jainism.[9]. The Nyaya and Vaisheshika schools developed elaborate theories of how atoms combined into more complex objects.[10] In the West, the references to atoms emerged with Leucippus,....”
Andriolo -- 84.222.74.121 ( talk) 10:19, 12 January 2011 (UTC)
Other sentence:
"Although the Indian and Greek concepts of the atom were based purely on philosophy, modern science has retained the name coined by Democritus.[7]"
The Scientific Method that is born in Western Europe and is based on classic culture (Greek and Latin) could use an another name ?
Andriolo -- 84.222.74.121 ( talk) 11:09, 12 January 2011 (UTC)
- In the wording "The earliest references..", the word "known" is implicit. I.e. "The earliest known reference..." implies a documented source. This doesn't require knowing when and where the concept first appeared; only where it was first mentioned. If you have a reference to an earlier such work from a different culture, I would appreciate it if you could provide such. Supposition isn't appropriate per
WP:RELIABLE. Thank you.—
RJH (
talk) 17:53, 12 January 2011 (UTC)
- RJH, I agree with you.-- Indian Chronicles ( talk) 03:45, 13 January 2011 (UTC)
I don’t want cancel the sentence in question but only the adjectives
The Indian Culture during VI century BCE was oral. The range of datation of oral sources varies from VIII century BCE until II century and the oral sources have written during a period corrispondent advanced European Middle Age with interpolations. The same question is in Europe with the Norse Culture or about the datation of the Bible texts.
To much to affirm "earliest", “first” and “before” ecc, and to sentence: "modern science has retained the name coined by Democritus" . The science, can it make a choice ? Why, at this point, can not the Science use the sanskrit or maghadi name in the place of greek name ? This is the intentional nationalist message.
Other thing I would have the book in question to see if the adjectives “first” and “before” are used in the same way or if they are put, (out of originary context) in the articles of atom and atomism of eng. wikipedia with political intent. I would be grateful to Indian Chronicle if he could give me an extract of the text in question on my talk page Gangopadhyaya, Mrinalkanti. Indian Atomism: history and sources. Atlantic Highlands, New Jersey: Humanities Press, 1981. ISBN 0-391-02177-X. Only the pages where Gangopadhyaya use these adjectives, with the the references.
Is it possible to have an judgement of a specialist committee on this question ?
-- Andriolo ( talk) 09:15, 13 January 2011 (UTC)
- Andriolo my friend, you may have a point. But try to understand, no is claimimg that Greek atomism is influenced by Indian atomism or that Greeks copied from Indians. Both developed the ideas of atomism seperately. Only thing is that Indian atomism is slightly older as far as documented history is concerned [This is what the scholars and historians say]. Unfortunately, what you are doing is original research, which is against WP policies.-- Indian Chronicles ( talk) 04:15, 14 January 2011 (UTC)
Western Nationalist (Greek?) POV
Androlio you are indulging in Original Research. Just becasue some things do not match your expectation that does not mean that it is wrong or POV. So you believe that everything good in this civilization is from Greeks? Maybe but you will have to reference it. You have tried this trick many times on the page of Atomism. Just take a break my friend.-- Indian Chronicles ( talk) 17:19, 12 January 2011 (UTC)
- Are there better sources available that could resolve this dispute?— RJH ( talk) 17:47, 12 January 2011 (UTC)
Dear lovely friend (Indian Cronicles)
You use mirror style debate but your affirmation are without scientific evidences.
I have no expectations, and I am sceptic and relativist and as any good westerner I can conciliate these things with my faith living in the contrast with the doubt.
While you, you have the absolute truth in your mind.
Andriolo -- 84.222.74.121 ( talk) 18:57, 12 January 2011 (UTC)
- Please see the template message at the top of the page. This is a place for a discussion of how to improve the article, rather than exchanging personal observations about other editors. Thank you.— RJH ( talk) 19:11, 12 January 2011 (UTC)
- RJH you are right. Although I can reply to Andriolo on his personal remarks, I will desist from it. Let us talk to improving this excellent article. Nationalism should have no say in this scientific article.-- Indian Chronicles ( talk) 03:44, 13 January 2011 (UTC)
The Vedas are sacred texts and in them, as in the Bible, Qur'an or others sacred texts you can find everything and its opposite.
-- Andriolo ( talk) 09:33, 13 January 2011 (UTC)
Page 317 and thereafter of The shape of ancient thought: comparative studies in Greek and Indian philosophies has an interesting discussion on the topic. Thoughts?— RJH ( talk) 16:27, 13 January 2011 (UTC)
---
In this book at pages 315-321 there is exactly the point in question. I have tried to use this book to explain to Indian Cronicles the question, in vain. (see Atomism talk)
In pag 317: " The canons of three [Indian] schools however, were systematized and written down centuries after the period in question, a situation which does not make easy. "
In pag 319 about Gangopadaya and Jain atomism the author says “...The Indian tradition places Jain atomism at least as early as the sixth century b.c.........Though there is no way at present to check the absolute dates of such texts, there is still reason to believe that Jain atomism is very early.....”
Infact to affirm " The earliest references to the concept of atoms date back to ancient India in the 6th century BCE,[8] appearing first in Jainism.[9]" is to much categorical, is better to write this sentence without the adjectives. For example: Other references to the concept of atoms date back to ancient India probably (or second the tradition) in the 6th century BCE [8] appearing in Jainism.[9] and in other indian schools as Ajivika and Charvaka etc etc
If Indian Cronicles don’t want to change the sentence, I think is better to put near this affirmation to balance: "Though there is no way at present to check the absolute dates of [oral] composition of [Indian] texts. "
The Indian schools could be more ancient, contemporary, or recent than greek or hellenistic schools but the latter are identified from written sources nearly contemporaneous. Aristotle is practically contemporary to Democritus (50 years). We know that Democritus wrote numerous books (see Thrasyllus of Mendes). Unfortunately the books are destroyed, from religious fanaticism during the fall of Roman Empire (as the bigger part of classic culture see Libraries of Alexandria and Pergamon and not only). But lot of fragments survives as citation in other books of tens of hellenistic and roman authors that fortunately are arrived to us.
While in the Indian schools the difference between the traditional date of oral composition and written sources is enormous: from 900 to 1500 years after. And without interpolations ? Could be. But we don’t know and the answers are only speculations
So the question is complex and unresolved.... but i think that we must write that is unresolved.
-- 84.223.59.79 ( talk) 13:52, 14 January 2011 (UTC) Sorry I haven't remember to firm with my wiki name. -- Andriolo ( talk) 14:04, 14 January 2011 (UTC)
- Although the author admits to difficulty in dating text, he is quite categorical in saying that there is reason to believe that Jain atomism is very early and shows archiac forms of thought and more primitive than Ajivika. Note use of superlative in very early, archiac and primotive instead of old or ancient. Hence the current way the edit stands seems quite reasonable. And by the way, dating of a text depends on when it was composed and not when it was written.-- Indian Chronicles ( talk) 14:21, 14 January 2011 (UTC)
And this sentence hasn't sense: "Although the Indian and Greek concepts of the atom were based purely on philosophy, modern science has retained the name coined by Democritus.[7]" infact we could use the Nahuatl word if the Scientific Revolution was born en Aztec Empire.
