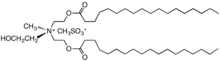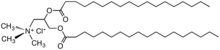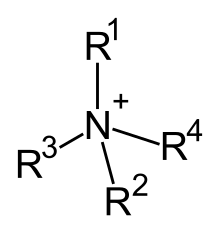
In organic chemistry, quaternary ammonium cations, also known as quats, are positively-charged polyatomic ions of the structure [NR4+, where R is an alkyl group, an aryl group [1] or organyl group. Unlike the ammonium ion (NH+4) and the primary, secondary, or tertiary ammonium cations, the quaternary ammonium cations are permanently charged, independent of the pH of their solution. Quaternary ammonium salts or quaternary ammonium compounds (called quaternary amines in oilfield parlance) are salts of quaternary ammonium cations. Polyquats are a variety of engineered polymer forms which provide multiple quat molecules within a larger molecule.
Quats are used in consumer applications including as antimicrobials (such as detergents and disinfectants), fabric softeners, and hair conditioners. As an antimicrobial, they are able to inactivate enveloped viruses (such as SARS-CoV-2). Quats tend to be gentler on surfaces than bleach-based disinfectants, and are generally fabric-safe. [2]


Synthesis
Quaternary ammonium compounds are prepared by the alkylation of tertiary amine. Industrial production of commodity quat salts usually involves hydrogenation of fatty nitriles, which can generate primary or secondary amines. These amines are then treated with methyl chloride. [4]
The quaternization of alkyl amines by alkyl halides is widely documented. [5] In older literature this is often called a Menshutkin reaction, however modern chemists usually refer to it simply as quaternization. [6] The reaction can be used to produce a compound with unequal alkyl chain lengths; for example when making cationic surfactants one of the alkyl groups on the amine is typically longer than the others. [7] A typical synthesis is for benzalkonium chloride from a long-chain alkyldimethylamine and benzyl chloride:
Reactions
Quaternary ammonium cations are unreactive toward even strong electrophiles, oxidants, and acids. They also are stable toward most nucleophiles. The latter is indicated by the stability of the hydroxide salts such as tetramethylammonium hydroxide and tetrabutylammonium hydroxide even at elevated temperatures. The halflife of Me4NOH in 6M NaOH at 160 °C is >61 h. [8]
Because of their resilience, many unusual anions have been isolated as the quaternary ammonium salts. Examples include
tetramethylammonium pentafluoroxenate, containing the highly reactive pentafluoroxenate (XeF−
5) ion.
Permanganate can be solubilized in
organic
solvents, when deployed as its N
Bu+
4 salt.
[9]
[10]
With exceptionally strong bases, quat cations degrade. They undergo Sommelet–Hauser rearrangement [11] and Stevens rearrangement, [12] as well as dealkylation under harsh conditions or in presence of strong nucleophiles, like thiolates. Quaternary ammonium cations containing N−C−C−H units can also undergo the Hofmann elimination and Emde degradation.

Examples
- Tetramethylammonium ion: (CH3)4N+, also denoted Me4N+ (Me = methyl group)
- Tetraethylammonium ion: (C2H5)4N+, also denoted Et4N+ (Et = ethyl group)
- Tetrapropylammonium ion: (n-C3H7)4N+, also denoted Pr4N+ (Pr = propyl group)
- Tetrabutylammonium ion: (n-C4H9)4N+, also denoted Bu4N+ (Bu = butyl group)
Applications
Quaternary ammonium salts are used as disinfectants, surfactants, fabric softeners, and as antistatic agents (e.g. in shampoos). In liquid fabric softeners, the chloride salts are often used. In dryer anticling strips, the sulfate salts are often used. Older aluminium electrolytic capacitors and spermicidal jellies also contain quaternary ammonium salts. Quats are also used in contraception formulations, veterinary products, diagnostic testing, vaccine production, and nasal formulations. [14]
Concerns have been raised about the level of understanding of safety profile of quat disinfectants on people. As of August 2020, half of disinfectants the United States Environmental Protection Agency suggested as effective against COVID-19 contained one of the quats, and often a quat as the sole ingredient. [15] Salmonella and E. coli O157:H7 exposed to quats have developed cross resistance to antibiotics. A subject of concern is the potential effect of increased use of quats related to COVID-19 pandemic on antibiotic resistance in a larger microbial community in nature and engineered environment. [16]
