| REV protein bound to RRE mRNA | |||||||||
|---|---|---|---|---|---|---|---|---|---|
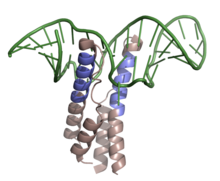 Shown above is the RNP complex formed by HIV-1 REV binding to the RRE upon mRNA of the env gene. Highlighted in slate is the ARM of the RNA binding domain, while colored in green is the mRNA with secondary stem-loop-like structure (PDB 4PMI). | |||||||||
| Identifiers | |||||||||
| Symbol | REV | ||||||||
| Pfam | PF00424 | ||||||||
| InterPro | IPR000625 | ||||||||
| SCOP2 | 484d / SCOPe / SUPFAM | ||||||||
| |||||||||
Rev is a transactivating protein that is essential to the regulation of HIV-1 (and other lentiviral) protein expression. A nuclear localization signal is encoded in the rev gene, which allows the Rev protein to be localized to the nucleus, where it is involved in the export of unspliced and incompletely spliced mRNAs. In the absence of Rev, mRNAs of the HIV-1 late (structural) genes are retained in the nucleus, preventing their translation.
History
A novel protein was found to be involved in the translation of gag and env mRNA. The unknown protein functioned by removing repression of regulatory sequences and was named Art (anti-repression transactivator). [1] Later studies suggested that the protein was involved in regulation of the RNA splicing mechanism. Therefore, the name of the protein was modified from Art to Trs (transregulator of splicing). [2] The most recent studies have shown that the protein has multiple functions in the regulation of HIV-1 proteins, and its name has been changed to Rev (regulator of expression of virion proteins), which more generally describes its function. [3]
Structure
Rev is a 13-k Da protein [4] that is composed of 116 amino acids. [5] Rev's sequence contains two specific domains which contribute to its nuclear import and export. The protein typically performs its function as a tetramer.[ citation needed]
Arginine-rich motif
The N-terminal region of Rev contains an arginine-rich sequence. The arginine-rich motif (ARM) is located between amino acids 38–49 of the rev gene [6] and forms an alpha-helical secondary structure. [7] The ARM is a highly specific sequence which allows for the multimerization of Rev proteins, prior to RNA binding. A single base substitution alters Rev's ability to form a tetramer. [8] The arginine-rich domain of Rev interacts with the rev-binding element (RBE), which is part of the HIV Rev response element (RRE) located in an intron downstream of the env gene. [9] The alpha-helical secondary structure specifically can be considered a helix-loop-helix motif, which allows the REV protein to stably bind to the RRE RNA to form the ribonucleoprotein complex. [10] The domain also contains a nuclear localization signal. [11]
Rev-activation domain (Nuclear export signal)
Rev's nuclear export signal is located in residues 71–82 [12] of the C-terminal region [13] and is leucine-rich. [14] Binding of Rev to viral RNAs containing the RRE allows for mRNA export out of the nucleus and into the cytoplasm by a mechanism different than that of cellular mRNAs.[ citation needed]
Function
HIV-1 regulatory proteins (including Rev) are translated from completely processed mRNA transcripts, while structural proteins are translated from incompletely spliced transcripts. Completely spliced transcripts are exported from the nucleus to the cytoplasm by the same mechanism as cellular mRNA. However, Rev is needed to export incompletely spliced mRNAs in order to produce the viral structural proteins.[ citation needed]
Rev localization to the nucleus
The arginine-rich domain of the Rev protein, containing a nuclear localization signal (NLS), allows Rev to enter the nucleus. Entry requires binding between a Rev multimer, Ran-GDP, and importin-β (a nuclear transport factor). [15] The Rev NLS is a highly similar sequence to that of the importin-β-binding site present within importin-α, [16] which allows for the interaction between Rev and importin-β. The NLS overlaps with the sequence required for RNA-binding. [17] This prevents the NLS from counteracting the export of RRE-containing mRNA transcripts.[ citation needed]

Binding of Rev to the RRE
The rev response element (RRE) is a 240 base-pair sequence located in the second intron of the HIV-1 genome, immediately downstream of the env gene. [18] The RRE remains functional if translocated, but needs to remain in the same orientation (cannot be inverted). The RRE is retained by incompletely processed mRNA transcripts. The secondary structure of the RRE creates eight stem-loops. Rev initially binds to the purine-rich stem-loop IIB, [19] then binds to a secondary site in stem-loop I. [20]
Within this purine-rich stem-loop, IIB, are non-canonical base pairs that form as a result of the mRNA stem loop-secondary structure. These base pairs include guanine-adenine (nucleotides 47 and 73, respectively) and guanine-guanine (nucleotides 48–71, respectively). The two base pairs are separated by a non-stacked and bulging uridine that points outwards, away from the ARM-RNA interactions.
