
The extended CDR3 loop is coloured orange.
A single-domain antibody (sdAb), also known as a Nanobody, is an antibody fragment consisting of a single monomeric variable antibody domain. Like a whole antibody, it is able to bind selectively to a specific antigen. With a molecular weight of only 12–15 kDa, single-domain antibodies are much smaller than common antibodies (150–160 kDa) which are composed of two heavy protein chains and two light chains, and even smaller than Fab fragments (~50 kDa, one light chain and half a heavy chain) and single-chain variable fragments (~25 kDa, two variable domains, one from a light and one from a heavy chain). [1]
The first single-domain antibodies were engineered from heavy-chain antibodies found in camelids; these are called VHH fragments. Cartilaginous fishes also have heavy-chain antibodies (IgNAR, 'immunoglobulin new antigen receptor'), from which single-domain antibodies called VNAR fragments can be obtained. [2] An alternative approach is to split the dimeric variable domains from common immunoglobulin G (IgG) from humans or mice into monomers. Although most research into single-domain antibodies is currently based on heavy chain variable domains, Nanobodies derived from light chains have also been shown to bind specifically to target epitopes. [3]
Camelid Nanobodies have been shown to be just as specific as antibodies, and in some cases they are more robust. They are easily isolated using the same phage panning procedure used for antibodies, allowing them to be cultured in vitro in large concentrations. The smaller size and single domain make these antibodies easier to transform into bacterial cells for bulk production, making them ideal for research purposes. [4]
Single-domain antibodies are being researched for multiple pharmaceutical applications, and have potential for use in the treatment of acute coronary syndrome, cancer, Alzheimer's disease, [5] [6] and Covid-19. [7] [8] [9]
Properties
A single-domain antibody is a peptide chain of about 110 amino acids long, comprising one variable domain (VH) of a heavy-chain antibody, or of a common IgG. These peptides have similar affinity to antigens as whole antibodies, but are more heat-resistant and stable towards detergents and high concentrations of urea. Those derived from camelid and fish antibodies are less lipophilic and more soluble in water, owing to their complementarity-determining region 3 (CDR3), which forms an extended loop (coloured orange in the ribbon diagram above) covering the lipophilic site that normally binds to a light chain. [10] [11] In contrast to common antibodies, two out of six single-domain antibodies survived a temperature of 90 °C (194 °F) without losing their ability to bind antigens in a 1999 study. [12] Stability towards gastric acid and proteases depends on the amino acid sequence. Some species have been shown to be active in the intestine after oral application, [13] [14] but their low absorption from the gut impedes the development of systemically active orally administered single-domain antibodies.

The comparatively low molecular mass leads to a better permeability in tissues, and to a short plasma half-life since they are eliminated renally. [1] Unlike whole antibodies, they do not show complement system triggered cytotoxicity because they lack an Fc region. Camelid and fish derived sdAbs are able to bind to hidden antigens that are not accessible to whole antibodies, for example to the active sites of enzymes. [15] This property has been shown to result from their extended CDR3 loop, which is able to penetrate such buried sites. [11] [16] [15]
Production
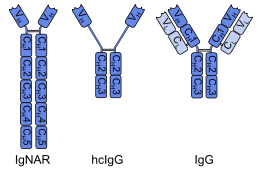
From heavy-chain antibodies
A single-domain antibody can be obtained by immunization of dromedaries, camels, llamas, alpacas or sharks with the desired antigen and subsequent isolation of the mRNA coding for the variable region (VNAR and VHH) of heavy-chain antibodies. Large phage displayed VNAR and VHH single domain libraries were established from nurse sharks [17] and dromedary camels. [18] [19] Screening techniques like phage display and ribosome display help to identify the clones binding the antigen. [20] [17] [21] [18] [22] [8] [19] The single domain antibodies including VNARs can be humanized for clinical applications. [23]
From conventional antibodies
Alternatively, single-domain antibodies can be made from common murine, [24] rabbit [25] or human IgG [26] with four chains. [27] The process is similar, comprising gene libraries from immunized or naïve donors and display techniques for identification of the most specific antigens. A problem with this approach is that the binding region of common IgG consists of two domains (VH and VL), which tend to dimerize or aggregate because of their lipophilicity. Monomerization is usually accomplished by replacing lipophilic by hydrophilic amino acids, but often results in a loss of affinity to the antigen. [28] If affinity can be retained, the single-domain antibodies can likewise be produced in E. coli, [25] [26] [29] S. cerevisiae or other organisms.