Medicines

Quaternary ammonium compounds have antimicrobial activity. [18] Quaternary ammonium compounds, especially those containing long alkyl chains, are used as antimicrobials and disinfectants. Examples are benzalkonium chloride, benzethonium chloride, methylbenzethonium chloride, cetalkonium chloride, cetylpyridinium chloride, cetrimonium, cetrimide, dofanium chloride, tetraethylammonium bromide, didecyldimethylammonium chloride and domiphen bromide. Also good against fungi, amoebas, and enveloped viruses (such as SARS-CoV-2), [19] [20] most quaternary ammonium compounds are believed to act by disrupting the cell membrane or viral envelope. [21] (Some QACs, such as dequalinium and similar bis-QACs, show evidence of a different mode of action.) [22]
Quaternary ammonium compounds are lethal to a wide variety of organisms except endospores and non-enveloped viruses, both having no accessible membrane coat to attack. It is possible to solve the endospore problem by adding chemicals which force them to germinate. [23] [24] They have reduced efficacy against gram-negative bacteria, mycobacteria, and bacteria in biofilms due to them having additional layers that need to be penetrated or disrupted. Some bacteria such as MRSA have acquired resistance genes, qacA/B and qacC/D, that pump the cation out of the cell. [22]
Phase transfer catalysts
In organic chemistry, quaternary ammonium salts are employed as phase transfer catalysts (PTCs). Such catalysts accelerate reactions between reagents dissolved in immiscible solvents. The highly reactive reagent dichlorocarbene is generated via PTC by reaction of chloroform and aqueous sodium hydroxide.[ citation needed]

Fabric softeners and hair conditioners
In the 1950s, distearyldimethylammonium chloride (DHTDMAC), was introduced as a fabric softener. This compound was discontinued because the cation biodegrades too slowly. Contemporary fabric softeners are based on salts of quaternary ammonium cations where the fatty acid is linked to the quaternary center via ester linkages; these are commonly referred to as betaine-esters or ester-quats and are susceptible to degradation, e.g., by hydrolysis. [26] Characteristically, the cations contain one or two long alkyl chains derived from fatty acids linked to an ethoxylated ammonium salt. [27] Other cationic compounds can be derived from imidazolium, guanidinium, substituted amine salts, or quaternary alkoxy ammonium salts. [28]
- Cationic surfactants used as fabric softeners
-
Distearyldimethylammonium chloride, an early generation fabric softener with low biodegradability that was phased out.
-
Another diesterquat, a contemporary fabric softener.
-
Diethyl ester dimethyl ammonium chloride used as a fabric softener.
-
Another diesterquat used as a fabric softener.
The antistatic qualities that make quaternary ammonium salts useful as fabric softeners also make them useful in hair conditioners and shampoos. [29] The idea was pioneered by Henkel with a 1984 patent. [30] Examples include cetrimonium chloride and behentrimonium chloride. [31]
Plant growth retardants
Cycocel (chlormequat chloride) reduces plant height by inhibiting the production of gibberellins, the primary plant hormones responsible for cell elongation. Therefore, their effects are primarily on stem, petiole, and flower stalk tissues. Lesser effects are seen in reductions of leaf expansion, resulting in thicker leaves with darker green color. [32]
Natural occurrence
Several quaternary ammonium derivatives exist in nature. [33] Prominent examples include glycine betaine, choline, carnitine. butyrobetaine, homarine, and trigonelline. Glycine betaine, an osmolyte, stabilizes osmotic pressure in cells. [34]
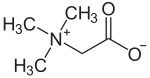
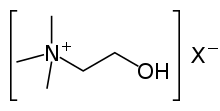
Choline is a precursor for the neurotransmitter acetylcholine. Choline is also a constituent of lecithin, which is present in many plants and animal organs. [35] It is found in phospholipids. For example, phosphatidylcholines, a major component of biological membranes, are a member of the lecithin group of fatty substances in animal and plant tissues. [36]
