| |

| |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
| |
| |
| Properties | |
| O8S2−2 | |
| Molar mass | 192.11 g·mol−1 |
| Conjugate acid | Peroxydisulfuric acid |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
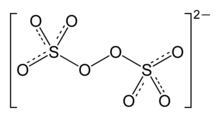
The peroxydisulfate ion, S
2O2−
8, is an
oxyanion, the anion of
peroxydisulfuric acid. It is commonly referred to as
persulfate, but this term also refers to the
peroxomonosulfate ion, SO2−
5. It is also called peroxodisulfate.
[2] Approximately 500,000 tons of salts containing this anion are produced annually. Important salts include
sodium persulfate (Na2S2O8),
potassium persulfate (K2S2O8), and
ammonium persulfate ((NH4)2S2O8). These salts are colourless, water-soluble solids that are strong oxidants.
[3]
Applications
Salts of peroxydisulfate are mainly used to initiate the polymerization of various alkenes, including styrene, acrylonitrile, and fluoroalkenes. Polymerization is initiated by the homolysis of the peroxydisulfate:
- [O3SO–OSO32− ⇌ 2 [SO4•−
Moreover, sodium peroxydisulfate can be used for soil and groundwater remediation, water and wastewater treatment, and etching of copper on circuit boards. [4] [2]
It has also been used to produce hair lighteners and bleaches, medical drugs, cellophane, rubber, soaps, detergents, adhesive papers, dyes for textiles, and in photography. [2]
In addition to its major commercial applications, peroxydisulfate participates in reactions of interest in the laboratory:
- Elbs persulfate oxidation
- Oxidation of Ag+ to Ag2+, such as the preparation of tetrakis(pyridine)silver(II) peroxydisulfate
Structure
Peroxydisulfate is a centrosymmetric anion. The O-O distance is 1.48 Å. The sulfur centers are tetrahedral. [5]
References
- ^ Ambiguous—see persulfate
- ^ a b c Shafiee, Saiful Arifin; Aarons, Jolyon; Hamzah, Hairul Hisham (2018). "Electroreduction of Peroxodisulfate: A Review of a Complicated Reaction". Journal of the Electrochemical Society. 165 (13): H785–H798. doi: 10.1149/2.1161811jes. S2CID 106396614.
-
^ Harald Jakob, Stefan Leininger, Thomas Lehmann, Sylvia Jacobi, Sven Gutewort. "Peroxo Compounds, Inorganic".
Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH.
doi:
10.1002/14356007.a19_177.pub2.
ISBN
978-3527306732.
{{ cite encyclopedia}}: CS1 maint: multiple names: authors list ( link) - ^ Wacławek, Stanisław; Lutze, Holger V.; Grübel, Klaudiusz; Padil, Vinod V.T.; Černík, Miroslav; Dionysiou, Dionysios.D. (2017). "Chemistry of persulfates in water and wastewater treatment: A review". Chemical Engineering Journal. 330: 44–62. doi: 10.1016/j.cej.2017.07.132.
- ^ Allan, David R. (2006). "Sodium Peroxodisulfate". Acta Crystallographica Section E. 62 (3): i44–i46. doi: 10.1107/S1600536806004302.