From human single-domain antibodies
Humans occasionally produce single domain antibodies by the random creation of a stop codon in the light chain. Human single-domain antibodies targeting various tumor antigens including mesothelin, [29] GPC2 [30] and GPC3 [26] [31] were isolated by phage display. The HN3 human single-domain antibodies have been used to create immunotoxins [31] [32] [33] and chimeric antigen receptor (CAR) T cells [34] for treating liver cancer. Blocking the Wnt binding domain of GPC3 by the HN3 human single-domain antibody inhibits Wnt activation in liver cancer cells. [35]
Potential applications
Single-domain antibodies allow a broad range of applications in biotechnical as well as therapeutic use due to their small size, simple production and high affinity. [36] [37] [15]
Biotechnological and diagnostic
The fusion of a fluorescent protein to a Nanobody generates a so-called chromobody. Chromobodies can be used to recognize and trace targets in different compartments of living cells. They can therefore increase the possibilities of live cell microscopy and will enable novel functional studies. [38] The coupling of an anti- GFP Nanobody to a monovalent matrix, called GFP-nanotrap, allows the isolation of GFP-fusion proteins and their interacting partners for further biochemical analyses. [39] Single molecule localization with super-resolution imaging techniques requires the specific delivery of fluorophores into close proximity with a target protein. Due to their large size the use of antibodies coupled to organic dyes can often lead to a misleading signal owing to the distance between the fluorophore and the target protein. The fusion of organic dyes to anti-GFP Nanobodies targeting GFP-tagged proteins allows nanometer spatial resolution and minimal linkage error because of the small size and high affinity. [40] The size dividend of Nanobodies also benefits the correlative light-electron microscopy study. Without any permeabilization agent, the cytoplasm of the chemically fixed cells are readily accessible to the fluorophore tagged Nanobodies. Their small size also allows them to penetrate deeper into volumetric samples than regular antibodies. High ultrastructural quality is preserved in the tissue that is imaged by fluorescence microscope and then electron microscope. This is especially useful for the neuroscience research that requires both molecular labeling and electron microscopic imaging. [41]
In diagnostic biosensor applications Nanobodies may be used prospectively as a tool. Due to their small size, they can be coupled more densely on biosensor surfaces. In addition to their advantage in targeting less accessible epitopes, their conformational stability also leads to higher resistance to surface regeneration conditions. After immobilizing single-domain antibodies on sensor surfaces sensing human prostate-specific antigen (hPSA) were tested. The Nanobodies outperformed the classical antibodies in detecting clinical significant concentrations of hPSA. [42]
To increase the crystallization probability of a target molecule, Nanobodies can be used as crystallization chaperones. As auxiliary proteins, they can reduce the conformational heterogeneity by binding and stabilizing just a subset of conformational states. They also can mask surfaces interfering with the crystallization while extending regions that form crystal contacts. [43] [37]
Therapeutic

Single-domain antibodies have been tested as a new therapeutic tool against multiple targets. In mice infected with influenza A virus subtype H5N1, Nanobodies directed against hemaglutinin suppressed replication of the H5N1 virus in vivo and reduced morbidity and mortality. [44] Nanobodies targeting the cell receptor binding domain of the virulence factors toxin A and toxin B of Clostridium difficile were shown to neutralize cytopathic effects in fibroblasts in vitro. [45] Nanobody conjugates recognizing antigen presenting cells have been successfully used for tumor detection [46] or targeted antigen delivery to generate strong immune response. [47]