This atom article is good, but it is very important, there are lot of little students that see it so we must do it better. -- Andriolo ( talk) 14:15, 14 January 2011 (UTC)
- The name coined for atom has nothing to do with its origin. Jungle is a word of Indian origin. That does not mean Jungles exist only in India! -- Indian Chronicles ( talk) 14:23, 14 January 2011 (UTC)
Infact in the Europe we have the boreal Forest, perhaps in future with global warming.....
Howewer could I modify the text in atom and atomism articles in the follow manner ?
..The earliest references to the concept of atoms date back to ancient India in the 6th century BCE,[8] appearing first in Jainism.[9] The more recent Nyaya and Vaisheshika schools developed elaborate theories of how atoms combined into more complex objects.[10] Though there is no way at present to check the absolute dates of oral composition of texts indeed the canons of Indian schools were systematized and written down centuries after the period in question. (x) In the West, the references to atoms emerged in 5th century BCE with Leucippus .....
With the references of the book in question
(x) Thomas McEvilley, The Shape of Ancient Thought: Comparative Studies in Greek and Indian Philosophies ISBM ecc ecc, pp. 317-321.
But this is confirmed but not only Thomas McEvilley book but also from other important Indian scholars. As in
S. Radhakrishnan (I have an italian translation), La filosofia indiana, vol 1, Dai Veda al Buddismo, pag 283. Le testimonianze più antiche a favore del Jainismo, di cui non si può contestare la veridicità, non risalgono oltre il V d.c. ;”
ISBN
88-85405-29-0
Substantially
S. Radhakrishnan claim that Ardha Magadhi probably is more recent than Pali. He gives other opinions as Colbrooke, Jacobi etc, but the author dubt. So the VI century BCE is not an absolute dating. It is only a plausible dating.
Have you (plural) an opinion ? Do you propose an adjustment ?
RJH, Can I post it ?
-- Andriolo ( talk) 22:39, 15 January 2011 (UTC)
- Well, nobody owns the page. It's more a matter of gaining consensus really.
- No offense intended, but the meaning of the third sentence is not quite clear. Might I suggest a slight re-write?
- At present, there is no means to check the absolute dates of oral composition of the ancient Indian texts. Indeed, the canons of Indian schools were systematized and written down centuries after the period in question.
- — RJH ( talk) 17:19, 16 January 2011 (UTC)
- Andriolo, Please remember this article is about Atom as understood by scientist and not about historical discussion as to when the texts were written down. You are blatantly indulging in Original Research and totally digressing from the main issue just to feed your nationalistic sentiments. This is against the policies of WP. You are basically trying to question the already referenced sentence. If you can find a well referenced reliable source that says otherwise then please go ahead. Else you are blatantly violating the WP policies.-- Indian Chronicles ( talk) 04:34, 17 January 2011 (UTC)
For RJH. I am not a english speaker, so I wanted a confirm about good english and the “politically correct” form. Your rewrite proposal is better. I will try to put this sentence in the atom and atomism articles, but I'm sure it will be deleted. -- Andriolo ( talk) 08:13, 17 January 2011 (UTC)
- This edit
[4] is a textbook case of nationalistic sentiment spoiling a featured article dedicated to one of the most important topic of science. I wil not call it vandalism but it stands out like a sore thumb. But I will not touch it. Maybe some sane editor will salvage the article and delete this edit.--
Indian Chronicles (
talk) 16:47, 17 January 2011 (UTC)
- I don't really want to get into what constitute a nationalistic sentiment here, but the statement as it stands doesn't appear especially biased. It's just a simple statement of fact. However, it is slightly off topic for the subject, so perhaps it would work better as a footnote. Would that be acceptable for you?—
RJH (
talk) 17:06, 17 January 2011 (UTC)
- Yes, its totally off-topic. The finer nuances of systematisation of Indian texts do not belong here. Maybe it is better off as a footnote. That should also pander Androlio's nationalistic sentiment without spoiling the 'scientificness' article.--
Indian Chronicles (
talk) 17:38, 17 January 2011 (UTC)
- Thank you. Regarding your last sentence, please take a look at WP:Civility at your earliest convenience.— RJH ( talk) 17:44, 17 January 2011 (UTC)
- Yes, its totally off-topic. The finer nuances of systematisation of Indian texts do not belong here. Maybe it is better off as a footnote. That should also pander Androlio's nationalistic sentiment without spoiling the 'scientificness' article.--
Indian Chronicles (
talk) 17:38, 17 January 2011 (UTC)
- I don't really want to get into what constitute a nationalistic sentiment here, but the statement as it stands doesn't appear especially biased. It's just a simple statement of fact. However, it is slightly off topic for the subject, so perhaps it would work better as a footnote. Would that be acceptable for you?—
RJH (
talk) 17:06, 17 January 2011 (UTC)
- This edit
[4] is a textbook case of nationalistic sentiment spoiling a featured article dedicated to one of the most important topic of science. I wil not call it vandalism but it stands out like a sore thumb. But I will not touch it. Maybe some sane editor will salvage the article and delete this edit.--
Indian Chronicles (
talk) 16:47, 17 January 2011 (UTC)
- Thanks. I took a look as you suggested and did not find anything offensive that I may have said considering the snide comments and holier than thou attitude that I have faced from Andriolo.-- Indian Chronicles ( talk) 08:11, 18 January 2011 (UTC)
But I just wanted to remove some adjectives..... only this. I agree the “balance sentence” should go to the footnotes. But first, to respect the Scientific Method and the Research, it would be necessary to modify the sentence of Indian Cronicles in probabilistic terms leaving the question open, as it is. My opinion is that the article must give evident informations of discernment especially for non specialists and children (little students for school research). They often take the article concepts as absolute.
I'm tired, I'm just an occasional and new editor of Wikipedia, I have not time but I have understand that this question is of “dogmatic importance” for them probably “for proselytism” and surely some sane editor will salvage the article and delete my edit soon.
Is it possible eventually to have a final judgement of a specialist committee on this question ?
-- Andriolo ( talk) 23:32, 17 January 2011 (UTC)
- There is no such specialist committee that resolves such matters; only us generic editors. Most of your suggested wording has been implemented by an anonymous editor. You agree with the idea of moving the "balance statement" to the notes. Are you now unhappy with the result? I am unclear, and it is starting to seem less than likely that we can make the both of you feel satisfied with the result.— RJH ( talk) 23:49, 17 January 2011 (UTC)
- RJH, I am okay with this compromise in footnote even though I feel that this does not belong here.-- Indian Chronicles ( talk) 08:11, 18 January 2011 (UTC)
I am the anonymous editor, I didn’t remember to make the login. I try to firm in the second time with minor modification.