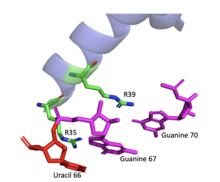
The ARM contains residues R35 and R39 that make base-specific contacts with residues on the RRE mRNA, specifically to bases uracil 66, guanine 67, and guanine 70, respectively. On the opposite side of these bases, residues N40 and R44 make base-specific contacts with nucleotides uracil 45, guanine 46, guanine 47, and adenine 73. In addition to these stabilizing contacts, additional Arg residues within the ARM, as well as T34, make nonspecific contacts with bases on the mRNA. [21]

The RRE sequence is cis-acting, and is necessary to achieve high levels of env mRNA in the cytoplasm. [22] The RRE also facilitates multimerization of the Rev proteins, which is required for Rev binding and function. [23] The Rev protein binds unspliced gag and pol transcripts and incompletely spliced env, vif, vpr and vpu transcripts at the RRE, facilitating export to the cytoplasm. [24]
Genomic export from the nucleus
Rev is continuously shuttled between the cytoplasm and nucleus. The shuttling of Rev is regulated by its nuclear localization signal and its nuclear export signal. Once Rev is inside the nucleus, Ran-GDP is phosphorylated into Ran-GTP, causing the importation complex to disassemble. Upon disassembly, Rev's NES forms a new complex with CRM1 ( exportin-1) and Ran-GTP at the RRE sequence within incompletely spliced transcripts. Following assembly of the complex, the intron-containing RNAs are exported from the nucleus into the cytoplasm. [25] Once the pre-mRNAs are in the cytoplasm, Rev dissociates, revealing the NLS. [26] Exposure of the NLS allows for Rev interaction with importin-β in order to shuttle Rev back to the nucleus.[ citation needed]
Rev-directed export of viral RNAs is similar to the mechanism by which snRNAs and the 5s rRNAs are exported, as opposed to the mechanism for export of cellular mRNAs. Rev is able to facilitate export of pre-mRNA transcripts that would otherwise typically remain in the nucleus, suggesting that the Rev NES is dominant over nuclear retention. [27]
Regulation of HIV gene expression
Rev acts post-transcriptionally to positively regulate the expression of structural genes and to negatively regulate the expression of regulatory genes. Rev positively regulates the expression of gag, pol, and env. Rev-mediated export from the nucleus increases cytoplasmic levels of the structural mRNAs (gag, pol, and env). [28] Gag, pol and env expression is lower in the absence of Rev and higher in the presence of Rev. [29] Rev negatively regulates the expression of the regulatory genes (rev and tat) by creating a negative feedback loop, which regulates Rev production. There is a decrease in Rev production when Rev protein levels are higher than is necessary for the given amount of HIV-1 genome encoded. Rev also decreases the quantity of completely spliced viral messages expressed by exporting pre-mRNA before it can be spliced. This results in decreased expression of the regulatory proteins, Rev and Tat. Since Rev is continuously shuttled between the nucleus and cytoplasm, small amounts of the protein are able to impact many mRNA transcripts. Maintenance of the proper balance between early and late viral gene quantities leads to an overall increase in virion production. [30]
Transition from early to late phase HIV-1 genes
HIV-1 genes are expressed from either completely spliced RNA or from intron-containing RNA. The export of fully spliced mRNAs (early, regulatory genes) occurs in the same manner as the normal export of cellular mRNAs. On the other hand, unspliced and incompletely spliced mRNAs which code for the late, structural proteins are Rev-dependent. The Rev protein is expressed as an early gene from completely spliced transcripts, so the expression of late phase structural proteins cannot occur until an initial amount of Rev is produced. [31]
Rev as a target for antiviral therapies
Since Rev is absolutely necessary for HIV-1 replication and it is expressed early on in infection, it has been suggested that Rev is a good target for antiviral therapies.[ citation needed]
Leptomycin B (LMB) binds to CRM1 which prevents the formation of the complex required for export(CRM1/NES/RanGTP/RRE) and ultimately reduces the production of incompletely spliced RNAs. [32] [33] Therefore, structural proteins, which are necessary for virion assembly, are not produced.