Orally available single-domain antibodies against E. coli-induced diarrhoea in piglets have been developed and successfully tested. [14] Other diseases of the gastrointestinal tract, such as inflammatory bowel disease and colon cancer, are also possible targets for orally available single-domain antibodies. [48]
Detergent-stable species targeting a surface protein of Malassezia furfur have been engineered for use in anti- dandruff shampoos. [10]
As an approach for photothermal therapy Nanobodies binding to the HER2 antigen, which is overexpressed in breast and ovarian cancer cells, were conjugated to branched gold nanoparticles (see figure). Tumor cells were destroyed photothermally using a laser in a test environment. [49]
Caplacizumab, a single-domain antibody targeting von Willebrand factor is in clinical trials for the prevention of thrombosis in patients with acute coronary syndrome. [50] A Phase II study examining ALX-0081 in high risk percutaneous coronary intervention has started in September 2009. [51]
Ablynx expects that their Nanobodies might cross the blood–brain barrier and permeate into large solid tumours more easily than whole antibodies, which would allow for the development of drugs against brain cancers. [48]
Nanobodies that tightly bind to the RBD domain of the spike protein of betacoronaviruses (including SARS-CoV-2 which causes COVID-19) and blocks interactions of spike with the cell receptor ACE2, has been recently identified [52] [18]
Application of various single domain antibodies (Nanobodies) for the prevention and treatment of infection by various highly pathogenic human coronaviruses (HPhCoVs) has been reported. The prospects, potency and challenges of deploying Nanobodies to bind and neutralize SARS-CoV-2 and akin have been recently highlighted. [53]
One of the most common causes of nagana – Trypanosoma brucei brucei – can be targeted by sdAbs. Stijlemans et al. 2004 succeeded in inducing effective sdAbs from rabbit and Camelus dromedarius by displaying a variable surface glycoprotein antigen to the vertebrates' immune systems using a phage. In the future, these therapies will surpass natural antibodies by reaching locations currently unreachable due to natural antibodies' larger size. [54]
References
- ^ a b Harmsen MM, De Haard HJ (November 2007). "Properties, production, and applications of camelid single-domain antibody fragments". Applied Microbiology and Biotechnology. 77 (1): 13–22. doi: 10.1007/s00253-007-1142-2. PMC 2039825. PMID 17704915.
- ^ English H, Hong J, Ho M (January 2020). "Ancient species offers contemporary therapeutics: an update on shark VNAR single domain antibody sequences, phage libraries and potential clinical applications". Antibody Therapeutics. 3 (1): 1–9. doi: 10.1093/abt/tbaa001. PMC 7034638. PMID 32118195.
- ^ Möller A, Pion E, Narayan V, Ball KL (December 2010). "Intracellular activation of interferon regulatory factor-1 by nanobodies to the multifunctional (Mf1) domain". The Journal of Biological Chemistry. 285 (49): 38348–38361. doi: 10.1074/jbc.M110.149476. PMC 2992268. PMID 20817723.
- ^ Ghannam A, Kumari S, Muyldermans S, Abbady AQ (March 2015). "Camelid nanobodies with high affinity for broad bean mottle virus: a possible promising tool to immunomodulate plant resistance against viruses". Plant Molecular Biology. 87 (4–5): 355–369. doi: 10.1007/s11103-015-0282-5. PMID 25648551. S2CID 17578111.
- ^ "Nanobodies herald a new era in cancer therapy". The Medical News. 12 May 2004.
- ^ "Pipeline". Ablynx. Retrieved 20 January 2010.
- ^ Gill, Victoria (22 September 2021). "Covid: Immune therapy from llamas shows promise". BBC News.
- ^ a b Buffington J, Duan Z, Kwon HJ, Hong J, Li D, Feng M, et al. (June 2023). "Identification of nurse shark VNAR single-domain antibodies targeting the spike S2 subunit of SARS-CoV-2". FASEB Journal. 37 (6): e22973. doi: 10.1096/fj.202202099RR. PMC 10715488. PMID 37191949. S2CID 258717083.
