Phosphatidylethanolamine (PE) is a class of phospholipids found in biological membranes. [1] They are synthesized by the addition of cytidine diphosphate- ethanolamine to diglycerides, releasing cytidine monophosphate. S-Adenosyl methionine can subsequently methylate the amine of phosphatidylethanolamines to yield phosphatidylcholines.
Function

In cells
Phosphatidylethanolamines are found in all living cells, composing 25% of all phospholipids. In human physiology, they are found particularly in nervous tissue such as the white matter of brain, nerves, neural tissue, and in spinal cord, where they make up 45% of all phospholipids. [2]
Phosphatidylethanolamines play a role in membrane fusion and in disassembly of the contractile ring during cytokinesis in cell division. [3] Additionally, it is thought that phosphatidylethanolamine regulates membrane curvature. Phosphatidylethanolamine is an important precursor, substrate, or donor in several biological pathways. [2]
As a polar head group, phosphatidylethanolamine creates a more viscous lipid membrane compared to phosphatidylcholine. For example, the melting temperature of di-oleoyl-phosphatidylethanolamine is -16 °C while the melting temperature of di-oleoyl-phosphatidylcholine is -20 °C. If the lipids had two palmitoyl chains, phosphatidylethanolamine would melt at 63 °C while phosphatidylcholine would melt already at 41 °C. [4] Lower melting temperatures correspond, in a simplistic view, to more fluid membranes.
In humans
In humans, metabolism of phosphatidylethanolamine is thought to be important in the heart. When blood flow to the heart is restricted, the asymmetrical distribution of phosphatidylethanolamine between membrane leaflets is disrupted, and as a result the membrane is disrupted. Additionally, phosphatidylethanolamine plays a role in the secretion of lipoproteins in the liver. This is because vesicles for secretion of very low-density lipoproteins coming off of the Golgi apparatus have a significantly higher phosphatidylethanolamine concentration when compared to other vesicles containing very low-density lipoproteins. [5] Phosphatidylethanolamine has also shown to be able to propagate infectious prions without the assistance of any proteins or nucleic acids, which is a unique characteristic of it. [6] Phosphatidylethanolamine is also thought to play a role in blood clotting, as it works with phosphatidylserine to increase the rate of thrombin formation by promoting binding to factor V and factor X, two proteins which catalyze the formation of thrombin from prothrombin. [7] The synthesis of endocannabinoid anandamide is performed from the phosphatidylethanolamine by the successive action of 2 enzymes, the N- acetyltransferase and phospholipase-D. [8]
In bacteria
Where phosphatidylcholine is the principal phospholipid in animals, phosphatidylethanolamine is the principal one in bacteria. One of the primary roles for phosphatidylethanolamine in bacterial membranes is to spread out the negative charge caused by anionic membrane phospholipids. In the bacterium E. coli, phosphatidylethanolamine play a role in supporting lactose permeases active transport of lactose into the cell, and may play a role in other transport systems as well. Phosphatidylethanolamine plays a role in the assembly of lactose permease and other membrane proteins. It acts as a 'chaperone' to help the membrane proteins correctly fold their tertiary structures so that they can function properly. When phosphatidylethanolamine is not present, the transport proteins have incorrect tertiary structures and do not function correctly. [9]
Phosphatidylethanolamine also enables bacterial multidrug transporters to function properly and allows the formation of intermediates that are needed for the transporters to properly open and close. [10]
Structure

As a lecithin, phosphatidylethanolamine consists of a combination of glycerol esterified with two fatty acids and phosphoric acid. Whereas the phosphate group is combined with choline in phosphatidylcholine, it is combined with ethanolamine in phosphatidylethanolamine. The two fatty acids may be identical or different, and are usually found in positions 1,2 (less commonly in positions 1,3).