It has been shown that various organic compounds have the ability to target the Rev/RRE interaction. Neomycin B, diphenylfuran cation, and proflavine are small molecules that can prevent Rev from binding to the RRE sequence. [34] [35] [36] If Rev is incapable of binding to the RRE on the pre-mRNA, the RNA will not be exported to the cytoplasm, also resulting in lack of necessary structural proteins.[ citation needed]
Other therapies target the Rev protein itself, since it is an essential component of HIV-1 infection. M10 is a mutated form of Rev and has a single amino acid substitution ( Aspartic acid to Leucine). If delivered to cells, Rev M10 will compete with the wild-type Rev protein for the RRE binding site, and therefore decrease Rev's normal cellular functions. [37]
Dihydrovaltrate was also identified as a Rev-export inhibitory congener.[ citation needed]
References
- ^ Sodroski J, Goh WC, Rosen C, Dayton A, Terwilliger E, Haseltine W (May 1986). "A second post-transcriptional trans-activator gene required for HTLV-III replication". Nature. 321 (6068): 412–7. Bibcode: 1986Natur.321..412S. doi: 10.1038/321412a0. PMID 3012355. S2CID 4306352.
- ^ Feinberg MB, Jarrett RF, Aldovini A, Gallo RC, Wong-Staal F (September 1986). "HTLV-III expression and production involve complex regulation at the levels of splicing and translation of viral RNA". Cell. 46 (6): 807–17. doi: 10.1016/0092-8674(86)90062-0. PMID 3638988. S2CID 37813869.
- ^ Gallo R, Wong-Staal F, Montagnier L, Haseltine WA, Yoshida M (June 1988). "HIV/HTLV gene nomenclature". Nature. 333 (6173): 504. Bibcode: 1988Natur.333..504G. doi: 10.1038/333504a0. PMID 2836736. S2CID 33832642.
- ^ Cochrane A, Kramer R, Ruben S, Levine J, Rosen CA (July 1989). "The human immunodeficiency virus rev protein is a nuclear phosphoprotein". Virology. 171 (1): 264–6. doi: 10.1016/0042-6822(89)90535-7. PMID 2741343.
- ^ Surendran R, Herman P, Cheng Z, Daly TJ, Ching Lee J (March 2004). "HIV Rev self-assembly is linked to a molten-globule to compact structural transition". Biophysical Chemistry. 108 (1–3): 101–19. doi: 10.1016/j.bpc.2003.10.013. PMID 15043924.
- ^ Kjems J, Calnan BJ, Frankel AD, Sharp PA (March 1992). "Specific binding of a basic peptide from HIV-1 Rev". The EMBO Journal. 11 (3): 1119–29. doi: 10.1002/j.1460-2075.1992.tb05152.x. PMC 556554. PMID 1547776.
- ^ Hope TJ (May 1999). "The ins and outs of HIV Rev". Archives of Biochemistry and Biophysics. 365 (2): 186–91. doi: 10.1006/abbi.1999.1207. PMID 10328811.
- ^ Zapp ML, Hope TJ, Parslow TG, Green MR (September 1991). "Oligomerization and RNA binding domains of the type 1 human immunodeficiency virus Rev protein: a dual function for an arginine-rich binding motif". Proceedings of the National Academy of Sciences of the United States of America. 88 (17): 7734–8. Bibcode: 1991PNAS...88.7734Z. doi: 10.1073/pnas.88.17.7734. PMC 52377. PMID 1715576.
- ^ Surendran R, Herman P, Cheng Z, Daly TJ, Ching Lee J (March 2004). "HIV Rev self-assembly is linked to a molten-globule to compact structural transition". Biophysical Chemistry. 108 (1–3): 101–19. doi: 10.1016/j.bpc.2003.10.013. PMID 15043924.
- ^ Auer, Manfred; Gremlich, Hans Ulrich; Seifert, Jan Marcus; Daly, Thomas J.; Parslow, Tristram G.; Casari, Georg; Gstach, Hubert (1994-03-15). "Helix-Loop-Helix Motif in HIV-1 Rev". Biochemistry. 33 (10): 2988–2996. doi: 10.1021/bi00176a031. ISSN 0006-2960. PMID 7510518.
- ^ Izaurralde E, Adam S (April 1998). "Transport of macromolecules between the nucleus and the cytoplasm". RNA. 4 (4): 351–64. PMC 1369623. PMID 9630243.
