The Schlenk line (also vacuum gas manifold) is a commonly used chemistry apparatus developed by Wilhelm Schlenk. [1] It consists of a dual manifold with several ports. [2] One manifold is connected to a source of purified inert gas, while the other is connected to a vacuum pump. The inert-gas line is vented through an oil bubbler, while solvent vapors and gaseous reaction products are prevented from contaminating the vacuum pump by a liquid-nitrogen or dry-ice/ acetone cold trap. Special stopcocks or Teflon taps allow vacuum or inert gas to be selected without the need for placing the sample on a separate line. [3]
Schlenk lines are useful for safely and successfully manipulating moisture- and air-sensitive compounds. The vacuum is also often used to remove the last traces of solvent from a sample. Vacuum and gas manifolds often have many ports and lines, and with care, it is possible for several reactions or operations to be run simultaneously.
When the reagents are highly susceptible to oxidation, traces of oxygen may pose a problem. Then, for the removal of oxygen below the ppm level, the inert gas needs to be purified by passing it through a deoxygenation catalyst. [4] This is usually a column of copper(I) or manganese(II) oxide, which reacts with oxygen traces present in the inert gas.
Techniques
The main techniques associated with the use of a Schlenk line include:
- counterflow additions, where air-stable reagents are added to the reaction vessel against a flow of inert gas;
- the use of syringes and rubber septa to transfer liquids and solutions; [5]
- cannula transfer, where liquids or solutions of air-sensitive reagents are transferred between different vessels stoppered with septa using a long thin tube known as a cannula. Liquid flow is supported by vacuum or inert-gas pressure. [6]
Glassware are usually connected by tightly fitting and greased ground glass joints. Round bends of glass tubing with ground glass joints may be used to adjust the orientation of various vessels. Glassware is necessarily purged of outside air by alternating application of vacuum and inert gas. The solvents and reagents that are used are also purged of air and water using various methods.
Filtration under inert conditions poses a special challenge that is usually tackled with specialized glassware. A Schlenk filter consists of sintered glass funnel fitted with joints and stopcocks. By fitting the pre-dried funnel and receiving flask to the reaction flask against a flow of nitrogen, carefully inverting the set-up, and turning on the vacuum appropriately, the filtration may be accomplished with minimal exposure to air.
Dangers
The main dangers associated with the use of a Schlenk line are the risks of an implosion or explosion. An implosion can occur due to the use of vacuum and flaws in the glass apparatus.
An explosion can occur due to the common use of liquid nitrogen in the cold trap, used to protect the vacuum pump from solvents. If a reasonable amount of air is allowed to enter the Schlenk line, liquid oxygen can condense into the cold trap as a pale blue liquid. An explosion may occur due to reaction of the liquid oxygen with any organic compounds also in the trap.
Gallery
-
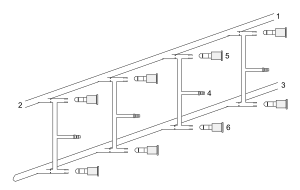
Vacuum/gas manifold setup: 1 inert gas in, 2 inert gas out (to bubbler), 3 vacuum (to cold traps) 4 reaction line, 5 Teflon tap to gas, 6 Teflon tap to vacuum
-
Vacuum/gas manifold setup: 1 inert gas in, 2 inert gas out (to bubbler), 3 vacuum (to cold traps), 4 reaction line, 5 double oblique stopcock (i.e. a glass tap with 2 separate parallel "channels/lines" that run diagonal to the axis of the tap)
-
The two reactants for an aldol reaction are prepared in adjacent flasks, ready for one to be transferred to the other while maintaining air-free conditions
-
A yellow suspension is filtered through a sintered-glass funnel into another Schlenk flask under air-free conditions
See also
-
Air-free technique gives a broad overview of methods including:
- Glovebox – used to manipulate air-sensitive (oxygen- or moisture-sensitive) chemicals.
- Schlenk flask – reaction vessel for handling air-sensitive compounds.
- Perkin triangle – used for the distillation of air-sensitive compounds.
References
-
^ The prototype of what became the "Schlenk line" appears in: Schlenk, Wilhelm; Thal, Alexander (1913).
"Über Metallketyle, eine große Klasse von Verbindungen mit dreiwertigem Kohlenstoff. II" [On metal ketyls, a large class of compounds with trivalent carbon. II.]. Berichte der Deutschen Chemischen Gesellschaft (in German). 46 (3): 2840–2854.
doi:
10.1002/cber.19130460356. See illustrations on pp. 2844–2845.
- The illustrations from Schlenk's 1913 paper are reproduced on pp. 960-963 of: Schlenk, Wilhelm (1924). "Organometallverbindungen [Organometallic compounds]". In Weyl, Josef (ed.). Die Methoden der Organischen Chemie [Methods of Organic Chemistry] (in German). Vol. 4 (2nd ed.). Leipzig, Germany: Georg Thieme. pp. 720–978.
- See also: Tidwell, Thomas T. (2001). "Wilhelm Schlenk: The Man Behind the Flask" (PDF). Angewandte Chemie International Edition. 40 (2): 331–337. doi: 10.1002/1521-3773(20010119)40:2<331::AID-ANIE331>3.0.CO;2-E.
- ^ Craig M. Davis and Kelly A. Curran (November 2007). "Manipulation of a Schlenk Line: Preparation of Tetrahydrofuran Complexes of Transition-Metal Chlorides" (abstract page). Journal of Chemical Education. 84 (11): 1822–3. Bibcode: 2007JChEd..84.1822D. doi: 10.1021/ed084p1822.
- ^ Borys, Andryj M. (2023). "An Illustrated Guide to Schlenk Line Techniques". Organometallics. 42 (3): 182–196. doi: 10.1021/acs.organomet.2c00535. S2CID 256409354.
- ^ C. R. McIlwrick and C. S. Phillips The removal of oxygen from gas streams: applications in catalysis and gas chromatography, Journal of Physics E: Scientific Instruments, 1973, 6:12, 1208–10.
- ^ Johansen, Martin B.; Kondrup, Jens C.; Hinge, Mogens; Lindhardt, Anders T. (13 June 2018). "Improved Safety during Transfer of Pyrophoric tert-Butyllithium from Flasks with Protective Seals". Organic Process Research & Development. 22 (7): 903–905. doi: 10.1021/acs.oprd.8b00151. S2CID 103573742.
- ^ Brown, H. C. "Organic Syntheses via Boranes" John Wiley & Sons, Inc., New York: 1975. ISBN 0-471-11280-1.
Further reading
- Sella, Andrea (January 2008). "Schlenk Apparatus". Chemistry World: 69. Retrieved 2008-01-30.
- Tidwell, Thomas (2001). "Wilhelm Schlenk: The Man Behind the Flask". Angewandte Chemie International Edition. 40 (2): 331–337. doi: 10.1002/1521-3773(20010119)40:2<331::AID-ANIE331>3.0.CO;2-E. PMID 11180319.
- Jürgen Heck. "The Integrated Synthesis Course: Schlenk Technique" (PDF). University of Hamburg. Archived from the original (reprint at Norwegian University of Science and Technology) on 2008-03-09.
- "Handling Air-Sensitive Reagents" (PDF). Sigma-Aldrich.
- "Handling Air-Sensitive Reagents" Sigma-Aldrich.
External links
- Rob Toreki (25 May 2004). "Schlenk Lines and Vacuum Lines". The Glassware Gallery. Interactive Learning Paradigms Incorporated.
- Preparation of a Manganese oxide column for inert gas purification from oxygen traces