- ^ Hong J, Kwon HJ, Cachau R, Chen CZ, Butay KJ, Duan Z, et al. (May 2022). "Dromedary camel nanobodies broadly neutralize SARS-CoV-2 variants". Proceedings of the National Academy of Sciences of the United States of America. 119 (18): e2201433119. Bibcode: 2022PNAS..11901433H. doi: 10.1073/pnas.2201433119. PMC 9170159. PMID 35476528.
- ^ a b Dolk E, van der Vaart M, Lutje Hulsik D, Vriend G, de Haard H, Spinelli S, et al. (January 2005). "Isolation of llama antibody fragments for prevention of dandruff by phage display in shampoo". Applied and Environmental Microbiology. 71 (1): 442–450. Bibcode: 2005ApEnM..71..442D. doi: 10.1128/AEM.71.1.442-450.2005. PMC 544197. PMID 15640220.
- ^ a b Stanfield RL, Dooley H, Flajnik MF, Wilson IA (September 2004). "Crystal structure of a shark single-domain antibody V region in complex with lysozyme". Science. 305 (5691): 1770–1773. Bibcode: 2004Sci...305.1770S. doi: 10.1126/science.1101148. PMID 15319492. S2CID 25137728.
- ^ van der Linden RH, Frenken LG, de Geus B, Harmsen MM, Ruuls RC, Stok W, et al. (April 1999). "Comparison of physical chemical properties of llama VHH antibody fragments and mouse monoclonal antibodies". Biochimica et Biophysica Acta (BBA) - Protein Structure and Molecular Enzymology. 1431 (1): 37–46. doi: 10.1016/S0167-4838(99)00030-8. PMID 10209277.
- ^ Harmsen MM, van Solt CB, Hoogendoorn A, van Zijderveld FG, Niewold TA, van der Meulen J (November 2005). "Escherichia coli F4 fimbriae specific llama single-domain antibody fragments effectively inhibit bacterial adhesion in vitro but poorly protect against diarrhoea". Veterinary Microbiology. 111 (1–2): 89–98. doi: 10.1016/j.vetmic.2005.09.005. PMID 16221532.
- ^ a b Harmsen MM, van Solt CB, van Zijderveld-van Bemmel AM, Niewold TA, van Zijderveld FG (September 2006). "Selection and optimization of proteolytically stable llama single-domain antibody fragments for oral immunotherapy". Applied Microbiology and Biotechnology. 72 (3): 544–551. doi: 10.1007/s00253-005-0300-7. PMID 16450109. S2CID 2319210.
- ^ a b c d Ho M (June 2018). "Inaugural Editorial: Searching for Magic Bullets". Antibody Therapeutics. 1 (1): 1–5. doi: 10.1093/abt/tby001. PMC 6086361. PMID 30101214.
- ^ Desmyter A, Transue TR, Ghahroudi MA, Thi MH, Poortmans F, Hamers R, et al. (September 1996). "Crystal structure of a camel single-domain VH antibody fragment in complex with lysozyme". Nature Structural Biology. 3 (9): 803–811. doi: 10.1038/nsb0996-803. PMID 8784355. S2CID 10419927.
- ^ a b Feng M, Bian H, Wu X, Fu T, Fu Y, Hong J, et al. (January 2019). "Construction and next-generation sequencing analysis of a large phage-displayed VNAR single-domain antibody library from six naïve nurse sharks". Antibody Therapeutics. 2 (1): 1–11. doi: 10.1093/abt/tby011. PMC 6312525. PMID 30627698.
- ^ a b c Hong J, Kwon HJ, Cachau R, Chen CZ, Butay KJ, Duan Z, et al. (May 2022). "Dromedary camel nanobodies broadly neutralize SARS-CoV-2 variants". Proceedings of the National Academy of Sciences of the United States of America. 119 (18): e2201433119. Bibcode: 2022PNAS..11901433H. doi: 10.1073/pnas.2201433119. PMC 9170159. PMID 35476528.
- ^ a b Li N, Quan A, Li D, Pan J, Ren H, Hoeltzel G, et al. (April 2023). "The IgG4 hinge with CD28 transmembrane domain improves VHH-based CAR T cells targeting a membrane-distal epitope of GPC1 in pancreatic cancer". Nature Communications. 14 (1): 1986. Bibcode: 2023NatCo..14.1986L. doi: 10.1038/s41467-023-37616-4. PMC 10082787. PMID 37031249.
