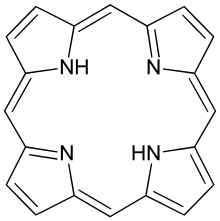
Porphyrins ( /ˈpɔːrfərɪn/ POR-fər-in) are a group of heterocyclic macrocycle organic compounds, composed of four modified pyrrole subunits interconnected at their α carbon atoms via methine bridges (=CH−). In vertebrates, an essential member of the porphyrin group is heme, which is a component of hemoproteins, whose functions include carrying oxygen in the bloodstream. In plants, an essential porphyrin derivative is chlorophyll, which is involved in light harvesting and electron transfer in photosynthesis.
The parent of porphyrins is porphine, a rare chemical compound of exclusively theoretical interest. Substituted porphines are called porphyrins. [1] With a total of 26 π-electrons, of which 18 π-electrons form a planar, continuous cycle, the porphyrin ring structure is often described as aromatic. [2] [3] One result of the large conjugated system is that porphyrins typically absorb strongly in the visible region of the electromagnetic spectrum, i.e. they are deeply colored. The name "porphyrin" derives from the Greek word πορφύρα (porphyra), meaning purple. [4]
Structure
Porphyrin complexes consist of a square planar MN4 core. The periphery of the porphyrins, consisting of sp2-hybridized carbons, generally display small deviations from planarity. "Ruffled" or saddle-shaped porphyrins is attributed to interactions of the system with its environment. [5] Additionally, the metal is often not centered in the N4 plane. [6] For free porphyrins, the two pyrrole protons are mutually trans and project out of the N4 plane. [7] These nonplanar distortions are associated with altered chemical and physical properties. Chlorophyll-rings are more distinctly nonplanar, but they are more saturated than porphyrins. [8]
Complexes of porphyrins
Concomitant with the displacement of two N-H protons, porphyrins bind metal ions in the N4 "pocket". The metal ion usually has a charge of 2+ or 3+. A schematic equation for these syntheses is shown:
- H2porphyrin + [MLn2+ → M(porphyrinate)Ln−4 + 4 L + 2 H+, where M = metal ion and L = a ligand
- Representative porphyrins and derivatives
-
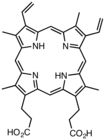
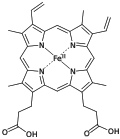
Derivatives of protoporphyrin IX are common in nature, the precursor to hemes.
-
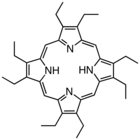
Octaethylporphyrin (H2OEP) is a synthetic analogue of protoporphyrin IX. Unlike the natural porphyrin ligands, OEP2− is highly symmetrical.
-
Tetraphenylporphyrin (H2TPP)is another synthetic analogue of protoporphyrin IX. Unlike the natural porphyrin ligands, TPP2− is highly symmetrical. Another difference is that its methyne centers are occupied by phenyl groups.
-
Simplified view of heme, a complex of a protoporphyrin IX.
-
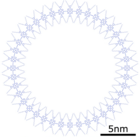
A macrocycle of 40 porphyrin molecules, model
-
A macrocycle of 40 porphyrin molecules, STM image
Ancient porphyrins
A geoporphyrin, also known as a petroporphyrin, is a porphyrin of geologic origin. [9] They can occur in crude oil, oil shale, coal, or sedimentary rocks. [9] [10] Abelsonite is possibly the only geoporphyrin mineral, as it is rare for porphyrins to occur in isolation and form crystals. [11]
The field of organic geochemistry had its origins in the isolation of porphyrins from petroleum.[ citation needed] This finding helped establish the biological origins of petroleum. Petroleum is sometimes "fingerprinted" by analysis of trace amounts of nickel and vanadyl porphyrins.[ citation needed]
Biosynthesis
In non-photosynthetic eukaryotes such as animals, insects, fungi, and protozoa, as well as the α-proteobacteria group of bacteria, the committed step for porphyrin biosynthesis is the formation of δ-aminolevulinic acid (δ-ALA, 5-ALA or dALA) by the reaction of the amino acid glycine with succinyl-CoA from the citric acid cycle. In plants, algae, bacteria (except for the α-proteobacteria group) and archaea, it is produced from glutamic acid via glutamyl-tRNA and glutamate-1-semialdehyde. The enzymes involved in this pathway are glutamyl-tRNA synthetase, glutamyl-tRNA reductase, and glutamate-1-semialdehyde 2,1-aminomutase. This pathway is known as the C5 or Beale pathway.