If we put the "balance statement" at the footnote. The principal and evident sentence in the text remain unbalanced with its “faith message” and the dogma “we are more ancient than others”. Statement that does not pass the critical analysis.
If Indian Cronicle will modify his sentence in probabilistic terms, the “balance statement” must go to footnote. Otherwise if the sentence of Indian Cronicle remain categorical and determinative, it is better that it remains in the text, indeed this "balance statement" give us a discerning information.
In this talk, I don’t want dispute the oral composition dating propose from Indian Cronicle, indeed is a Pandora's box that I don’t want open. Indian Cronicles oral composition dating is not shared from all scholars, there are lot of different suppositions (with a range of 1300 years. From VIII BCE (black centuries of India) and V century CE without to consider the question of written sources ranging from 50 BCE(?) to 1500 CE. The datations of oral compositions are based on analysis of the
Prakrit languages used in the compositions or generally only on the tradition
How we can resolve the question at moment it is in a dead point ?
-- 84.222.75.240 ( talk) 10:06, 18 January 2011 (UTC)-- Andriolo ( talk) 10:10, 18 January 2011 (UTC)
- This tinkering with edits that have no relevence is spoiling this featured article. A lot of people have taken a lot of efforts to make it and maintain it as a featured article. Discussion of composition of oral texts does not fall here. Andriolo, You claim many scholars have disputed but you are not able to come up with one reference to the contrary. Yet, I give up for the sake of this article. You can have your way as far as this article is concerned. We can go with your version: References to the concept of atoms date back to ancient India probably in the 6th century BCE,[8] appearing in Jainism.[9]. The Nyaya and Vaisheshika schools developed elaborate theories of how atoms combined into more complex objects.[10] We can simply delete the misfit: At present, there is no means to check the absolute dates of oral composition of the ancient Indian texts. Indeed, the canons of Indian schools were systematized and written down centuries after the period in question. [11]. I think that should resolve this discussion. [PS: This does not mean that I agree with the compromise. Its simply my wish for consensus and peace and hopeful that this article does not lose its featured status because of some silly content dispute.] -- Indian Chronicles ( talk) 11:15, 18 January 2011 (UTC)
How much vanity....... OK, I agree:
References to the concept of atoms date back to ancient India probably in the 6th century BCE,[8] appearing in Jainism.[9]. The Nyaya and Vaisheshika schools developed elaborate theories of how atoms combined into more complex objects.[10] The “balance sentence” is no more necessary but if you haven’t fear of it, and if you love the history, you leave it in the footnotes. Will you change the same sentence in atomism article ?
Finally I have a citation for you: "... you want to remove the immovable foundations of our faith in the gods, when you ask for each of them a rational demonstration. The ancestral faith of our fathers is based on itself.... we cannot to find clearer evidence of this..... if only a point is called in the analysis his solidity and the general belief are shaken.....” (Amatorius, 13, 756 B) Plutarch
Now I can go back into my greek well.
-- Andriolo ( talk) 13:25, 18 January 2011 (UTC)
- Thanks Andriolo for the quotation. I have a better creed to live by and that is anekantavada. Do go through it whenever you have time. Thanks RJH for moderating the discussion.-- Indian Chronicles ( talk) 04:19, 19 January 2011 (UTC)
I do it, and I have finished. Thank you RJH, and Thank you Indian Cronicles I have studied a bit Indian philosophy for me is a new world. I think that contacts with roman-hellenistic world was absolute. Existed
Suez Canal and the cosmopolitan
Alexandria. I live in a city that has founded its richness on the pepper trade during the Middle Ages, without roman paved roads and the persian-hellenistic-roman Suez Canal. The marchants of my city knew goods, mathematics, mesures. You know Pegolotti "
pratica della mercatura". There are hundreds in the archives. Why in the the ancient world the commerce should be different ? The greek were merchants and mercenaires as in my medieval city they absorbed everything. For greek philosophy the concepts are no important but the method and the proposition of the mind and that has created a black hole, every knowledges is not enough.
Remain opens others question on atomism article, perhaps in future I will write something directly with references. The phoenician Mochus of Sidon the reference you can find in the specific article. The chinese atomism: Mohism haunted from Quin emperor. The Hermeticism tradition (Egyptian-Ptolemaic) (alchemy) more important than ancient atomism for modern concept of atom see Boyle Sceptical Chymist. There are a links between Hermeticism-Freemasonry and Courpoularism. To define the concept of ancient atom (because it is material and indivisible) other atomisms are non absolutely indivisibles and have theistic part). I see that jain atomism discriminates from material and immaterial and it is relativistic but I see that in other indian school could have atom concept resambling arché concept as probably in chinese Mohism. There is also a grammarian atomism but is it atomism ?
Sorry RJH for this outline.
-- Andriolo ( talk) 02:14, 19 January 2011 (UTC)
It is not absolutely important for me, but Indian Cronicles have chosen to delete the footnotes in object... perhaps for some obscure reason he has fear of the phrase. It seems that democracy knowledge of wikipedia (often influenced from faith) perhaps could be in contrast with scientific knowledge. But I hope with the law of big numbers and aristotelian logic..... with time the science comes out.
(No offense intended)
« Veritas est adaequatio intellectus ad rem; adaequatio rei ad intellectum; adaequatio rei et intellectus. » Thomas Aquinas
In this dispute I see the inability to abstract the subject (not only the object). Or using the more simple metaphor of the elephant present in the Anekantavada that Indian Cronicles can to understand. ...He is constantly touching the elephant's nose....
Howewer the hard relationship between faith and reason divides my country from centuries and maybe I give too much weight on this thing. But often the “message” is not into the concepts but into the semantics. And often the citations are interpreted. -- Andriolo ( talk) 15:08, 19 January 2011 (UTC)
- I don't really have any skin in this issue, and it seems to be somewhat minor. To me, whether the addition is included or not isn't going to significantly impact the quality of the article.
- The difference between the two versions appears to be the footnote statement:
- At present, there is no means to check the absolute dates of oral composition of the ancient Indian texts. Indeed, the canons of Indian schools were systematized and written down centuries after the period in question.
