
A speleothem ( /ˈspiːliəθɛm/; from Ancient Greek σπήλαιον (spḗlaion) 'cave', and θέμα (théma) 'deposit') is a geological formation by mineral deposits that accumulate over time in natural caves. [1] Speleothems most commonly form in calcareous caves due to carbonate dissolution reactions. They can take a variety of forms, depending on their depositional history and environment. Their chemical composition, gradual growth, and preservation in caves make them useful paleoclimatic proxies.
Chemical and physical characteristics
More than 300 variations of cave mineral deposits have been identified. [2] The vast majority of speleothems are calcareous, composed of calcium carbonate (CaCO3) minerals ( calcite or aragonite). Less commonly, speleothems are made of calcium sulfate ( gypsum or mirabilite) or opal. [2] Speleothems of pure calcium carbonate or calcium sulfate are translucent and colorless. The presence of iron oxide or copper provides a reddish brown color. The presence of manganese oxide can create darker colors such as black or dark brown. Speleothems can also be brown due to the presence of mud and silt. [2]
Many factors impact the shape and color of speleothems, including the chemical composition of the rock and water, water seepage rate, water flow direction, cave temperature, cave humidity, air currents, aboveground climate, and aboveground plant cover. Weaker flows and short travel distances form narrower stalagmites, while heavier flow and a greater fall distance tend to form broader ones.
Formation processes
Most cave chemistry involves calcium carbonate (CaCO3) containing rocks such as limestone or dolomite, composed of calcite or aragonite minerals. Carbonate minerals are more soluble in the presence of higher carbon dioxide (CO2) and lower temperatures. Calcareous speleothems form via carbonate dissolution reactions whereby rainwater reacts with soil CO2 to create weakly acidic water via the reaction: [3]
- H2O + CO2 → H2CO3
As the acidic water travels through the calcium carbonate bedrock from the surface to the cave ceiling, it dissolves the bedrock via the reaction:
- CaCO3 + H2CO3 → Ca2+ + 2 HCO3−
When the solution reaches a cave, the lower pCO2 in the cave drives the precipitation of CaCO3 via the reaction:
- Ca2+ + 2 HCO3− → CaCO3 + H2O + CO2
Over time, the accumulation of these precipitates form dripstones ( stalagmites, stalactites), and flowstones, two of the major types of speleothems.
Climate proxies
Speleothem transects can provide paleoclimate records similar to those from ice cores or tree rings. [4] Slow geometrical growth and incorporation of radioactive elements enables speleothems to be accurately and precisely dated over much of the late Quaternary by radiocarbon dating and uranium-thorium dating, as long as the cave is a closed system and the speleothem has not undergone recrystallization. [5] Oxygen ( δ18O) and carbon ( δ13C) stable isotopes are used to track variation in rainfall temperature, precipitation, and vegetation changes over the past ~500,000 years. [6] [7] The Mg/Ca proxy has likewise been used as a moisture indicator, although its reliability as a palaeohygrometer can be affected by cave ventilation during dry seasons. [8] Variations in precipitation alter the width of speleothem rings: closed rings indicates little rainfall, wider spacing indicates heavier rainfall, and denser rings indicate higher moisture. Drip rate counting and trace element analysis of the water drops record short-term climate variations, such as El Niño–Southern Oscillation (ENSO) climate events. [9] Exceptionally, climate proxy data from the early Permian period have been retrieved from speleothems dated to 289 million years ago sourced from infilled caves exposed by quarrying at the Richards Spur locality in Oklahoma. [10]
Types and categories
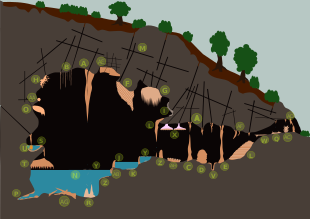