- ^ Arbabi Ghahroudi M, Desmyter A, Wyns L, Hamers R, Muyldermans S (September 1997). "Selection and identification of single domain antibody fragments from camel heavy-chain antibodies". FEBS Letters. 414 (3): 521–526. doi: 10.1016/S0014-5793(97)01062-4. PMID 9323027.
- ^ Li D, English H, Hong J, Liang T, Merlino G, Day CP, Ho M (March 2022). "A novel PD-L1-targeted shark VNAR single-domain-based CAR-T cell strategy for treating breast cancer and liver cancer". Molecular Therapy Oncolytics. 24: 849–863. doi: 10.1016/j.omto.2022.02.015. PMC 8917269. PMID 35317524.
- ^ Li D, Wang R, Liang T, Ren H, Park C, Tai CH, et al. (September 2023). "Camel nanobody-based B7-H3 CAR-T cells show high efficacy against large solid tumours". Nature Communications. 14 (1): 5920. Bibcode: 2023NatCo..14.5920L. doi: 10.1038/s41467-023-41631-w. PMC 10517151. PMID 37739951.
- ^ Zhang YF, Sun Y, Hong J, Ho M (January 2023). "Humanization of the Shark VNAR Single Domain Antibody Using CDR Grafting". Current Protocols. 3 (1): e630. doi: 10.1002/cpz1.630. PMC 9813873. PMID 36594750.
- ^ Ward ES, Güssow D, Griffiths AD, Jones PT, Winter G (October 1989). "Binding activities of a repertoire of single immunoglobulin variable domains secreted from Escherichia coli". Nature. 341 (6242): 544–546. Bibcode: 1989Natur.341..544W. doi: 10.1038/341544a0. PMID 2677748. S2CID 4329440.
- ^ a b Feng R, Wang R, Hong J, Dower CM, Croix BS, Ho M (January 2020). "Isolation of rabbit single domain antibodies to B7-H3 via protein immunization and phage display". Antibody Therapeutics. 3 (1): 10–17. doi: 10.1093/abt/tbaa002. PMC 7052794. PMID 32166218.
- ^ a b c Feng M, Gao W, Wang R, Chen W, Man YG, Figg WD, et al. (March 2013). "Therapeutically targeting glypican-3 via a conformation-specific single-domain antibody in hepatocellular carcinoma". Proceedings of the National Academy of Sciences of the United States of America. 110 (12): E1083–E1091. Bibcode: 2013PNAS..110E1083F. doi: 10.1073/pnas.1217868110. PMC 3607002. PMID 23471984.
- ^ Holt LJ, Herring C, Jespers LS, Woolven BP, Tomlinson IM (November 2003). "Domain antibodies: proteins for therapy". Trends in Biotechnology. 21 (11): 484–490. doi: 10.1016/j.tibtech.2003.08.007. PMID 14573361.
- ^ Borrebaeck CA, Ohlin M (December 2002). "Antibody evolution beyond Nature". Nature Biotechnology. 20 (12): 1189–1190. doi: 10.1038/nbt1202-1189. PMID 12454662. S2CID 7937915.
- ^ a b Tang Z, Feng M, Gao W, Phung Y, Chen W, Chaudhary A, et al. (April 2013). "A human single-domain antibody elicits potent antitumor activity by targeting an epitope in mesothelin close to the cancer cell surface". Molecular Cancer Therapeutics. 12 (4): 416–426. doi: 10.1158/1535-7163.MCT-12-0731. PMC 3624043. PMID 23371858.
- ^ Li N, Fu H, Hewitt SM, Dimitrov DS, Ho M (August 2017). "Therapeutically targeting glypican-2 via single-domain antibody-based chimeric antigen receptors and immunotoxins in neuroblastoma". Proceedings of the National Academy of Sciences of the United States of America. 114 (32): E6623–E6631. Bibcode: 2017PNAS..114E6623L. doi: 10.1073/pnas.1706055114. PMC 5559039. PMID 28739923.