- Do you agree? This appears to boils down the statement that we don't have an exact date, along with an explanation of why we don't know the date. However, the first sentence states, "probably in the 6th century". This would seem to imply the same thing. Am I mistaken?— RJH ( talk) 18:51, 19 January 2011 (UTC)
No, for my friend was a great effort to accept the probabilistic sentence. I left him the freedom to choose about the notes. --
Andriolo (
talk) 20:19, 19 January 2011 (UTC)
- This seems to be a priority dispute between Greece and India. Judging by the McEvilley book (
ISBN
1581152035), the Indian dating is uncertain and it's not known who first developed the atomist philosophy. So, I think the first two sentences should be rewritten to clarify this, for example, to:
References to the concept of atoms date back to ancient Greece and India. In India, the Ajivika, Jain, and Carvaka schools of atomism may date back to the 6th century BCE. (McEvilley, p. 317) The Nyaya and Vaisheshika schools later developed theories on how atoms combined into more complex objects. (King, pp. 105-107)
- The claim in the second sentence is the same as in McEvilley. Also, it's better not to use Teresi as a source as he's a popularizer. The King reference is to Indian philosophy: an introduction to Hindu and Buddhist thought, Richard King, Edinburgh University Press, 1999, ISBN 0748609547. Spacepotato ( talk) 04:29, 20 January 2011 (UTC)
- This seems to be a priority dispute between Greece and India. Judging by the McEvilley book (
ISBN
1581152035), the Indian dating is uncertain and it's not known who first developed the atomist philosophy. So, I think the first two sentences should be rewritten to clarify this, for example, to:
- If it is a priority dispute between India and Greece and if we are to go by McEvily, then McEvily thinks that Indian atomism is earlier than Greek and probably influenced the Greeks. Let me quote McEvily:
- Ajivika, Jain, and Carvaka brands of atomism, on the other hand may habve been in existence by 6th Century BC and are candidates to have influenced thoughts of Leucippus and Democritus.p317
- Indian Tradition places Jain atomism at least as early as sixth century BC—that is atleast a century before the earliest extant traces of Greek atomism. None of the authors deny this, only qualify the dating part due to oral tradition and not the priority part. And as far as difficulty in dating is concerned, they are not sure about the exact year or decade, but confidently quote century. Hence, I think we should maintain the status quo on the article as of now. Maybe the debate of priority and different viewpoints may be discussed in
atomism article--
Indian Chronicles (
talk) 05:29, 20 January 2011 (UTC)
- He says that they "may have" been in existence by the 6th century B.C., but that "The canons of all three schools...were systematized and written down centuries after the period in question". On p. 321, he says that "Atomism...may have been among the varied philosophical merchandise that contact by way of the Persian Empire carried from East to West or from West to East". Also: (p. 520) "In fact, there is no certain proof of the existence of atomism in any Indian philosophy until after 326 B.C.", which is followed by speculation on transmission from the Greco-Roman world to India. From these quotes, it seems that he does not believe the priority question, or the question of influence and transmission, have been resolved one way or the other. Spacepotato ( talk) 05:53, 20 January 2011 (UTC)
- Spacepotato, what you are saying is right. Prima facie what the author is saying in pg 510 is in contradiction with what he said earlier in p.317. I don't know how he has come to that conclusion on pg 510 and what is the significance of 326 BCE. Since google books is the only source, other pages are blocked hence we cannot come to conclusion in what is in authors mind. On pg. 510 additionally he discusses speculation by other authors on transmission from the Greco-Roman world to India without himself giving an opinion. On the other hand on pg quoted by me above, he himself speculated on Indian influence on Greeks. What ever the case maybe, we cannot come to a conclusion as we are like archeologists digging up data that can be found and arriving at conclusions. In the end we can go with what you say if RJH also agrees. However too much discussion on this should not be in this article but in atomism.-- Indian Chronicles ( talk) 08:53, 20 January 2011 (UTC)
- Yes, the first sentence proposed by Spacepotato does establish a solid baseline level of neutrality. The fact that the Indian heritage is presented next can be ascribed to the chronological order of the dates presented; in contrast to the otherwise logical alphabetical order. Overall, the wording favors neither party, so this proposal works for me. Thank you.— RJH ( talk) 16:04, 21 January 2011 (UTC)
The sentence proposed by Spacepotato is better. It gives more information (about other indian schools) and it is probabilistic in the form. -- Andriolo ( talk) 10:58, 20 January 2011 (UTC)
- As there was agreement on the proposal I made the change. I believe the significance of 326 BC is that it's the date of Alexander's invasion of India. Spacepotato ( talk) 02:47, 22 January 2011 (UTC)
The 326 is intended as the date when India (the Ganges valley) becomes the object of study for the western style
historiography This historiography can not to be used before this date.
It is necessary to use others methods but these do not give us the same precision:
The India of
Mahajanapadas didn’t had complex painting, masonry or stone building, cities, sculpture, writing system (see
Brahmi),
coins. All sources considered from western historiography. Before, Indians surely used wood for temples and houses, palaces, but unfortunately the monsoon climate is destructive for everything that is organic.
From
Indica and other
classic authors and oral Indian tradition we know about the existence of a rich, powerful, divided and segmented civilisation.
The only sources (before 326) are:
1) the wonderful and well preserved
oral tradition (but indian historiography used it in a bit different manner than western historiography)(
Phylology-Anthropology)
2) villages’s pottery and manufacts. (
Archeology-
Anthropology)
3) linguistic analisys.
In Europe the same questions we have for the
Norse culture.
I don't know if in wikipedia there is a talk about this interesting argument.
Correct me if I am wrong. but that explain the 326
-- 84.222.73.144 ( talk) 21:35, 22 January 2011 (UTC)-- Andriolo ( talk) 21:38, 22 January 2011 (UTC)
Indian Cronicles don’t want to change the usual sentence that in this moment exist in atomism article 1/20/2011:
The earliest references to the concept of atoms date back to ancient India in the 6th century BC,[2] appearing first in Jainism.[3] The Nyaya and Vaisheshika schools developed elaborate theories of how atoms combined into more complex objects.[4]
Can you help me Spacepotato ? I am tired to debate, he don't want to understand.
-- Andriolo ( talk) 11:24, 20 January 2011 (UTC)
WP:LDR
I've been implementing WP:LDR on a number of new and existing articles because I find that it makes editing and maintenance of an article easier. With LDR, the inline citation templates are all moved to the end of the article, thereby leaving mostly the article text in the body. (See the source for the Earth article, as an example.) Does anybody have an objection to implementing WP:LDR on this article? Thanks.— RJH ( talk) 20:47, 15 March 2011 (UTC)
- Actually this is a good idea. I dont think anyone would have any objection to it.-- Indian Chronicles ( talk) 04:26, 16 March 2011 (UTC)
Most non-bot edits are made to the text, rather than to the citations. With a massive clutter of inline citations, editing the text is made much more difficult. Thus, the optimum circumstances for most editors is having the references out of the way. I might also add that having the references together actually makes reading the history page easier and simplifies the task of performing mass updates to the citations. The benefits of WP:LDR outway the drawbacks, I believe, and so in most cases it makes sense to implement; especially for well-developed and stable articles such as FA'd topics.— RJH ( talk) 14:21, 18 March 2011 (UTC)
- I agree with you. Even I am unable to understand Headbombs objection.-- Indian Chronicles ( talk) 10:41, 19 March 2011 (UTC)
To be an atom, or not to be an atom.....