- ^ Malim MH, McCarn DF, Tiley LS, Cullen BR (August 1991). "Mutational definition of the human immunodeficiency virus type 1 Rev activation domain". Journal of Virology. 65 (8): 4248–54. doi: 10.1128/JVI.65.8.4248-4254.1991. PMC 248862. PMID 2072452.
- ^ Meyer BE, Meinkoth JL, Malim MH (April 1996). "Nuclear transport of human immunodeficiency virus type 1, visna virus, and equine infectious anemia virus Rev proteins: identification of a family of transferable nuclear export signals". Journal of Virology. 70 (4): 2350–9. doi: 10.1128/JVI.70.4.2350-2359.1996. PMC 190077. PMID 8642662.
- ^ Fornerod M, Ohno M, Yoshida M, Mattaj IW (September 1997). "CRM1 is an export receptor for leucine-rich nuclear export signals". Cell. 90 (6): 1051–60. doi: 10.1016/s0092-8674(00)80371-2. PMID 9323133. S2CID 15119502.
- ^ Pollard VW, Malim MH (October 1998). "The HIV-1 Rev protein". Annual Review of Microbiology. 52 (1): 491–532. doi: 10.1146/annurev.micro.52.1.491. PMID 9891806.
- ^ Görlich D, Henklein P, Laskey RA, Hartmann E (April 1996). "A 41 amino acid motif in importin-alpha confers binding to importin-beta and hence transit into the nucleus". The EMBO Journal. 15 (8): 1810–7. doi: 10.1002/j.1460-2075.1996.tb00530.x. PMC 450097. PMID 8617226.
- ^ Kubota S, Siomi H, Satoh T, Endo S, Maki M, Hatanaka M (August 1989). "Functional similarity of HIV-I rev and HTLV-I rex proteins: identification of a new nucleolar-targeting signal in rev protein". Biochemical and Biophysical Research Communications. 162 (3): 963–70. doi: 10.1016/0006-291x(89)90767-5. PMID 2788417.
- ^ Hadzopoulou-Cladaras M, Felber BK, Cladaras C, Athanassopoulos A, Tse A, Pavlakis GN (March 1989). "The rev (trs/art) protein of human immunodeficiency virus type 1 affects viral mRNA and protein expression via a cis-acting sequence in the env region". Journal of Virology. 63 (3): 1265–74. doi: 10.1128/JVI.63.3.1265-1274.1989. PMC 247823. PMID 2783738.
- ^ Nakatani K, Horie S, Goto Y, Kobori A, Hagihara S (August 2006). "Evaluation of mismatch-binding ligands as inhibitors for Rev-RRE interaction". Bioorganic & Medicinal Chemistry. 14 (15): 5384–8. doi: 10.1016/j.bmc.2006.03.038. PMID 16603366.
- ^ Daugherty MD, D'Orso I, Frankel AD (September 2008). "A solution to limited genomic capacity: using adaptable binding surfaces to assemble the functional HIV Rev oligomer on RNA". Molecular Cell. 31 (6): 824–34. doi: 10.1016/j.molcel.2008.07.016. PMC 2651398. PMID 18922466.
- ^ Rausch, Jason; Grice, Stuart (June 2015). "HIV Rev Assembly on the Rev Response Element (RRE): A Structural Perspective". Viruses. 7 (6): 3053–3075. doi: 10.3390/v7062760. PMC 4488727. PMID 26075509.
- ^ Hammarskjöld ML, Heimer J, Hammarskjöld B, Sangwan I, Albert L, Rekosh D (May 1989). "Regulation of human immunodeficiency virus env expression by the rev gene product". Journal of Virology. 63 (5): 1959–66. doi: 10.1128/JVI.63.5.1959-1966.1989. PMC 250609. PMID 2704072.
- ^ Zapp ML, Hope TJ, Parslow TG, Green MR (September 1991). "Oligomerization and RNA binding domains of the type 1 human immunodeficiency virus Rev protein: a dual function for an arginine-rich binding motif". Proceedings of the National Academy of Sciences of the United States of America. 88 (17): 7734–8. Bibcode: 1991PNAS...88.7734Z. doi: 10.1073/pnas.88.17.7734. PMC 52377. PMID 1715576.
