
Nonmetallic material, or in nontechnical terms a nonmetal, refers to materials which are not metals. Depending upon context it is used in slightly different ways. In everyday life it would be a generic term for those materials such as plastics, wood or ceramics which are not typical metals such as the iron alloys used in bridges. In some areas of chemistry, particularly the periodic table, it is used for just those chemical elements which are not metallic at standard temperature and pressure conditions. It is also sometimes used to describe broad classes of dopant atoms in materials. In general usage in science, it refers to materials which do not have electrons that can readily move around, more technically there are no available states at the Fermi energy, the equilibrium energy of electrons. For historical reasons there is a very different definition of metals in astronomy, with just hydrogen and helium as nonmetals. The term may also be used as a negative of the materials of interest such as in metallurgy or metalworking.
Variations in the environment, particularly temperature and pressure can change a nonmetal into a metal, and vica versa; this is always associated with some major change in the structure, a phase transition. Other external stimuli such as electric fields can also lead to a local nonmetal, for instance in certain semiconductor devices. There are also many physical phenomena which are only found in nonmetals such as piezoelectricity or flexoelectricity.
General definition

A nonmetal has a gap in the energy levels of the electrons at the Fermi level. [1]: Chpt 8 & 19 In contrast, a metal has at least one partially occupied band at the Fermi level; [1] in a semiconductor or insulator there are no states at the Fermi level, see for instance Ashcroft and Mermin. [1] These definitions are equivalent to stating that metals conduct electricity at absolute zero, as suggested by Nevill Francis Mott, [2]: 257 and the equivalent definition at other temperatures is also commonly used as in textbooks such as Chemistry of the Non-Metals by Ralf Steudel [3] and work on metal–insulator transitions. [4] [5]
Band structure definitions of metallicity are the most widely used, and apply both to single elements such as insulating boron [6] as well as compounds such as strontium titanate. [7] (There are many compounds which have states at the Fermi level and are metallic, for instance titanium nitride. [8]) There are many experimental methods of checking for nonmetals by measuring the band gap, or by ab-initio quantum mechanical calculations. [9]
Functional definition

An alternative in metallurgy is to consider various malleable alloys such as steel, aluminium alloys and similar as metals, and other materials as nonmetals; [10] fabricating metals is termed metalworking, [11] but there is no corresponding term for nonmetals. A loose definition such as this is often the common useage, but can also be inaccurate. For instance, in this useage plastics are nonmetals, but in fact there are (electrically) conducting polymers [12] [13] which should be described formally as metals. Similar, but slightly more complex, many materials which are (nonmetal) semiconductors behave like metals when they contain a high concentration of dopants, being called degenerate semiconductors. [14] A general introduction to much of this can be found in the 2017 book by Fumiko Yonezawa [2]: Chpt 1
Periodic table elements
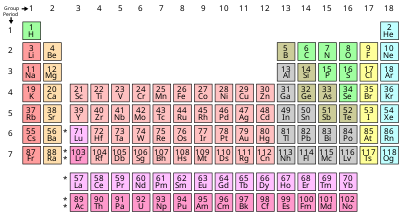
The term nonmetal (chemistry) is also used for those elements which are not metallic in their normal ground state; compounds are sometimes excluded from consideration. Some textbooks use the term nonmetallic elements such as the Chemistry of the Non-Metals by Ralf Steudel, [15]: 4 which also uses the general definition in terms of conduction and the Fermi level. [15]: 154 The approach based upon the elements is often used in teaching to help students understand the periodic table of elements, [16] although it is a teaching oversimplification. [17] [18] Those elements towards the top right of the periodic table are nonmetals, those towards the center ( transition metal and lanthanide) and the left are metallic. An intermediate designation metalloid is used for some elements.
The term is sometimes also used when describing dopants of specific elements types in compounds, alloys or combinations of materials, using the periodic table classification. For instance metalloids are often used in high-temperature alloys, [19] and nonmetals in precipitation hardening in steels and other alloys. [20] Here the description implicitly includes information on whether the dopants tend to be electron acceptors that lead to covalently bonded compounds rather than metallic bonding or electron acceptors.
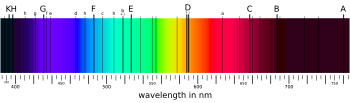
Nonmetals in astronomy
A quite different approach is used in astronomy where the term metallicity is used for all elements heavier than helium, so the only nonmetals are hydrogen and helium. This is a historical anomaly. In 1802, William Hyde Wollaston [21] noted the appearance of a number of dark features in the solar spectrum. [22] In 1814, Joseph von Fraunhofer independently rediscovered the lines and began to systematically study and measure their wavelengths, and they are now called Fraunhofer lines. He mapped over 570 lines, designating the most prominent with the letters A through K and weaker lines with other letters. [23] [24] [25]
About 45 years later, Gustav Kirchhoff and Robert Bunsen [26] noticed that several Fraunhofer lines coincide with characteristic emission lines identifies in the spectra of heated chemical elements. [27] They inferred that dark lines in the solar spectrum are caused by absorption by chemical elements in the solar atmosphere. [28] Their observations [29] were in the visible range where the strongest lines come from metals such as Na, K, Fe and thus all the extra elements beyond just hydrogen and helium were termed metallic.
The astrophysicst Carlos Jaschek, and the stellar astronomer and spectroscopist Mercedes Jaschek, in their book The Classification of Stars, observed that: [30]
- Stellar interior specialists use 'metals' to designate any element other than hydrogen and helium, and in consequence ‘metal abundance’ implies all elements other than the first two. For spectroscopists this is very misleading, because they use the word in the chemical sense. On the other hand photometrists, who observe combined effects of all lines (i.e. without distinguishing the different elements) often use this word 'metal abundance', in which case it may also include the effect of the hydrogen lines.
Metal-insulator transition