Two molecules of dALA are then combined by porphobilinogen synthase to give porphobilinogen (PBG), which contains a pyrrole ring. Four PBGs are then combined through deamination into hydroxymethyl bilane (HMB), which is hydrolysed to form the circular tetrapyrrole uroporphyrinogen III. This molecule undergoes a number of further modifications. Intermediates are used in different species to form particular substances, but, in humans, the main end-product protoporphyrin IX is combined with iron to form heme. Bile pigments are the breakdown products of heme.
The following scheme summarizes the biosynthesis of porphyrins, with references by EC number and the OMIM database. The porphyria associated with the deficiency of each enzyme is also shown:

Laboratory synthesis

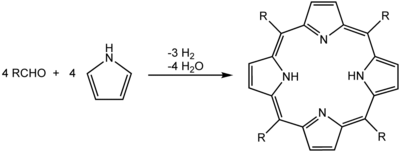
A common synthesis for porphyrins is the Rothemund reaction, first reported in 1936, [12] [13] which is also the basis for more recent methods described by Adler and Longo. [14] The general scheme is a condensation and oxidation process starting with pyrrole and an aldehyde.
Potential applications
Photodynamic therapy
Porphyrins have been evaluated in the context of photodynamic therapy (PDT) since they strongly absorb light, which is then converted to heat in the illuminated areas. [15] This technique has been applied in macular degeneration using verteporfin. [16]
PDT is considered a noninvasive cancer treatment, involving the interaction between light of a determined frequency, a photo-sensitizer, and oxygen. This interaction produces the formation of a highly reactive oxygen species (ROS), usually singlet oxygen, as well as superoxide anion, free hydroxyl radical, or hydrogen peroxide. [17] These high reactive oxygen species react with susceptible cellular organic biomolecules such as; lipids, aromatic amino acids, and nucleic acid heterocyclic bases, to produce oxidative radicals that damage the cell, possibly inducing apoptosis or even necrosis. [18]
Molecular electronics and sensors
Porphyrin-based compounds are of interest as possible components of molecular electronics and photonics. [19] Synthetic porphyrin dyes have been incorporated in prototype dye-sensitized solar cells. [20] [21]
Biological applications
Porphyrins have been investigated as possible anti-inflammatory agents [22] and evaluated on their anti-cancer and anti-oxidant activity. [23] Several porphyrin-peptide conjugates were found to have antiviral activity against HIV in vitro. [24]
Toxicology
Heme biosynthesis is used as biomarker in environmental toxicology studies. While excess production of porphyrins indicate organochlorine exposure, lead inhibits ALA dehydratase enzyme. [25]
Gallery
-
Lewis structure for meso-tetraphenylporphyrin
-
UV–vis readout for meso-tetraphenylporphyrin
-
Light-activated porphyrin. Monatomic oxygen. Cellular aging
Related species
In nature
Several heterocycles related to porphyrins are found in nature, almost always bound to metal ions. These include
| N4-macrocycle | Cofactor name | metal | comment |
|---|---|---|---|
| chlorin | chlorophyll | magnesium | several versions of chlorophyll exist (sidechain; exception being chlorophyll c) |
| bacteriochlorin | bacteriochlorophyll (in part) | magnesium | several versions of bacteriochlorophyll exist (sidechain; some use a usual chlorin ring) |
| sirohydrochlorin (an isobacteriochlorin) | siroheme | iron | Important cofactor in sulfur assimilation |