- ^ a b Gao W, Tang Z, Zhang YF, Feng M, Qian M, Dimitrov DS, Ho M (March 2015). "Immunotoxin targeting glypican-3 regresses liver cancer via dual inhibition of Wnt signalling and protein synthesis". Nature Communications. 6: 6536. Bibcode: 2015NatCo...6.6536G. doi: 10.1038/ncomms7536. PMC 4357278. PMID 25758784.
- ^ Wang C, Gao W, Feng M, Pastan I, Ho M (May 2017). "Construction of an immunotoxin, HN3-mPE24, targeting glypican-3 for liver cancer therapy". Oncotarget. 8 (20): 32450–32460. doi: 10.18632/oncotarget.10592. PMC 5464801. PMID 27419635.
- ^ Fleming BD, Urban DJ, Hall MD, Longerich T, Greten TF, Pastan I, Ho M (May 2020). "Engineered Anti-GPC3 Immunotoxin, HN3-ABD-T20, Produces Regression in Mouse Liver Cancer Xenografts Through Prolonged Serum Retention". Hepatology. 71 (5): 1696–1711. doi: 10.1002/hep.30949. PMC 7069773. PMID 31520528.
- ^ Kolluri A, Li D, Li N, Duan Z, Roberts LR, Ho M (February 2023). "Human VH-based chimeric antigen receptor T cells targeting glypican 3 eliminate tumors in preclinical models of HCC". Hepatology Communications. 7 (2): e0022. doi: 10.1097/HC9.0000000000000022. PMC 9851680. PMID 36691969.
- ^ Li N, Wei L, Liu X, Bai H, Ye Y, Li D, et al. (October 2019). "A Frizzled-Like Cysteine-Rich Domain in Glypican-3 Mediates Wnt Binding and Regulates Hepatocellular Carcinoma Tumor Growth in Mice". Hepatology. 70 (4): 1231–1245. doi: 10.1002/hep.30646. PMC 6783318. PMID 30963603.
- ^ Muyldermans S (2013). "Nanobodies: natural single-domain antibodies". Annual Review of Biochemistry. 82: 775–797. doi: 10.1146/annurev-biochem-063011-092449. PMID 23495938.
- ^ a b Gulati S, Jin H, Masuho I, Orban T, Cai Y, Pardon E, et al. (May 2018). "Targeting G protein-coupled receptor signaling at the G protein level with a selective nanobody inhibitor". Nature Communications. 9 (1): 1996. Bibcode: 2018NatCo...9.1996G. doi: 10.1038/s41467-018-04432-0. PMC 5959942. PMID 29777099.
- ^ Rothbauer U, Zolghadr K, Tillib S, Nowak D, Schermelleh L, Gahl A, et al. (November 2006). "Targeting and tracing antigens in live cells with fluorescent nanobodies". Nature Methods. 3 (11): 887–889. doi: 10.1038/nmeth953. PMID 17060912. S2CID 29985078.
- ^ Rothbauer U, Zolghadr K, Muyldermans S, Schepers A, Cardoso MC, Leonhardt H (February 2008). "A versatile nanotrap for biochemical and functional studies with fluorescent fusion proteins". Molecular & Cellular Proteomics. 7 (2): 282–289. doi: 10.1074/mcp.M700342-MCP200. PMID 17951627.
- ^ Ries J, Kaplan C, Platonova E, Eghlidi H, Ewers H (June 2012). "A simple, versatile method for GFP-based super-resolution microscopy via nanobodies". Nature Methods. 9 (6): 582–584. doi: 10.1038/nmeth.1991. PMID 22543348. S2CID 12190938.
- ^ Fang T, Lu X, Berger D, Gmeiner C, Cho J, Schalek R, et al. (December 2018). "Nanobody immunostaining for correlated light and electron microscopy with preservation of ultrastructure". Nature Methods. 15 (12): 1029–1032. doi: 10.1038/s41592-018-0177-x. PMC 6405223. PMID 30397326.