Carnitine participates in the beta-oxidation of fatty acids.[ citation needed]

Health effects
Quaternary ammonium compounds can display a range of health effects, amongst which are mild skin and respiratory irritation [37] up to severe caustic burns on skin and the gastrointestinal wall (depending on concentration), gastrointestinal symptoms (e.g., nausea and vomiting), coma, convulsions, hypotension and death. [38]
They are thought to be the chemical group responsible for anaphylactic reactions that occur with use of neuromuscular blocking drugs during general anaesthesia in surgery. [39] Quaternium-15 is the single most often found cause of allergic contact dermatitis of the hands (16.5% in 959 cases). [40]
Possible reproductive effects in laboratory animals
Quaternary ammonium-based disinfectants (Virex and Quatricide) were tentatively identified as the most probable cause of jumps in birth defects and fertility problems in caged lab mice. The quat ingredients in the disinfectants include alkyl dimethyl benzyl ammonium chloride (ADBAC) and didecyl dimethyl ammonium chloride (DDAC). [41] [42] A similar link was tentatively identified in nurses. [43] The studies contradict earlier toxicology data reviewed by the U.S. Environmental Protection Agency (U.S. EPA) and the EU Commission. [44]
Quantification
The quantification of quaternary ammonium compounds can be challenging. Some methods include precipitation of solid salts with tetraphenylborate. Another method, an Epton titration, involves partitioning between water- chloroform in the presence of an anionic dye. Individual cations are detectable by ESI-MS and NMR spectroscopy. [4]
See also
References
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) " quaternary ammonium compounds". doi: 10.1351/goldbook.Q05003
- ^ Heffernan T (14 April 2021). "The Best Surface Cleaners, Wipes, and Disinfectants". The New York Times. ISSN 0362-4331. Retrieved 2022-02-17.
- ^ a b Kern A, Näther C, Studt F, Tuczek F (August 2004). "Application of a universal force field to mixed Fe/Mo-S/Se cubane and heterocubane clusters. 1. Substitution of sulfur by selenium in the series [Fe4X4(YCH3)4]2-; X = S/Se and Y = S/Se". Inorganic Chemistry. 43 (16): 5003–5010. doi: 10.1021/ic030347d. PMID 15285677.
- ^ a b Weston, Charles W.; Papcun, John R.; Dery, Maurice (2003). "Ammonium Compounds". Kirk-Othmer Encyclopedia of Chemical Technology. doi: 10.1002/0471238961.0113131523051920.a01.pub2. ISBN 0471238961.
- ^ Brasen WR, Hauser CR (1954). "o-Methylbenzyl Alcohol". Organic Syntheses. 34: 58. doi: 10.15227/orgsyn.034.0058.
- ^ Smith MB, March J (2001). Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (5th ed.). New York, NY: Wiley-Interscience. ISBN 0-471-58589-0.
- ^ Kosswig K. "Surfactants". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.a25_747. ISBN 978-3527306732.
- ^ Marino MG, Kreuer KD (February 2015). "Alkaline stability of quaternary ammonium cations for alkaline fuel cell membranes and ionic liquids". ChemSusChem. 8 (3): 513–523. doi: 10.1002/cssc.201403022. PMID 25431246.
- ^ Herriott AW (1977). "Purple benzene: Solubilization of anions in organic solvents". Journal of Chemical Education. 54 (4): 229. Bibcode: 1977JChEd..54Q.229H. doi: 10.1021/ed054p229.1.
- ^ Doheny Jr AJ, Ganem B (1980). "Purple benzene revisited". Journal of Chemical Education. 57 (4): 308. Bibcode: 1980JChEd..57..308D. doi: 10.1021/ed057p308.1.
- ^ Brasen WR, Hauser CR (1963). "2-Methylbenzyldimethylamine". Organic Syntheses; Collected Volumes, vol. 4, p. 585.
- ^ Pine SH (2011). "The Base-Promoted Rearrangements of Quaternary Ammonium Salts". Organic Reactions. pp. 403–464. doi: 10.1002/0471264180.or018.04. ISBN 978-0471264187.
- ^ Lackner M, Guggenbichler JP (2013). "Antimicrobial Surfaces". Ullmann's Encyclopedia of Industrial Chemistry. pp. 1–13. doi: 10.1002/14356007.q03_q01. ISBN 9783527306732.
- ^ "Quats applications". 22 October 2021.
- ^ Lim XZ (August 2, 2020). "Do we know enough about the safety of quat disinfectants?". Chemical and Engineering News. Retrieved 2023-01-08.
