| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
| ECHA InfoCard | 100.034.740 |
| EC Number |
|
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| O4PPr | |
| Molar mass | 235.877 g·mol−1 |
| Appearance | solid |
| Hazards | |
| GHS labelling: [1] | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P264+P265, P271, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
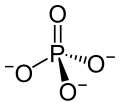
Praseodymium(III) phosphate is an inorganic compound with the chemical formula PrPO4.
Preparation
Praseodymium(III) phosphate hemihydrate can be obtained by reacting praseodymium chloride and phosphoric acid: [2]
- PrCl3 + H3PO4 → PrPO4 + 3 HCl
It can also be produced by reacting silicon pyrophosphate (SiP2O7) and praseodymium(III,IV) oxide (Pr6O11) at 1200 °C. [3]
Properties
Praseodymium(III) phosphate forms light green crystals in the monoclinic crystal system, with space group P21/n and cell parameters a = 0.676 nm, b = 0.695 nm, c = 0.641 nm, β = 103.25°, Z = 4. [4] [5]
It forms a crystal hydrate of the composition PrPO4·nH2O, where n < 0.5, with light green crystals of hexagonal crystal system, space group P6222, and cell parameters a = 0.700 nm, c = 0.643 nm, Z = 3. [6] [7]
Praseodymium(III) phosphate reacts with sodium fluoride to obtain Na2PrF2(PO4). [8]
- PrPO4 + 2 NaF → Na2PrF2(PO4)
References
- ^ "Praseodymium(III) phosphate". pubchem.ncbi.nlm.nih.gov.
- ^ Hikichi, Yasuo; Hukuo, Ken-iti; Shiokawa, Jiro (Dec 1978). "Syntheses of Rare Earth Orthophosphates". Bulletin of the Chemical Society of Japan. 51 (12): 3645–3646. doi: 10.1246/bcsj.51.3645. ISSN 0009-2673.
-
^ Carlos E. Bamberger, George M. Begun, Dale E. Heatherly (Nov 1983).
"Synthesis of Metal Phosphates Using SiP2O7". Journal of the American Ceramic Society. 66 (11): c208–c209.
doi:
10.1111/j.1151-2916.1983.tb10575.x.
ISSN
0002-7820. Retrieved 2022-03-06.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ Garkavi, Andrey Vladimirovich (2023), "How to Draw Up and Complete a Dissertation, 2nd ed., revised and enlarged", How to Draw up and Complete a Dissertation, 2nd ed., revised and enlarged, OOO "GEOTAR-Media" Publishing Group, pp. 1–80, doi: 10.33029/9704-7930-8-hdc-2023-1-80, ISBN 978-5-9704-7930-8, retrieved 2023-12-06
- ^ Horchani-Naifer, K.; Férid, M. (2009-04-20). "Crystal structure, energy band and optical characterizations of praseodymium monophosphate PrPO4". Inorganica Chimica Acta. 362 (6): 1793–1796. doi: 10.1016/j.ica.2008.08.021. ISSN 0020-1693.
- ^ Инязовские чтения. Сборник научных статей Первой международной научно-практической конференции Совета молодых ученых МГЛУ (in Russian). Москва: Московский государственный лингвистический университет. 2022. doi: 10.52070/978-5-00120-359-9_2022. ISBN 978-5-00120-359-9.
- ^ Hezel, A.; Ross, S. D. (1967-08-01). "X-ray powder data and cell dimensions of some rare earth orthophosphates". Journal of Inorganic and Nuclear Chemistry. 29 (8): 2085–2089. doi: 10.1016/0022-1902(67)80469-X. ISSN 0022-1902.
- ^ ZIMINA, G. V.; SMIRNOVA, I. N.; GORKOVENKO, M. YU.; SPIRIDONOV, F. M.; KOMISSAROVA, L. N.; KALOEV, N. I. (1995-02-21). "ChemInform Abstract: Synthesis and Studies of Fluorophosphates of Rare Earth Elements Na2LnF2PO4". ChemInform. 26 (8). doi: 10.1002/chin.199508015. ISSN 0931-7597.