This article says, "However, the hydrogen-1 atom has no neutrons and a positive hydrogen ion has no electrons." Isn't the electron necessary to call it an atom?
siNkarma86—Expert Sectioneer of Wikipedia
86 = 19+9+14 + karma = 19+9+14 +
talk 20:17, 13 July 2011 (UTC)
- Yes, although technically we're calling it a hydrogen 'ion' rather than a hydrogen 'atom'. I have seen instances of a "Lithium atom that is fully ionized", for example, so usage varies. Regards, RJH ( talk) 22:32, 13 July 2011 (UTC)
Duplicate reference
There are two different references under one name ref name=schroeder, one for L.Meitner, the other for the magnetic properties.-- 77.85.4.229 ( talk) 11:05, 29 July 2011 (UTC)
- Fixed, thanks. Materialscientist ( talk) 11:51, 29 July 2011 (UTC)
English
In the opening paragraph the writing uses "less" when talking about things that can counted ("less electrons"). I suggest the corrected phrase should be "fewer electrons". Use "less" when talking about things that cannot be counted e.g. "less sugar means fewer sugar crystals". — Preceding unsigned comment added by 174.34.217.49 ( talk) 14:20, 28 October 2011 (UTC)
- Changed, thanks. Materialscientist ( talk) 14:23, 28 October 2011 (UTC)
Book reference
Is there room in the book reference section for Asimov's book "Atom, Journey across the subatomic cosmos, Penguin Books 1992. WFPM ( talk) 22:20, 26 November 2011 (UTC) ISBN 0-452-26834-6
Well you know Asimov! He talks interestingly about everything. And he almost makes you think you should understand it! And remember that he died in April 1992 and the book was published in August 1992. WFPM ( talk) 10:16, 14 December 2011 (UTC)
- Yes I do. However, the Book References section is intended for citations of material in the article. If it can't be used for that purpose, then I think the book would perhaps be better placed under a Wikipedia:Further reading section heading. Thanks. Regards, RJH ( talk) 15:34, 14 December 2011 (UTC)
You might note that the book is not among those listed in his biography. But it's a good chronological discussion of science progress, and probably his most up to date writing. And he must have had a fantastic memory. WFPM ( talk) 23:53, 14 December 2011 (UTC)
OK! At your instigation, I reread it, and it's a tour de force and chronological history of the physical/chemical history and concepts of atomic physics as only Asimov could write it, (328 pages). But he still models nucleons with spheres, which should be banned, and I like magnetized cylinders better.Regards. WFPM ( talk) 01:02, 16 December 2011 (UTC)Truman Talley Books/Plume
MASS: An update to nuclear properties is needed
The large majority of an atom's mass comes from the protons and neutrons that make it up. The total number of these particles (called "nucleons") in a given atom is called the mass number. The mass number is a simple whole number, and has units of "nucleons." An example of use of a mass number is "carbon-12," which has 12 nucleons (six protons and six neutrons).
This statement should be changed. Mass Defect is an important concept of the mass of a nucleus and as such, should be included in this section. — Preceding unsigned comment added by 108.17.134.163 ( talk) 09:50, 14 April 2012 (UTC)
- The mass number is defined to be the number of nucleons. Mass defects are irrelevant here. Headbomb { talk / contribs / physics / books} 13:58, 14 April 2012 (UTC)
Classification of quarks
In the article is the sentence:
- The quark belongs to the fermion group of particles, and is one of the two basic constituents of matter — the other being the lepton, of which the electron is an example.
I find this somewhat missleading: It sounds like there would be two types of particles: fermions and leptons. I would like to suggest to replace the sentence by something like:
- The quarks and the electron belong to the fermion group of particles.
It is maybe the best to leave the classification of fermions into leptons and quarks to the standart model page. That way the paragraph only contains the two distinct classes fermions and (gauge-) bosons.
-- Falktan ( talk) 15:06, 19 June 2012 (UTC)
- I think it could be re-written in a manner that retains the intended information. Perhaps:
- Quarks and electrons are the two basic constituents of matter, and together they form the fermion group of particles.
- Regards, RJH ( talk) 15:26, 19 June 2012 (UTC)
- I think we are getting closer :)
- I still would leave the class of leptons out here, since the electron is not only an example, but the only lepton that plays any role at all. If not filled with any further information the word lepton is meaningless here anyway.
- Regards, Falktan ( talk) 19:41, 19 June 2012 (UTC)
- In that case there's no need to mention electrons either because it's out of scope because of the exclusion of leptons from the sentence. I suggest changing it to:
- The quark belongs to the fermion group of particles, and is one of the basic constituents of matter.
- Regards, RJH ( talk)
- Agreed. We should throw out the electrons here. Actually the remaining sentence is not so meaningful either. I would even consider removing the hole sentence, or at least the part about "constituents of matter". This point is a bit tricky: Even though the quarks are the only particle in the nucleus having a rest mass, also the gluons play an important role for the overall mass of the nucleus, as can most clearly be seen from the mass defect. I would leave this hole point to the articles about protons and neutrons. So my suggestion would be
- In the Standard Model of physics, both protons and neutrons are composed of elementary particles called quarks. There are six types of quarks, each having a fractional electric charge of either +2⁄3 or −1⁄3. Protons are composed of two up quarks and one down quark, while a neutron consists of one up quark and two down quarks. This distinction accounts for the difference in mass and charge between the two particles. The quarks are held together by the strong nuclear force, which is mediated by gluons. While the quarks are fermions, the gluon is a member of the family of gauge bosons, which are elementary particles that mediate physical forces. [4] [5]
- For comparision the original:
- In the Standard Model of physics, both protons and neutrons are composed of elementary particles called quarks. The quark belongs to the fermion group of particles, and is one of the two basic constituents of matter—the other being the lepton, of which the electron is an example. There are six types of quarks, each having a fractional electric charge of either +2⁄3 or −1⁄3. Protons are composed of two up quarks and one down quark, while a neutron consists of one up quark and two down quarks. This distinction accounts for the difference in mass and charge between the two particles. The quarks are held together by the strong nuclear force, which is mediated by gluons. The gluon is a member of the family of gauge bosons, which are elementary particles that mediate physical forces. [4] [5]
- I moved the part about fermions to the last sentence. I would also consider throwing out or rephrasing the sentence
- This distinction accounts for the difference in mass and charge between the two particles.
