 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.233.114 |
| Chemical and physical data | |
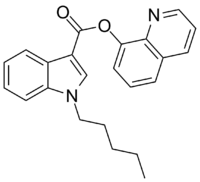
| Formula | C23H22N2O2 |
| Molar mass | 358.441 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
PB-22 (QUPIC, SGT-21 or 1-pentyl-1H-indole-3-carboxylic acid 8-quinolinyl ester) is a designer drug offered by online vendors as a cannabimimetic agent, and detected being sold in synthetic cannabis products in Japan in 2013. [1] [2] PB-22 represents a structurally unique synthetic cannabinoid chemotype, since it contains an ester linker at the indole 3-position, rather than the precedented ketone of JWH-018 and its analogs, or the amide of APICA and its analogs.
PB-22 has an EC50 of 5.1 nM for human CB1 receptors, and 37 nM for human CB2 receptors. [3] PB-22 produces bradycardia and hypothermia in rats at doses of 0.3–3 mg/kg, suggesting potent cannabinoid-like activity. [3] The magnitude and duration of hypothermia induced in rats by PB-22 was notably greater than JWH-018, AM-2201, UR-144, XLR-11, APICA, or STS-135, with a reduction of body temperature still observable six hours after dosing. [3] One clinical toxicology study found PB-22 to be the cause of seizures in a human and his dog. [4]
History
PB-22 was originally developed by New Zealand legal highs company Stargate International in 2012 as SGT-21, intended to be a structural hybrid of QMPSB and JWH-018. [5] However, no intellectual property protection was applied for and the compound quickly became subject to widespread grey-market sales outside the control of the inventors.
Detection
A forensic standard of PB-22 is available, and the compound has been posted on the Forendex website of potential drugs of abuse. [6]
Legal status
As of 9 May 2014, PB-22 is no longer legal in New Zealand. [7]
In January 2014, PB-22 was designated as a Schedule I controlled substance in the United States. [8] [9]
In Ohio, PB-22 is illegal. [10]
Florida also has banned PB-22. [11]
Since 13 December 2014 it is also illegal in Germany because of adding the substance to the BtMG Anlage II.
As of October 2015 PB-22 is a controlled substance in China. [12]
See also
References
- ^ Uchiyama N, Matsuda S, Kawamura M, Kikura-Hanajiri R, Goda Y (2013). "Two new-type cannabimimetic quinolinyl carboxylates, QUPIC and QUCHIC, two new cannabimimetic carboxamide derivatives, ADB-FUBINACA and ADBICA, and five synthetic cannabinoids detected with a thiophene derivative α-PVT and an opioid receptor agonist AH-7921 identified in illegal products". Forensic Toxicology. 31 (2): 223–240. doi: 10.1007/s11419-013-0182-9. S2CID 1279637.
- ^ Lin M, Ellis B, Eubanks LM, Janda KD (July 2021). "Pharmacokinetic Approach to Combat the Synthetic Cannabinoid PB-22". ACS Chemical Neuroscience. 12 (14): 2573–2579. doi: 10.1021/acschemneuro.1c00360. PMID 34254505. S2CID 235808519.
- ^ a b c Banister SD, Stuart J, Kevin RC, Edington A, Longworth M, Wilkinson SM, et al. (August 2015). "Effects of bioisosteric fluorine in synthetic cannabinoid designer drugs JWH-018, AM-2201, UR-144, XLR-11, PB-22, 5F-PB-22, APICA, and STS-135". ACS Chemical Neuroscience. 6 (8): 1445–1458. doi: 10.1021/acschemneuro.5b00107. PMID 25921407.
- ^ Gugelmann H, Gerona R, Li C, Tsutaoka B, Olson KR, Lung D (July 2014). "'Crazy Monkey' poisons man and dog: Human and canine seizures due to PB-22, a novel synthetic cannabinoid". Clinical Toxicology. 52 (6): 635–638. doi: 10.3109/15563650.2014.925562. PMID 24905571. S2CID 207647659.
- ^ Brandt SD, Kavanagh PV, Westphal F, Dreiseitel W, Dowling G, Bowden MJ, Williamson JP (January 2021). "Synthetic cannabinoid receptor agonists: Analytical profiles and development of QMPSB, QMMSB, QMPCB, 2F-QMPSB, QMiPSB, and SGT-233". Drug Testing and Analysis. 13 (1): 175–196. doi: 10.1002/dta.2913. PMID 32880103.
- ^ Forendex entry, Southern Association of Forensic Scientists
- ^ Jones N (1 May 2014). "Legal highs pulled from shelves". New Zealand Herald. New Zealand Media and Entertainment. Retrieved 18 August 2020.
- ^ Behonick G, Shanks KG, Firchau DJ, Mathur G, Lynch CF, Nashelsky M, et al. (October 2014). "Four postmortem case reports with quantitative detection of the synthetic cannabinoid, 5F-PB-22". Journal of Analytical Toxicology. 38 (8): 559–562. doi: 10.1093/jat/bku048. PMC 4334789. PMID 24876364.
- ^ "PB-22 and 5F-PB-22" (PDF). Drug Enforcement Administration, Office of Diversion Control.
- ^ Pelzer J (April 17, 2014). "Ohio bans two synthetic marijuana drugs sold as "herbal incense"". cleveland.com.
- ^ "Statutes & Constitution :View Statutes : Online Sunshine". Leg.state.fl.us. 1997-05-06. Retrieved 2014-07-12.
- ^ "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" (in Chinese). China Food and Drug Administration. 27 September 2015. Archived from the original on 1 October 2015. Retrieved 1 October 2015.