| biosynthetic intermediate en route to cofactor F430 and B12 | |||
| corrin | vitamin B12 | cobalt | several variants of B12 exist (sidechain) |
| corphin | Cofactor F430 | nickel | highly reduced macrocycle |
Synthetic
A benzoporphyrin is a porphyrin with a benzene ring fused to one of the pyrrole units. e.g. verteporfin is a benzoporphyrin derivative. [26]
Non-natural porphyrin isomers

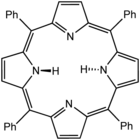
The first synthetic porphyrin isomer was reported by Emanual Vogel and coworkers in 1986. [27] This isomer [18]porphyrin-(2.0.2.0) is named as porphycene, and the central N4 Cavity forms a rectangle shape as shown in figure. [28] Porphycenes showed interesting photophysical behavior and found versatile compound towards the photodynamic therapy. [29] This inspired Vogel and Sessler to took up the challenge of preparing [18]porphyrin-(2.1.0.1) and named it as corrphycene or porphycerin. [30] The third porphyrin that is [18]porphyrin-(2.1.1.0), was reported by Callot and Vogel-Sessler. Vogel and coworkers reported successful isolation of [18]porphyrin-(3.0.1.0) or isoporphycene. [31] The Japanese scientist Furuta [32] and Polish scientist Latos-Grażyński [33] almost simultaneously reported the N-confused porphyrins. The inversion of one of the pyrrolic subunits in the macrocyclic ring resulted in one of the nitrogen atoms facing outwards from the core of the macrocycle.

See also
- A porphyrin-related disease: porphyria
- Porphyrin coordinated to iron: heme
- A heme-containing group of enzymes: Cytochrome P450
- Porphyrin coordinated to magnesium: chlorophyll
- The one-carbon-shorter analogues: corroles, including vitamin B12, which is coordinated to a cobalt
- Corphins, the highly reduced porphyrin coordinated to nickel that binds the Cofactor F430 active site in methyl coenzyme M reductase (MCR)
- Nitrogen-substituted porphyrins: phthalocyanine
References
- ^ Rayati S, Malekmohammadi S (2016). "Catalytic activity of multi-wall carbon nanotube supported manganese (III) porphyrin: an efficient, selective and reusable catalyst for oxidation of alkenes and alkanes with urea–hydrogen peroxide". Journal of Experimental Nanoscience. 11 (11): 872. Bibcode: 2016JENan..11..872R. doi: 10.1080/17458080.2016.1179802.
- ^ Ivanov AS, Boldyrev AI (August 2014). "Deciphering aromaticity in porphyrinoids via adaptive natural density partitioning". Organic & Biomolecular Chemistry. 12 (32): 6145–6150. doi: 10.1039/C4OB01018C. PMID 25002069.
- ^ Lash TD (2011). "Origin of aromatic character in porphyrinoid systems". Journal of Porphyrins and Phthalocyanines. 15 (11n12): 1093–1115. doi: 10.1142/S1088424611004063.
- ^ Harper D, Buglione DC. "porphyria (n.)". The Online Etymology Dictionary. Retrieved 14 September 2014.
- ^ Senge MO, MacGowan SA, O'Brien JM (December 2015). "Conformational control of cofactors in nature - the influence of protein-induced macrocycle distortion on the biological function of tetrapyrroles". Chemical Communications. 51 (96): 17031–17063. doi: 10.1039/C5CC06254C. hdl: 2262/75305. PMID 26482230.
- ^ Walker FA, Simonis U (2011). "Iron Porphyrin Chemistry". Encyclopedia of Inorganic and Bioinorganic Chemistry. doi: 10.1002/9781119951438.eibc0104. ISBN 9781119951438.
- ^ Jentzen W, Ma JG, Shelnutt JA (February 1998). "Conservation of the conformation of the porphyrin macrocycle in hemoproteins". Biophysical Journal. 74 (2 Pt 1): 753–763. Bibcode: 1998BpJ....74..753J. doi: 10.1016/S0006-3495(98)74000-7. PMC 1302556. PMID 9533688.