Synthesis
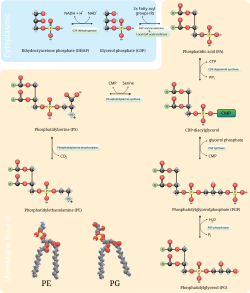
The phosphatidylserine decarboxylation pathway and the cytidine diphosphate-ethanolamine pathways are used to synthesize phosphatidylethanolamine. Phosphatidylserine decarboxylase is the enzyme that is used to decarboxylate phosphatidylserine in the first pathway. The phosphatidylserine decarboxylation pathway is the main source of synthesis for phosphatidylethanolamine in the membranes of the mitochondria. Phosphatidylethanolamine produced in the mitochondrial membrane is also transported throughout the cell to other membranes for use. In a process that mirrors phosphatidylcholine synthesis, phosphatidylethanolamine is also made via the cytidine diphosphate-ethanolamine pathway, using ethanolamine as the substrate. Through several steps taking place in both the cytosol and endoplasmic reticulum, the synthesis pathway yields the end product of phosphatidylethanolamine. [11] Phosphatidylethanolamine is also found abundantly in soy or egg lecithin and is produced commercially using chromatographic separation.
Regulation
Synthesis of phosphatidylethanolamine through the phosphatidylserine decarboxylation pathway occurs rapidly in the inner mitochondrial membrane. However, phosphatidylserine is made in the endoplasmic reticulum. Because of this, the transport of phosphatidylserine from the endoplasmic reticulum to the mitochondrial membrane and then to the inner mitochondrial membrane limits the rate of synthesis via this pathway. The mechanism for this transport is currently unknown but may play a role in the regulation of the rate of synthesis in this pathway. [12]
Presence in food, health issues
Phosphatidylethanolamines in food break down to form phosphatidylethanolamine-linked Amadori products as a part of the Maillard reaction. [13] These products accelerate membrane lipid peroxidation, causing oxidative stress to cells that come in contact with them. [14] Oxidative stress is known to cause food deterioration and several diseases. Significant levels of Amadori-phosphatidylethanolamine products have been found in a wide variety of foods such as chocolate, soybean milk, infant formula, and other processed foods. The levels of Amadori-phosphatidylethanolamine products are higher in foods with high lipid and sugar concentrations that have high temperatures in processing. [13] Additional studies have found that Amadori-phosphatidylethanolamine may play a role in vascular disease, [15] act as the mechanism by which diabetes can increase the incidence of cancer, [16] and potentially play a role in other diseases as well. Amadori-phosphatidylethanolamine has a higher plasma concentration in diabetes patients than healthy people, indicating it may play a role in the development of the disease or be a product of the disease. [17]
See also
References
- ^ Wellner, Niels; Diep, Thi Ai; Janfelt, Christian; Hansen, Harald Severin (2012). "N-acylation of phosphatidylethanolamine and its biological functions in mammals". Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids. 1831 (3): 652–62. doi: 10.1016/j.bbalip.2012.08.019. PMID 23000428.
- ^ a b Vance, Jean E.; Tasseva, Guergana (2012). "Formation and function of phosphatidylserine and phosphatidylethanolamine in mammalian cells". Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids. 1831 (3): 543–54. doi: 10.1016/j.bbalip.2012.08.016. PMID 22960354.
- ^ Emoto, K.; Kobayashi, T; Yamaji, A; Aizawa, H; Yahara, I; Inoue, K; Umeda, M (1996). "Redistribution of phosphatidylethanolamine at the cleavage furrow of dividing cells during cytokinesis". Proceedings of the National Academy of Sciences. 93 (23): 12867–72. Bibcode: 1996PNAS...9312867E. doi: 10.1073/pnas.93.23.12867. JSTOR 40713. PMC 24012. PMID 8917511.
- ^ See references in Wan et al. Biochemistry 47 2008[ verification needed]
- ^ Vance, J. E. (2008). "Thematic Review Series: Glycerolipids. Phosphatidylserine and phosphatidylethanolamine in mammalian cells: Two metabolically related aminophospholipids". The Journal of Lipid Research. 49 (7): 1377–87. doi: 10.1194/jlr.R700020-JLR200. PMID 18204094.
- ^ Deleault, N. R.; Piro, J. R.; Walsh, D. J.; Wang, F.; Ma, J.; Geoghegan, J. C.; Supattapone, S. (2012). "Isolation of phosphatidylethanolamine as a solitary cofactor for prion formation in the absence of nucleic acids". Proceedings of the National Academy of Sciences. 109 (22): 8546–51. Bibcode: 2012PNAS..109.8546D. doi: 10.1073/pnas.1204498109. PMC 3365173. PMID 22586108.