- Regards, Falktan ( talk) 12:51, 20 June 2012 (UTC)
- Agreed. We should throw out the electrons here. Actually the remaining sentence is not so meaningful either. I would even consider removing the hole sentence, or at least the part about "constituents of matter". This point is a bit tricky: Even though the quarks are the only particle in the nucleus having a rest mass, also the gluons play an important role for the overall mass of the nucleus, as can most clearly be seen from the mass defect. I would leave this hole point to the articles about protons and neutrons. So my suggestion would be
For the same reason, we really don't want to discuss the origin of the atom's mass here, since that leads into the origin of baryon mass, which is a complicated and ill-understood subject. Save that for the baryon article, or the articles on proton and neutron, where it is needed NOW and doesn't exist . Only 1% (or so) of baryon mass is due to rest mass of quarks, but there exist also sea quarks (virtual quarks), the mass of the quark kinetic energy, gluons with their energy (and therefore invariant mass) and other fields (static electric fields have mass, even though made of virtual photons, not real ones).
Somwhat likewise, the "mass defect" of nucleon binding is also not worth looking at mechistically. It's not just "quarks." Just say that it never exceeds 1% of nuclear mass (as compared with the free baryon masses in toto-- that 1% figure occuring again here, is coincidental, and of course can be smaller). This missing mass would actually be twice as large as it is, if not for the mass of the static electric potential energy *added* (not subtracted) when the positive charges were squashed together to make nuclei. This electric added-mass is always more than made up for, by missing mass from nuclear force field destroyed, but this nuclear force field is made of virtual mesons (pions and some rho mesons) and we don't want to go THERE, either! No. We can (maybe) just note that nuclear force is more energetic (in terms of potential) than the electric field potential, and that's why nuclei stick together.
But fission destroys electric potential at the expense of creating nuclear potential, and fusion does the opposite (destroys nuclear potential and creates electrical potential, as charges are pushed together) and that is all a matter of discussion in THOSE articles ( nuclear fission and nuclear fusion, with perhaps some more in binding energy and atomic nucleus). But atom is not the place for it, as we haven't room. S B H arris 18:04, 20 June 2012 (UTC)
- I totally agree. I like the changes in the article. Thanks, Falktan ( talk) 11:53, 21 June 2012 (UTC)
The big bang, hydrogen not being mentioned.
I remember watching the talk show something from nothing with evolutionary biologist Richard Dawkins and Lawrence Krauss, Lawrence said that the first element created after the big bang were hydrogen helium, and a bit of lithium. Why did you not put hydrogen there, And protons and electrons were created 3 seconds after the big bang. — Preceding unsigned comment added by Anastronomer ( talk • contribs) 09:58, 20 October 2012 (UTC)
- Please sign your talk page messages with four tildes (~~~~). Thanks.
- As you can see in the opening sentence of Big Bang nucleosynthesis, this nucleosynthesis does not produce hydrogen. It produces hydrogen isotopes and other nuclei from hydrogen. Cheers - DVdm ( talk) 12:07, 20 October 2012 (UTC)
Allotropes
In the section on 'States', a correction needs made. The article is locked, so I can't. The article currently reads:
"Within a state, a material can also exist in different phases. An example of this is solid carbon, which can exist as graphite or diamond."
This is not a phase. It is an allotrope. See the Wikipedia article on Allotropy. The statement should instead be worded:
"Within a state, an element can also exist as different allotropes. An example of this is solid carbon, which can exist as graphite or diamond."
Perhaps even linking 'allotropes' to the Wikipedia Allotropy article.
Thanks!
66.62.194.228 ( talk) 00:37, 17 November 2012 (UTC)
- Fixed. Get a username , make 10 good edits, wait 4 days, and you too can edit these " locked" articles , which are only protected against IP editing. S B H arris 09:38, 18 November 2012 (UTC)
Please add 'positronium' as another example of exotic matter
Positronium. I'd do it myself, but as you know the article is in lockdown. Thanks. 24.79.75.240 ( talk) 07:36, 5 August 2012 (UTC)
 Done by adding the category:exotic atoms to the
this section, which provides a list including Positronium. Also added the p. exotic matter with the same idea. (You could also get an account), Cheers, -- chat
O'Brien 08:41, 3 December 2012 (UTC)
Done by adding the category:exotic atoms to the
this section, which provides a list including Positronium. Also added the p. exotic matter with the same idea. (You could also get an account), Cheers, -- chat
O'Brien 08:41, 3 December 2012 (UTC)
individual
the english word that matches the greek word is individual. we dont say indivisible (αδιαίρετο) or uncuttable (άτμητο) in greek. and i think it is important cause the meaning changed with chemistry. i mean until the microscope was invented or Hook's Micrographia, people didnt talk about microorganisms Wassermagier ( talk) 17:14, 7 March 2013 (UTC)
Anaxagoras
What about the Ionian School and Anaxagoras?
Why is the history section bungled?
More elaboration is needed about Ionian Greek Atomic Theory (philosophy). This article has it wrong, or very incomplete.
The error was obvious and the article does not give an old enough date for the ancient Greek atomic theory. Anaxagoras was born 510 BC and as many know the Ionians are the creators of the atomic model and cosmic mind. Democritus was from the Traki city state in northern Greece and lived slightly later (at least 50 years).
The Ionian Atomic Theory is famous to anyone that reads philosophy; Wikipedia editors have just botched it. — Preceding unsigned comment added by 64.252.6.62 ( talk) 05:21, 24 March 2013 (UTC)
- Many philosophy readers have just botched to find the atomic theory article. Reading clever books does not improve their cognitive abilities enough. Incnis Mrsi ( talk) 14:39, 24 March 2013 (UTC)
grading this article
Grading “The Atom”
the first sentence in the article is inaccurate. There is no such thing as 'clouds of negatively charged electrons' the second sentence is confusing – it introduces new materials without a contextual background – we are introduced to 'the stability of nuclides', and forced to differentiate between protons and nuetrons without any anecodotal or empirical evidence which may allow us to do so effectively. The third sentence of the introduction is incomplete – the 'electromagnetic force' is only one of the nuclear forces – the article makes no mention to any other nuclear force. The fourt sentence – is inaccurate, it suggests that 'chemical bonds' are electromagnetic in nature – suggesting that molecular stability (the stability of mult-chemical compounds) is electro-magnetic-- for instance, that the electro-magnetic bond is most essential to molecular AIR. The following sentence is false – furthermore, the usage of the term “proton” “neutron” and “electron” which referred to different kinds of sub-atomic particles, is not pedagogically useful, without corrborating information which may serve to DEFINE these particles in function and effect (the term ION is useless, and it is only confusing and does not help to clarify anything in this context). The following sentence is a mis-statement: atoms are classified on the basis primarily of WEIGHT – atomic weight which corresponds to atomic number – less dense atoms (such as hydrogen or oxygen) versus those which are HEAVIER (salts, iron) – as aforementioned, the terms PROTON and NEUTRON are not useful in this context.