- ^ Favaro JP, Borg KT, Arrigo SJ, Schmidt MG (September 1998). "Effect of Rev on the intranuclear localization of HIV-1 unspliced RNA". Virology. 249 (2): 286–96. doi: 10.1006/viro.1998.9312. PMID 9791020.
- ^ Hope TJ (May 1997). "Viral RNA export". Chemistry & Biology. 4 (5): 335–44. doi: 10.1016/s1074-5521(97)90124-1. PMID 9195877.
- ^ Kubota S, Siomi H, Satoh T, Endo S, Maki M, Hatanaka M (August 1989). "Functional similarity of HIV-I rev and HTLV-I rex proteins: identification of a new nucleolar-targeting signal in rev protein". Biochemical and Biophysical Research Communications. 162 (3): 963–70. doi: 10.1016/0006-291x(89)90767-5. PMID 2788417.
- ^ Fischer U, Huber J, Boelens WC, Mattaj IW, Lührmann R (August 1995). "The HIV-1 Rev activation domain is a nuclear export signal that accesses an export pathway used by specific cellular RNAs". Cell. 82 (3): 475–83. doi: 10.1016/0092-8674(95)90436-0. PMID 7543368. S2CID 15414237.
- ^ Feinberg MB, Jarrett RF, Aldovini A, Gallo RC, Wong-Staal F (September 1986). "HTLV-III expression and production involve complex regulation at the levels of splicing and translation of viral RNA". Cell. 46 (6): 807–17. doi: 10.1016/0092-8674(86)90062-0. PMID 3638988. S2CID 37813869.
- ^ Hadzopoulou-Cladaras M, Felber BK, Cladaras C, Athanassopoulos A, Tse A, Pavlakis GN (March 1989). "The rev (trs/art) protein of human immunodeficiency virus type 1 affects viral mRNA and protein expression via a cis-acting sequence in the env region". Journal of Virology. 63 (3): 1265–74. doi: 10.1128/JVI.63.3.1265-1274.1989. PMC 247823. PMID 2783738.
- ^ Hope TJ (May 1999). "The ins and outs of HIV Rev". Archives of Biochemistry and Biophysics. 365 (2): 186–91. doi: 10.1006/abbi.1999.1207. PMID 10328811.
- ^ Felber BK, Drysdale CM, Pavlakis GN (August 1990). "Feedback regulation of human immunodeficiency virus type 1 expression by the Rev protein". Journal of Virology. 64 (8): 3734–41. doi: 10.1128/JVI.64.8.3734-3741.1990. PMC 249668. PMID 2196381.
- ^ Fornerod M, Ohno M, Yoshida M, Mattaj IW (September 1997). "CRM1 is an export receptor for leucine-rich nuclear export signals". Cell. 90 (6): 1051–60. doi: 10.1016/s0092-8674(00)80371-2. PMID 9323133. S2CID 15119502.
- ^ Xiao G, Kumar A, Li K, Rigl CT, Bajic M, Davis TM, et al. (May 2001). "Inhibition of the HIV-1 rev-RRE complex formation by unfused aromatic cations". Bioorganic & Medicinal Chemistry. 9 (5): 1097–113. doi: 10.1016/s0968-0896(00)00344-8. PMID 11377168.
- ^ Baba M (2004). "Inhibitors of HIV-1 gene expression and transcription". Current Topics in Medicinal Chemistry. 4 (9): 871–82. doi: 10.2174/1568026043388466. PMID 15134546.
- ^ DeJong ES, Chang CE, Gilson MK, Marino JP (July 2003). "Proflavine acts as a Rev inhibitor by targeting the high-affinity Rev binding site of the Rev responsive element of HIV-1". Biochemistry. 42 (26): 8035–46. doi: 10.1021/bi034252z. PMID 12834355.
- ^ Xiao G, Kumar A, Li K, Rigl CT, Bajic M, Davis TM, et al. (May 2001). "Inhibition of the HIV-1 rev-RRE complex formation by unfused aromatic cations". Bioorganic & Medicinal Chemistry. 9 (5): 1097–113. doi: 10.1016/s0968-0896(00)00344-8. PMID 11377168.
- ^ Plavec I, Agarwal M, Ho KE, Pineda M, Auten J, Baker J, et al. (February 1997). "High transdominant RevM10 protein levels are required to inhibit HIV-1 replication in cell lines and primary T cells: implication for gene therapy of AIDS". Gene Therapy. 4 (2): 128–39. doi: 10.1038/sj.gt.3300369. PMID 9081703.