- ^ Saerens D, Frederix F, Reekmans G, Conrath K, Jans K, Brys L, et al. (December 2005). "Engineering camel single-domain antibodies and immobilization chemistry for human prostate-specific antigen sensing". Analytical Chemistry. 77 (23): 7547–7555. doi: 10.1021/ac051092j. PMID 16316161.
- ^ Koide S (August 2009). "Engineering of recombinant crystallization chaperones". Current Opinion in Structural Biology. 19 (4): 449–457. doi: 10.1016/j.sbi.2009.04.008. PMC 2736338. PMID 19477632.
- ^ Ibañez LI, De Filette M, Hultberg A, Verrips T, Temperton N, Weiss RA, et al. (April 2011). "Nanobodies with in vitro neutralizing activity protect mice against H5N1 influenza virus infection". The Journal of Infectious Diseases. 203 (8): 1063–1072. doi: 10.1093/infdis/jiq168. hdl: 1854/LU-1212323. PMID 21450996.
- ^ Hussack G, Arbabi-Ghahroudi M, van Faassen H, Songer JG, Ng KK, MacKenzie R, Tanha J (March 2011). "Neutralization of Clostridium difficile toxin A with single-domain antibodies targeting the cell receptor binding domain". The Journal of Biological Chemistry. 286 (11): 8961–8976. doi: 10.1074/jbc.M110.198754. PMC 3058971. PMID 21216961.
- ^ Fang T, Duarte JN, Ling J, Li Z, Guzman JS, Ploegh HL (February 2016). "Structurally Defined αMHC-II Nanobody-Drug Conjugates: A Therapeutic and Imaging System for B-Cell Lymphoma". Angewandte Chemie. 55 (7): 2416–2420. doi: 10.1002/anie.201509432. PMC 4820396. PMID 26840214.
- ^ Fang T, Van Elssen CH, Duarte JN, Guzman JS, Chahal JS, Ling J, Ploegh HL (August 2017). "Targeted antigen delivery by an anti-class II MHC VHH elicits focused αMUC1(Tn) immunity". Chemical Science. 8 (8): 5591–5597. doi: 10.1039/c7sc00446j. PMC 5618788. PMID 28970938.
- ^ a b "Nanobodies". Nanobody.org. 2006.
- ^ Van de Broek B, Devoogdt N, D'Hollander A, Gijs HL, Jans K, Lagae L, et al. (June 2011). "Specific cell targeting with nanobody conjugated branched gold nanoparticles for photothermal therapy". ACS Nano. 5 (6): 4319–4328. doi: 10.1021/nn1023363. PMID 21609027.
- ^ "Ablynx Announces Interim Results of First NANOBODY® Phase I Study of,ALX-0081 (ANTI-VWF)". Bio-Medicine.org. 2 July 2007.
- ^ Clinical trial number NCT01020383 for "Comparative Study of ALX-0081 Versus GPIIb/IIIa Inhibitor in High Risk Percutaneous Coronary Intervention (PCI) Patients" at ClinicalTrials.gov
- ^ Wrapp D, De Vlieger D, Corbett KS, Torres GM, Wang N, Van Breedam W, et al. (May 2020). "Structural Basis for Potent Neutralization of Betacoronaviruses by Single-Domain Camelid Antibodies". Cell. 181 (5): 1004–1015.e15. doi: 10.1016/j.cell.2020.04.031. PMC 7199733. PMID 32375025.
- ^ Konwarh R (23 June 2020). "Nanobodies: Prospects of Expanding the Gamut of Neutralizing Antibodies Against the Novel Coronavirus, SARS-CoV-2". Frontiers in Immunology. 11: 1531. doi: 10.3389/fimmu.2020.01531. PMC 7324746. PMID 32655584.
-
^ Muyldermans S (2013-06-02). "Nanobodies: natural single-domain antibodies". Annual Review of Biochemistry. 82 (1).
Annual Reviews: 775–797.
doi:
10.1146/annurev-biochem-063011-092449.
PMID
23495938. p. 788:
Moreover...