(A) Stalactite (B) Soda straws (C) Stalagmites (D) Coned stalagmite (E) Stalagnate or column (F) Drapery (G) Drapery (H) Helictites (I) Moonmilk (J) Sinter pool, rimstone (K) Calcite crystals (L) Sinter terrace (M) Karst (N) Body of water (O) Shield (P) Cave clouds (Q) Cave pearls (R) Tower cones (S) Shelfstones (T) Baldacchino canopy (U) Bottlebrush stalactite (V) Conulite (W) Flowstone (X) Trays (Y) Calcite rafts (Z) Cave popcorn or coralloids (AA) Frostworks (AB) Flowstone (AC) Splattermite (AD) Speleoseismites (AE) Boxworks (AF) Oriented stalactite (AG) collapsed rubble
Speleothems take various forms, depending on whether the water drips, seeps, condenses, flows, or ponds. Many speleothems are named for their resemblance to man-made or natural objects. Types of speleothems include: [11]
- Dripstone is calcium carbonate in the form of stalactites or stalagmites
-
Stalactites are pointed pendants hanging from the cave ceiling, from which they grow
- Soda straws are very thin but long stalactites with an elongated cylindrical shape rather than the usual more conical shape of stalactites
-
Helictites are stalactites that have a central canal with twig-like or spiral projections that appear to defy gravity
- Include forms known as ribbon helictites, saws, rods, butterflies, hands, curly-fries, and "clumps of worms"
- Chandeliers are complex clusters of ceiling decorations
- Ribbon stalactites, or simply "ribbons", are shaped accordingly
-
Stalagmites are the "ground-up" counterparts of stalactites, often blunt mounds
- Broomstick stalagmites are very tall and spindly
- Totem pole stalagmites are also tall and shaped like their namesakes
- Fried egg stalagmites are small, typically wider than they are tall
- Stalagnate results when stalactites and stalagmites meet or when stalactites reach the floor of the cave
-
Stalactites are pointed pendants hanging from the cave ceiling, from which they grow
-
Flowstone is sheet like and found on cave floors and walls
- Draperies or curtains are thin, wavy sheets of calcite hanging downward
- Bacon is a drapery with variously colored bands within the sheet
- Rimstone dams, or gours, occur at stream ripples and form barriers that may contain water
- Stone waterfall formations simulate frozen cascades
- Draperies or curtains are thin, wavy sheets of calcite hanging downward
- Cave crystals
- Dogtooth spar are large calcite crystals often found near seasonal pools
- Frostwork is needle-like growths of calcite or aragonite
- Moonmilk is white and cheese-like
- Anthodites are flower-like clusters of aragonite crystals
- Cryogenic calcite crystals are loose grains of calcite found on the floors of caves formed by segregation of solutes during freezing of water.
-
Speleogens (technically distinct from speleothems) are formations within caves that are created by the removal of
bedrock, rather than as secondary deposits. These include:
- Pillars
- Scallops
- Boneyard
- Boxwork
- Others
- Cave popcorn, also known as "coralloids" or "cave coral", are small, knobby clusters of calcite
- Cave pearls are the result of water dripping from high above, causing small "seed" crystals to turn over so often that they form into near-perfect spheres of calcium carbonate
- Snottites are colonies of predominantly sulfur oxidizing bacteria and have the consistency of "snot", or mucus
- Calcite rafts are thin accumulations of calcite that appear on the surface of cave pools
- Hells Bells, a particular speleothem found in the El Zapote cenote of Yucatan in the form of submerged, bell-like shapes
- Lava tubes contain speleothems composed of sulfates, mirabilite or opal. When the lava cools, precipitation occurs.
Calthemites
The usual definition of speleothem excludes secondary mineral deposits derived from concrete, lime, mortar, or other calcareous material (e.g. limestone and dolomite) outside the cave environment or in artificial caves (e.g. mines, tunnels), which can have similar shapes and forms as speleothems. Such secondary deposits in man-made structures are termed calthemites. Calthemites are often associated with concrete degradation, or due to leaching of lime, mortar, or other calcareous material.