- ^ Senge MO, Ryan AA, Letchford KA, MacGowan SA, Mielke T (2014). "Chlorophylls, Symmetry, Chirality, and Photosynthesis". Symmetry. 6 (3): 781–843. Bibcode: 2014Symm....6..781S. doi: 10.3390/sym6030781. hdl: 2262/73843.
- ^ a b Kadish KM, ed. (1999). The Porphyrin Handbook. Elsevier. p. 381. ISBN 9780123932006.
- ^ Zhang B, Lash TD (September 2003). "Total synthesis of the porphyrin mineral abelsonite and related petroporphyrins with five-membered exocyclic rings". Tetrahedron Letters. 44 (39): 7253. doi: 10.1016/j.tetlet.2003.08.007.
- ^ Mason GM, Trudell LG, Branthaver JF (1989). "Review of the stratigraphic distribution and diagenetic history of abelsonite". Organic Geochemistry. 14 (6): 585. Bibcode: 1989OrGeo..14..585M. doi: 10.1016/0146-6380(89)90038-7.
- ^ Rothemund P (1936). "A New Porphyrin Synthesis. The Synthesis of Porphin". J. Am. Chem. Soc. 58 (4): 625–627. doi: 10.1021/ja01295a027.
- ^ Rothemund P (1935). "Formation of Porphyrins from Pyrrole and Aldehydes". J. Am. Chem. Soc. 57 (10): 2010–2011. doi: 10.1021/ja01313a510.
- ^ Adler AD, Longo FR, Finarelli JD, Goldmacher J, Assour J, Korsakoff L (1967). "A simplified synthesis for meso-tetraphenylporphine". J. Org. Chem. 32 (2): 476. doi: 10.1021/jo01288a053.
- ^ Giuntini F, Boyle R, Sibrian-Vazquez M, Vicente MG (2014). "Porphyrin conjugates for cancer therapy". In Kadish KM, Smith KM, Guilard R (eds.). Handbook of Porphyrin Science. Vol. 27. pp. 303–416.
- ^ Wormald R, Evans J, Smeeth L, Henshaw K (July 2007). "Photodynamic therapy for neovascular age-related macular degeneration" (PDF). The Cochrane Database of Systematic Reviews (3): CD002030. doi: 10.1002/14651858.CD002030.pub3. PMID 17636693.
- ^ Price M, Terlecky SR, Kessel D (2009). "A role for hydrogen peroxide in the pro-apoptotic effects of photodynamic therapy". Photochemistry and Photobiology. 85 (6): 1491–1496. doi: 10.1111/j.1751-1097.2009.00589.x. PMC 2783742. PMID 19659920.
- ^ Singh S, Aggarwal A, Bhupathiraju NV, Arianna G, Tiwari K, Drain CM (September 2015). "Glycosylated Porphyrins, Phthalocyanines, and Other Porphyrinoids for Diagnostics and Therapeutics". Chemical Reviews. 115 (18): 10261–10306. doi: 10.1021/acs.chemrev.5b00244. PMC 6011754. PMID 26317756.
- ^ Lewtak JP, Gryko DT (October 2012). "Synthesis of π-extended porphyrins via intramolecular oxidative coupling". Chemical Communications. 48 (81): 10069–10086. doi: 10.1039/c2cc31279d. PMID 22649792.
- ^ Walter MG, Rudine AB, Wamser CC (2010). "Porphyrins and phthalocyanines in solar photovoltaic cells". Journal of Porphyrins and Phthalocyanines. 14 (9): 759–792. doi: 10.1142/S1088424610002689.
- ^ Yella A, Lee HW, Tsao HN, Yi C, Chandiran AK, Nazeeruddin MK, et al. (November 2011). "Porphyrin-sensitized solar cells with cobalt (II/III)-based redox electrolyte exceed 12 percent efficiency". Science. 334 (6056): 629–634. Bibcode: 2011Sci...334..629Y. doi: 10.1126/science.1209688. PMID 22053043. S2CID 28058582.