- ^ Hora PI, Pati SG, McNamara PJ, Arnold WA (2020-09-08). "Increased Use of Quaternary Ammonium Compounds during the SARS-CoV-2 Pandemic and Beyond: Consideration of Environmental Implications". Environmental Science & Technology Letters. 7 (9): 622–631. doi: 10.1021/acs.estlett.0c00437. ISSN 2328-8930. PMC 7341688.
- ^ Bungardt E, Mutschler E (2000). "Spasmolytics". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.a24_515. ISBN 978-3527306732.
- ^ Jia Z, shen D, Xu W (June 2001). "Synthesis and antibacterial activities of quaternary ammonium salt of chitosan". Carbohydrate Research. 333 (1): 1–6. doi: 10.1016/S0008-6215(01)00112-4. PMID 11423105.
- ^ Abedon ST. "Specific Antimicrobials". Ohio State University. Archived from the original on 2008-10-15. Retrieved 2020-02-14.
- ^ Schrank CL, Minbiole KP, Wuest WM (July 2020). "Are Quaternary Ammonium Compounds, the Workhorse Disinfectants, Effective against Severe Acute Respiratory Syndrome-Coronavirus-2?". ACS Infectious Diseases. 6 (7): 1553–1557. doi: 10.1021/acs.estlett.0c00437. PMC 7341688. PMID 32412231.
- ^ Cocco AR, Rosa WL, Silva AF, Lund RG, Piva E (November 2015). "A systematic review about antibacterial monomers used in dental adhesive systems: Current status and further prospects". Dental Materials. 31 (11): 1345–1362. doi: 10.1016/j.dental.2015.08.155. PMID 26345999.
- ^ a b Tischer, Maximilian; Pradel, Gabriele; Ohlsen, Knut; Holzgrabe, Ulrike (2 January 2012). "Quaternary Ammonium Salts and Their Antimicrobial Potential: Targets or Nonspecific Interactions?". ChemMedChem. 7 (1): 22–31. doi: 10.1002/cmdc.201100404. PMID 22113995. S2CID 26326417.
- ^ Nerandzic, MM; Donskey, CJ (October 2016). "A Quaternary Ammonium Disinfectant Containing Germinants Reduces Clostridium difficile Spores on Surfaces by Inducing Susceptibility to Environmental Stressors". Open Forum Infectious Diseases. 3 (4): ofw196. doi: 10.1093/ofid/ofw196. PMC 5198585. PMID 28066792.
- ^ Tuladhar, E; de Koning, MC; Fundeanu, I; Beumer, R; Duizer, E (April 2012). "Different virucidal activities of hyperbranched quaternary ammonium coatings on poliovirus and influenza virus". Applied and Environmental Microbiology. 78 (7): 2456–8. Bibcode: 2012ApEnM..78.2456T. doi: 10.1128/AEM.07738-11. PMC 3302582. PMID 22287007.
- ^ De Dardel, François; Arden, Thomas V. (2008). "Ion Exchangers". Ullmann's Encyclopedia of Industrial Chemistry. p. 5. doi: 10.1002/14356007.a14_393.pub2. ISBN 978-3527306732.
- ^ Hellberg PE, Bergström K, Holmberg K (January 2000). "Cleavable surfactants". Journal of Surfactants and Detergents. 3 (1): 81–91. doi: 10.1007/s11743-000-0118-z. S2CID 195343430.
- ^ "Henkel Consumer Info". Henkelconsumerinfo.com. Archived from the original on 2019-10-18. Retrieved 2009-06-04.
- ^ Smulders E, Sung E. "Laundry Detergents, 2. Ingredients and Products". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.o15_013. ISBN 978-3527306732.
- ^ Distearyldimonium chloride in the Consumer Product Information Database
- ^ "Quaternary ammonium compound hair conditioners US4744977". Google Patents. 14 November 1985.
- ^ "What is cetrimonium chloride?". naturallycurly.com. November 2007. Archived from the original on 2012-03-04.
- ^ "Growth Retardants". Archived from the original on September 25, 2011. Retrieved July 19, 2012. Wageningen Agricultural University, The Netherlands
- ^ Anthoni, U.; Christophersen, C.; Hougaard, L.; Nielsen, P.H. (1991). "Quaternary ammonium compounds in the biosphere—An Example of a Versatile Adaptive Strategy". Comparative Biochemistry and Physiology Part B: Comparative Biochemistry. 99: 1–18. doi: 10.1016/0305-0491(91)90002-U.