- Chemical bonds ARE electromagnetic in nature! "Molecular air" is a mixture not a compound-- the only chemical bonds in "molecular air" are between atoms in air molecules (at least those air molecules that contain more than one atom; this isn't true in argon). Atomic weight does NOT correspond to atomic number. If you order atoms by atomic weight, you will run up against next door pairs that are the same or even reversed in weight (potassium/argon, cobalt/nickel, tellurium/iodine) but of course different in number (place on the periodic table). This was a big problem in the days before people knew what atomic number was besides place on the periodic table. It was not understood until 1914. S B H arris 02:02, 5 June 2013 (UTC)
The Next Paragraph!
The opening sentence posits the existence of “objects” with the diameter of a few nanometers – suggesting that “atoms” may also correspond to such a description, without any supporting evidence or providing information regarding the manner in which measurements may be achieved (though it does suggest the existence of a tool referred to as a “scanning tunneling microscope” – a thing which may or may not exist – this raises an important issue, but fails to resolve it or explore it at sufficient depth – the integrity of “atomic theory” depended on the ability to provide empirical evidence which confirmed the atom's existence.
- The scanning tunneling microscope may or may not exist?? Maybe you should read the article? S B H arris 02:02, 5 June 2013 (UTC)
It may be true that an atom's mass is concentrated in the nucleus – but again, the usage of protons and neutrons and their possession of 'equal mass' is not relevant if we have ignored the issue of 'ATOMIC STABILITY', which proves to be more important – a discussion of nuclear forces may be more appropriate in this section. Modern “atomic theory” – (and it may be useful to reference the idea, that for MOST PEOPLE, atoms remain theoretical, as these were for early Greco-Roman scientists, becauase WE as amateur scientists and theoreticians, LACK the ability to indisputably prove their existence) – includes the idea of the weak and strong nuclear forces – the forces relevant to the stability of the atom include the electro-magnetic force, the gravitational force, as well as the 'physical force' (as in physical bonding)
- "Physical force"?? "Physical bonding"? What? Are you making up your own terms? Gravitational force has nothing to do wtih the stability of atoms. S B H arris 02:02, 5 June 2013 (UTC)
a understanding of the creation of photons/electrons at the sub-atomic level presupposes a understanding of nuclear forces – it is not practical to introduce the idea of orbitals, at this stage of argumentation – furthermore, the inclusion of this idea suggests a non-NPOV argumentation – these development borrows rigorously from established APE curriculum (and includes the fallacious reasoning, sometimes used for 'pedagogical' purposes).
Given the problems in the introduction of the material – I do not believe that this article at present may pass a fair review. Furthermore, the introduction should include some of the historical background of atomic theory – or provide a link to “atomic theory” – as for example, by such a device, : “the 'atom' is the smallest unit of stable matter of any chemical element supposed to exist in the universe, as described in 'atomic theory'” (and here the link) --- it may be useful to include this in the introduction, as this is where that sort of material belongs, rather than, in one of the body paragraphs.
I would also like to know why this article has been "locked" to prevent editing -- as it is in such a obvious state of disrepair. Is there an "administrator" who is responsible for this? Please step forward and respond to these queries-- I would appreciate this. -Anaceus. — Preceding unsigned comment added by 68.51.85.203 ( talk) 16:38, 3 June 2013 (UTC)
- You misunderstand what WP:Verifiability means. It is not about “evidences which indisputably prove” something. It is about reliable sources. Can you, as an amateur geographer, prove indisputably that Pacific Ocean is larger than all other planet’s bodies of water combined? Or, as an amateur historian, prove the existence of Thirteen Colonies? I am sure than the answer to both questions is “no”. Then, why are you so preoccupied with atom while numerous things exist beyond your personal experience? Incnis Mrsi ( talk) 17:39, 3 June 2013 (UTC)
certainly-- i have no problem with what you are suggesting. however-- to re-write an article on the atom, consistent with the wikipedian philosophy of expressing NPOV -- a neutral perspective -- would require for us to work with the essential facts as they are reported to us -- for instance, to accept that there are, lets say, 3 sub-atomic particles which are essential to an undertanding of the atom. we can accept this. to suggest, that these particles in their function, collectively, allow for 'atomic stability' -- furthermore, to include well-documented and important information -- such as the existence of 'nuclear forces'-- which is to say, of forces essential to nuclear stability (the stability of an atomic nucleus)-- in all cases, in the physical sciences, it is not merely a question of sources -- but we are charged with the responsibility of interpration -- in order to re-write a clear and accurate article, it is necesary for us to accept as credible some of the basic constituent parts, as related to us in the atomic theory of some conventional sources, while of necessity arranging them in a manner that is most consistent with PHYSICAL REALITY, such as we understand it.
likewise, it is necessary for us to reject information that is incomplete, misleading, causes confusion, makes a shallow usage of the "nomenclature" of the physical sciences, and leads to a superficial comprehension of the material.
--- as for the pacific ocean-- yes, we have merely to consult available information, such as referring to any of the available satellites which provide geographical information on the internet-- supposing we accept the formal definition of "pacific ocean", we then have merely to find an acceptable means of measurement, and draw our conclusions. as for the 13 colonies-- we may find sufficient anecedotal evidence to support the existence of the 13 colonies in published literature to accept the usage of the term-- and if we are interested as historians to uncover further corrborating evidence, we may look for the historical artifacts of that period-- we start with some hypotheses, that there were people, perhaps english-speaking people, who lived in a place that we may loosely define as the east coast of america, during a specific period of time, and that these people were the predecessors of the contemporary nation-state that is the USA, and with this as our point of departure attempt to find supporting evidence in order to confirm our hypotheses (or, perhaps, to our disappointment, find that there is no evidence to support such an idea)-- what if we, for instance, stared with the idea that vessels had departed the great oceans of barsoom during the 75th century by the ancient (*$*$*$*) calendar and that THESE settlers were the founders of the nation which currently occupies the territories we consider as specific to the United States of America (as between a certain latitude and longitude, ... )-- in order to prove this hypothesis, we would need to uncover evidence and prove its authenticity.
this is besides the point. for our purposes, we have the evidence of our senses-- in physical reality, all matter maintains its stability irrespective of the rotation of the earth, and our orbit, and the phases of the moon, and all the rest of that-- we suppose that matter must be composed of something, we attempt an analysis-- we accept the theses of "atomic theory" rather than accepting the idea that "all rocks are made of grains", "all socks made of strands" and so forth-- the atom is the proposed answer to this question-- what is the atom? the atom is the proposed, hypothetical "smallest possible unit of a chemical element that maintains its stability" -- this hypothesis includes our understanding of the existence of chemical elements-- we suppose that an atom of hydrogen may differ from an atom of carbon, or an atom of iron -- or we may reject this theory entirely, and create something which is more satisfying to our reasoning and intellect. this is our choice.