- ^ Majumder, R.; Liang, X.; Quinn-Allen, M. A.; Kane, W. H.; Lentz, B. R. (2011). "Modulation of Prothrombinase Assembly and Activity by Phosphatidylethanolamine". Journal of Biological Chemistry. 286 (41): 35535–42. doi: 10.1074/jbc.M111.260141. PMC 3195639. PMID 21859710.
- ^ Isidro, F. (2014). "Cannabinoids for treatment of Alzheimer's disease: moving toward the clinic". Frontiers in Pharmacology. 5: 37. doi: 10.3389/fphar.2014.00037. PMC 3942876. PMID 24634659.
- ^ Christie, W.W. (April 16, 2012). "Phosphatidylethanolamine and Related Lipids". The AOCS Lipid Library. Archived from the original on August 21, 2012. Retrieved September 3, 2012.
- ^ Gbaguidi, B.; Hakizimana, P.; Vandenbussche, G.; Ruysschaert, J.-M. (2007). "Conformational changes in a bacterial multidrug transporter are phosphatidylethanolamine-dependent" (PDF). Cellular and Molecular Life Sciences. 64 (12): 1571–82. doi: 10.1007/s00018-007-7031-0. PMID 17530171. S2CID 2078590.
- ^ Kelly, Karen (July 28, 2011). "Phospholipid Biosynthesis". The AOCS Lipid Library. Retrieved September 3, 2012.
- ^ Kuge, Osamu; Nishijima, Masahiro (1 April 2003). "Biosynthetic Regulation and Intracellular Transport of phosphatidylserine in Mammalian Cells". The Journal of Biochemistry. 133 (4): 397–403. doi: 10.1093/jb/mvg052. PMID 12761285. Archived from the original on 30 January 2021. Retrieved 30 January 2021.
- ^ a b Oak, Jeong-Ho; Nakagawa, Kiyotaka; Miyazawa, Teruo (2002). "UV analysis of Amadori-glycated phosphatidylethanolamine in foods and biological samples". The Journal of Lipid Research. 43 (3): 523–9. doi: 10.1016/S0022-2275(20)30158-9. PMID 11893788.
- ^ Oak, Jeong-Ho; Nakagawa, Kiyotaka; Miyazawa, Teruo (2000). "Synthetically prepared Amadori-glycated phosphatidylethanolamine can trigger lipid peroxidation via free radical reactions". FEBS Letters. 481 (1): 26–30. doi: 10.1016/S0014-5793(00)01966-9. PMID 10984609. S2CID 23265125.
- ^ Oak, Jeong-Ho; Nakagawa, Kiyotaka; Oikawa, Shinichi; Miyazawa, Teruo (2003). "Amadori-glycated phosphatidylethanolamine induces angiogenic differentiations in cultured human umbilical vein endothelial cells". FEBS Letters. 555 (2): 419–23. doi: 10.1016/S0014-5793(03)01237-7. PMID 14644453. S2CID 33974755.
- ^ Eitsuka, Takahiro; Nakagawa, Kiyotaka; Ono, Yuichi; Tatewaki, Naoto; Nishida, Hiroshi; Kurata, Tadao; Shoji, Naoki; Miyazawa, Teruo (2012). "Amadori-glycated phosphatidylethanolamine up-regulates telomerase activity in PANC-1 human pancreatic carcinoma cells". FEBS Letters. 586 (16): 2542–7. doi: 10.1016/j.febslet.2012.06.027. PMID 22750441. S2CID 5452160.
- ^ Ariizumi, Ken; Koike, T; Ohara, S; Inomata, Y; Abe, Y; Iijima, K; Imatani, A; Oka, T; Shimosegawa, T (2008). "Incidence of reflux esophagitis and H pylori infection in diabetic patients". World Journal of Gastroenterology. 14 (20): 3212–7. doi: 10.3748/wjg.14.3212. PMC 2712855. PMID 18506928.
External links
- Phosphatidylethanolamines at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Phosphatidylethanolamine at the AOCS Lipid Library.