- ^ Slama, Inès; Abdelly, Chedly; Bouchereau, Alain; Flowers, Tim; Savouré, Arnould (2015). "Diversity, distribution and roles of osmoprotective compounds accumulated in halophytes under abiotic stress". Annals of Botany. 115 (3): 433–447. doi: 10.1093/aob/mcu239. PMC 4332610. PMID 25564467.
- ^ Zeisel SH, da Costa KA (November 2009). "Choline: an essential nutrient for public health". Nutrition Reviews. 67 (11): 615–623. doi: 10.1111/j.1753-4887.2009.00246.x. PMC 2782876. PMID 19906248.
- ^ Jackowski S, Cronan Jr JE, Rock CO (1991). "Chapter 2: Lipid metabolism in procaryotes". In Vance DE, Vance J (eds.). Biochemistry of Lipids, Lipoproteins and Membranes. Elsevier. pp. 80–81. ISBN 0-444-89321-0.
- ^ Bello A, Quinn MM, Perry MJ, Milton DK (March 2009). "Characterization of occupational exposures to cleaning products used for common cleaning tasks--a pilot study of hospital cleaners". Environmental Health. 8: 11. doi: 10.1186/1476-069X-8-11. PMC 2678109. PMID 19327131.
- ^ "Quaternary ammonium". Internationally Peer Reviewed Chemical Safety Information (INCHEM). World Health Organization. PIM G022.
- ^ Harper NJ, Dixon T, Dugué P, Edgar DM, Fay A, Gooi HC, et al. (February 2009). "Suspected anaphylactic reactions associated with anaesthesia". Anaesthesia. 64 (2): 199–211. doi: 10.1111/j.1365-2044.2008.05733.x. PMC 3082210. PMID 19143700.
- ^ Warshaw EM, Ahmed RL, Belsito DV, DeLeo VA, Fowler JF, Maibach HI, et al. (August 2007). "Contact dermatitis of the hands: cross-sectional analyses of North American Contact Dermatitis Group Data, 1994-2004". Journal of the American Academy of Dermatology. 57 (2): 301–314. doi: 10.1016/j.jaad.2007.04.016. PMID 17553593.
- ^ Hunt P (June 2008). "Lab disinfectant harms mouse fertility. Patricia Hunt interviewed by Brendan Maher". Nature. 453 (7198): 964. doi: 10.1038/453964a. PMID 18563110.
- ^ Melin VE, Potineni H, Hunt P, Griswold J, Siems B, Werre SR, Hrubec TC (December 2014). "Exposure to common quaternary ammonium disinfectants decreases fertility in mice". Reproductive Toxicology. 50: 163–170. doi: 10.1016/j.reprotox.2014.07.071. PMC 4260154. PMID 25483128.
- ^ Gaskins AJ, Chavarro JE, Rich-Edwards JW, Missmer SA, Laden F, Henn SA, Lawson CC (March 2017). "Occupational use of high-level disinfectants and fecundity among nurses". Scandinavian Journal of Work, Environment & Health. 43 (2): 171–180. doi: 10.5271/sjweh.3623. PMC 5840865. PMID 28125764.
- ^ Office of Prevention, Pesticides and Toxic Substances, US EPA (August 2006). "Reregistration Eligibility Decision for Aliphatic Alkyl Quaternaries (DDAC)". Epa739-R-06-008.
Further reading
- Zhang C, Cui F, Zeng GM, Jiang M, Yang ZZ, Yu ZG, et al. (June 2015). "Quaternary ammonium compounds (QACs): a review on occurrence, fate and toxicity in the environment". The Science of the Total Environment. 518–519: 352–362. Bibcode: 2015ScTEn.518..352Z. doi: 10.1016/j.scitotenv.2015.03.007. PMID 25770948.
External links
- "Toxicities of quaternary ammonium". Internationally Peer Reviewed Chemical Safety Information (INCHEM). World Health Organization.
![{\displaystyle {\ce {CH3(CH2)_{\mathit {n}}N(CH3)2{}+ClCH2C6H5->{}[CH3(CH2)_{\mathit {n}}N(CH3)2CH2C6H5]+Cl-}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/75c6828bdcf8dc9add4d8323e2b870fe1dd7d73d)