Throughout history there have been questions as to the “verfiability” of the existence of the atom – this question is as essential to the study of atomic theory as the electrons and photons which are speculated to be emitted from an atomic nucleus. This article does not concern itself with any of the advanced research and science which relates to particle physics – we can, however, use the starting point which is our definition of the atom – rather than our definition of the “material grain” or “cotton strand” – to draw useful conclusions regarding atomic structure and particle physics – for instance, that if the atom is the “smallest possible unit of any chemical element to function as stable matter”, that sub-atomic particles such as neutrons, protons, and electrons do not function as “stable matter”, for instance.
- It's hard to say what "stable matter" means, but it usually it is a term is taken to be the "stuff" of which everyday objects are made. Neurons, protons, and electrons alone will not do to make such stuff. The charge on protons and electrons, if they were collected together withough equal amounts of the other, would blow even small collections of them. And (except in incredible mass) neutrons are radioactive, when alone. S B H arris 02:02, 5 June 2013 (UTC)
My criticism is of the incompleteness, vagueness, poor use of terminology, inaccuracy, and structural weakness of this article. Your criticism appears to be of my interest in this article.
It is reasonable to take any text which concerns itself with the physical sciences as a possible source – all of them will make reference to positive and negative electrical charge – to alpha and beta decay – to gamma radiation – to positively changed particles, to negatively charged particles, to particles of a neutral charge – to nuclear forces.
To reference these terms is insufficient: it not acceptable to say, “ions are the byproduct of alpha decay” without placing such an idea in a context – it is even less useful to say “The electrons determine the chemical properties of an element, and strongly influence an atom's magnetic properties” (paragraph 2, sentence 7). --anaceus. — Preceding unsigned comment added by 68.51.85.203 ( talk) 15:02, 4 June 2013 (UTC)
- This encyclopedia works with hyperlinks, which place terms immediately in context. If you don't know what alpha decay is, you can simply click on the term and read the article, then come back. So it's not like the texts you're used to. S B H arris 23:16, 4 June 2013 (UTC)
yes-- I understand that is true-- however 'alpha decay' is a term that is relative-- in physics, such terms may be defined for different purposes-- they are interchangeable-- we designate something as 'alpha', something as 'beta', thus may refer to an alpha and beta decay-- there is, in organic and inorganic chemistry-- organic decay which is a molecular decay which occurs in such things as fruit and vegetation, as well as non-organic decay, such as the minor decrease in atomic weight which is the result of atomic radioactivity.
yes. i understand! wikipedia is rooted in html, of course-- it is the world's first encylopedia written entirely with the help of html, which allows for hyperlinks, well 'why didn't i think of that', haha?
it is not so hard to say what "stable matter" is-- our daily life provides us with direct experience of a great variety of stable matter.
my point is that this article is very conventional-- and furthermore, not very useful, from the purpose of improving a reader's understanding of nuclear forces, of atomic theory, of the structure/role of the atom. it does not challenge the reader to re-examine any of their beliefs regarding the fundaments of matter, and further makes statements that are inaccurate and wrong-- it is an article that is in need of serious editing. it was my recommendation that a project overview-- a kind of, well-mannered backing away from the accepted terminology of science, in order to allow for a more objective perspective, may be useful-- if we accept, for instance, the idea that 'neutrality' must be guide, and that furthemore that science requires of us that we only take as factual things which we are capable of empirically verifying, then, for many of us-- the atom is consigned to the realm of theory-- from this standpoint, what is the usefulness of thing of the equally theoretical sub-atomic particles by specific names-- we are better off, if we wish to improve our understanding, by using more general terms-- and by developing definitions which serve to refine our comprehension--- and yes, gravitational forces are quite important to atomic stability, SbHarris.
Is there a neutral arbitrater or some procedure to which we may appeal in order to resolve this issue? This article is essential, it is poorly developed, and I see no reason to waste a great deal of time bickering if it is with people who have no interest in working to improve this particular article-- or have no interest in the subject. --anaceus. — Preceding unsigned comment added by 68.51.85.203 ( talk) 07:49, 5 June 2013 (UTC)
"Chemical bonds ARE electromagnetic in nature! "Molecular air" is a mixture not a compound" -- I suppose we should break this into two parts, I will respond in reverse order:
1. "molecular air" from this standpoint, would be both a mixture and a compound-- if we include in the phenomena of air, both clouds, which frequently include molecular compounds of hydrogen and oxygen, and 'air' which is the un-bonded mixture of hydrogen and oxygen 2. "chemical bonds ARE electromagnetic in nature!" -- well, if in this statement you mean to suggest that the *bonds* at the atomic level, between sub-atomic particles are electro-magnetic in nature, then yes, you are in part correct, because electr-magnetism is another primary nuclear force-- however, if you are suggesting, as in the previous example, that the bonds between for instance, molecular compounds of hydrogen and oxygen are "ARE electromagnetic", then no-- those are bonds that exist as the result of changes in atmosphere pressure, and physical processes such as preciptation, and are most certainly not electromagnetic. To rephrase this, some of the bonds which characterize the fundamental differences between chemical elements are electromagnetic, however, the bonds between molecular compounds, such as the bonds between hydrogen and carbon in hydrocarbons, or the bonds between oxygen and hydrogen in hydroxides are in no ways electromagnetic "in nature!" --anaceus. — Preceding unsigned comment added by 68.51.85.203 ( talk) 08:24, 5 June 2013 (UTC)
- It's sad that you don't even know that air is oxygen and nitrogen (not hydrogen). Or that the hydrogen bonds between water molecules are electrmagnetic as are all significant interactions between molecules. Read chemical bond and learn. Hydrocarbon and carbohydrate are well-defined very different chemical terms. Read and learn. Their atoms are held together no differently from H and O in water molecules (or hydroxides for that matter). The fundamentals of chemistry elude you and you want to edit the article on atoms. Your ideas on gravitational stability of atoms are fringe. You try to find textbook references and you will see how fringe. Wikipedia's job is to summarize the well-accepted. All these links are available, such as van der Waals interaction. Educate yourself. S B H arris 19:34, 5 June 2013 (UTC)
- Grinchnote - see User talk:68.51.85.203. - DVdm ( talk) 20:39, 5 June 2013 (UTC)
- ^ Protons and neutrons are over 1800 times more massive than electrons
-
^ Staff (
August 1
2007).
"Radioactive Decays". Stanford Linear Accelerator Center, Stanford University. Retrieved 2007-01-02.
{{ cite web}}: Check date values in:|date=( help) - ^ {{cite news | author=Staff | date=July 1, 2008 | title=Physicists Create Millimeter-sized 'Bohr Atom' | publisher= Science Daily | url= http://www.sciencedaily.com/releases/2008/06/080630173921.htm
- ^
a
b Cite error: The named reference
pdg2002was invoked but never defined (see the help page). - ^
a
b Cite error: The named reference
schombert2006was invoked but never defined (see the help page).