- ^ Alonso-Castro AJ, Zapata-Morales JR, Hernández-Munive A, Campos-Xolalpa N, Pérez-Gutiérrez S, Pérez-González C (May 2015). "Synthesis, antinociceptive and anti-inflammatory effects of porphyrins". Bioorganic & Medicinal Chemistry. 23 (10): 2529–2537. doi: 10.1016/j.bmc.2015.03.043. PMID 25863493.
- ^ Bajju GD, Ahmed A, Devi G (December 2019). "Synthesis and bioactivity of oxovanadium(IV)tetra(4-methoxyphenyl)porphyrinsalicylates". BMC Chemistry. 13 (1): 15. doi: 10.1186/s13065-019-0523-9. PMC 6661832. PMID 31384764.
- ^ Mendonça DA, Bakker M, Cruz-Oliveira C, Neves V, Jiménez MA, Defaus S, et al. (June 2021). "Penetrating the Blood-Brain Barrier with New Peptide-Porphyrin Conjugates Having anti-HIV Activity". Bioconjugate Chemistry. 32 (6): 1067–1077. doi: 10.1021/acs.bioconjchem.1c00123. PMC 8485325. PMID 34033716.
- ^ Walker CH, Silby RM, Hopkin SP, Peakall DB (2012). Principles of Ecotoxicology. Boca Raton, FL: CRC Press. p. 182. ISBN 978-1-4665-0260-4.
- ^ Scott LJ, Goa KL (February 2000). "Verteporfin". Drugs & Aging. 16 (2): 139–146, discussion 146–8. doi: 10.2165/00002512-200016020-00005. PMID 10755329. S2CID 260491296.
- ^ Vogel E, Köcher M (March 1986). "Porphycene—a Novel Porphin Isomer". Angewandte Chemie. 25 (3): 257. doi: 10.1002/anie.198602571.
- ^ Nagamaiah J, Dutta A, Pati NN, Sahoo S, Soman R, Panda PK (March 2022). "3,6,13,16-Tetrapropylporphycene: Rational Synthesis, Complexation, and Halogenation". The Journal of Organic Chemistry. 87 (5): 2721–2729. doi: 10.1021/acs.joc.1c02652. PMID 35061396. S2CID 246165814.
- ^ Dougherty TJ (2001). "Basic principles of photodynamic therapy". Journal of Porphyrins and Phthalocyanines. 5 (2): 105. doi: 10.1002/jpp.328.
- ^ Vogel E, Guilard R (November 1993). "New Porphycene Ligands: Octaethyl‐ and Etioporphycene (OEPc and EtioPc)—Tetra‐ and Pentacoordinated Zinc Complexes of OEPc". Angewandte Chemie International Edition. 32 (11): 1600. doi: 10.1002/anie.199316001.
- ^ Vogel E, Scholz P, Demuth R, Erben C, Bröring M, Schmickler H, et al. (October 1999). "Isoporphycene: The Fourth Constitutional Isomer of Porphyrin with an N(4) Core-Occurrence of E/Z Isomerism". Angewandte Chemie. 38 (19): 2919–2923. doi: 10.1002/(SICI)1521-3773(19991004)38:19<2919::AID-ANIE2919>3.0.CO;2-W. PMID 10540393.
- ^ Hiroyuki F (1994). ""N-Confused Porphyrin": A New Isomer of Tetraphenylporphyrin". J. Am. Chem. Soc. 116 (2): 767. doi: 10.1021/ja00081a047.
- ^ Chmielewski PJ, Latos-Grażyński L, Rachlewicz K, Glowiak T (18 April 1994). "Tetra‐p‐tolylporphyrin with an Inverted Pyrrole Ring: A Novel Isomer of Porphyrin". Angewandte Chemie International Edition. 33 (7): 779. doi: 10.1002/anie.199407791.