Gallery
-
Various formations in the Hall of the Mountain Kings, Ogof Craig a Ffynnon, South Wales, Great Britain
-
Stalactites and columns in Natural Bridge Caverns, Texas
-
More formations in Natural Bridge Caverns, Texas
-
California Caverns, Calaveras County, California; one of many caverns located in the Sierra Foothills of California
-
Image of Cave Pearl formation
-
Image of flowstone in Mammoth Cave, KY
See also
References
- ^ White, W. B. (2019). "Speleothems". Encyclopedia of Caves: 1006–17. doi: 10.1016/B978-0-12-814124-3.00117-5. ISBN 9780128141243.
- ^ a b c White, William (2016). "Chemistry and karst". Acta Carsologica. 44 (3). doi: 10.3986/ac.v44i3.1896. ISSN 0583-6050.
-
^ J., Fairchild, Ian (2012).
Speleothem science: from process to past environments. Wiley-Blackwell.
ISBN
978-1-4051-9620-8.
OCLC
813621194.
{{ cite book}}: CS1 maint: multiple names: authors list ( link) - ^ Bradley, Raymond S. (2015). Paleoclimatology: Reconstructing Climates of the Quaternary. Academic Press. pp. 291–318. doi: 10.1016/b978-0-12-386913-5.00008-9. ISBN 978-0-12-386913-5.
- ^ Richards, David A.; Dorale, Jeffrey A. (2003). "Uranium-series Chronology and Environmental Applications of Speleothems". Reviews in Mineralogy and Geochemistry. 52 (1): 407–460. Bibcode: 2003RvMG...52..407R. doi: 10.2113/0520407. ISSN 1529-6466.
- ^ Fairchild, Ian J.; Smith, Claire L.; Baker, Andy; Fuller, Lisa; Spötl, Christoph; Mattey, Dave; McDermott, Frank; E.I.M.F. (2006). "Modification and preservation of environmental signals in speleothems" (PDF). Earth-Science Reviews. ISOtopes in PALaeoenvironmental reconstruction (ISOPAL). 75 (1–4): 105–153. Bibcode: 2006ESRv...75..105F. doi: 10.1016/j.earscirev.2005.08.003.
- ^ Hendy, C. H (1971). "The isotopic geochemistry of speleothems–I. The calculation of the effects of different modes of formation on the isotopic composition of speleothems and their applicability as palaeoclimatic indicators". Geochimica et Cosmochimica Acta. 35 (8): 801–824. Bibcode: 1971GeCoA..35..801H. doi: 10.1016/0016-7037(71)90127-X.
- ^ Ronay, Elli R.; Breitenbach, Sebastian F. M.; Oster, Jessica L. (25 March 2019). "Sensitivity of speleothem records in the Indian Summer Monsoon region to dry season infiltration". Scientific Reports. 9 (1): 5091. Bibcode: 2019NatSR...9.5091R. doi: 10.1038/s41598-019-41630-2. ISSN 2045-2322. PMC 6434041. PMID 30911101.
- ^ McDonald, Janece; Drysdale, Russell; Hill, David (2004). "The 2002–2003 El Niño recorded in Australian cave drip waters: Implications for reconstructing rainfall histories using stalagmites". Geophysical Research Letters. 31 (22): L22202. Bibcode: 2004GeoRL..3122202M. doi: 10.1029/2004gl020859. hdl: 1959.13/29201. ISSN 1944-8007.
- ^ Woodhead, Jon; Reisz, Robert; Fox, David; Drysdale, Russell; Hellstrom, John; Maas, Roland; Cheng, Hai; Edwards, R. Lawrence (2010-05-01). "Speleothem climate records from deep time? Exploring the potential with an example from the Permian". Geology. 38 (5): 455–458. Bibcode: 2010Geo....38..455W. doi: 10.1130/G30354.1. hdl: 1959.13/931960. ISSN 0091-7613.
- ^ Hill, C A, and Forti, P, (1997). Cave Minerals of the World, (2nd edition). [Huntsville, Alabama: National Speleological Society Inc.] pp. 217, 225
External links
- The Virtual Cave: an online guide to speleothems
- Mineral aggregates from carst caves, formed in capillary film solutions
- Gallery of speleothems from NPS Cave and Karst Program (archived on 23 January 2013)