There are many cases where an element or compound is metallic under certain circumstances, but a nonmetal in others. One example is metallic hydrogen which forms under very high pressures. [32] There are many other cases as discussed by Mott, [4] Inada et al [5] and more recently by Yonezawa. [2]
There can also be local transitions to a nonmetal, particularly in semiconductor devices. One example is a field-effect transistor where an electric field can lead to a region where there are no electrons at the Fermi energy ( depletion zone). [33] [34]
Properties specific to nonmetals

Nonmetals have a wide range of properties, for instance the nonmetal diamond is the hardest known material, while the nonmetal molybdenum disulfide is a solid lubricants used in space. [35] There are some properties specific to them not having electrons at the Fermi energy. The main ones, for which more details are available in the links are: [1]: Chpt 27-29 [36]
- Dielectric polarization, [37] approximately equivalent to alignment of local dipoles with an electric field, as in capacitors.
- Electrostriction, [38] a change in volume due to an electric field, or more accurately polarization density.
- Flexoelectricity, where there is a coupling between strain gradients and polarization. [39] This plays a role in the generation of static electricity due to the triboelectric effect. [40]
- Piezoelectricity, a coupling between polarization and linear strains. [38]
- A decreased resistance with temperature, due to having more carriers (via Fermi–Dirac statistics) available in partially occupied higher energy bands [1] [41]
- Increased conductivity when illuminated with light or ultraviolet radiation, called photoconductivity. This is similar to the effect of temperature, but with the photons exciting electrons into partially occupied states. [42]
- Transmit electric fields as in the capacitor figure above; in a metal there is electric-field screening that prevents this beyond very small distances, see Classical Electrodynamics. [43]
See also
- Abundance of the chemical elements – Measure of the relative occurrences of chemical elements in a given context
- List of data references for chemical elements
- List of manufacturing processes – Manufacturing processes
- List of materials properties
- Metallicity distribution function – Distribution within a group of stars of the ratio of iron to hydrogen in a star
- Superconductor-insulator transition – Type of quantum phase transition
References
- ^ a b c d e Ashcroft, Neil W.; Mermin, N. David (1976). Solid state physics. Fort Worth Philadelphia San Diego [etc.]: Saunders college publ. ISBN 978-0-03-083993-1.
- ^ a b c Yonezawa, Fumiko (2017). Physics of metal-nonmetal transitions. Washington, DC: IOS Press. ISBN 978-1-61499-786-3.
- ^ Steudel, Ralf (2020). Chemistry of the Non-Metals: Syntheses - Structures - Bonding - Applications. De Gruyter. p. 154. doi: 10.1515/9783110578065. ISBN 978-3-11-057806-5.
- ^ a b Mott, N. F. (1968). "Metal-Insulator Transition". Reviews of Modern Physics. 40 (4): 677–683. Bibcode: 1968RvMP...40..677M. doi: 10.1103/RevModPhys.40.677. ISSN 0034-6861.
- ^ a b Imada, Masatoshi; Fujimori, Atsushi; Tokura, Yoshinori (1998). "Metal-insulator transitions". Reviews of Modern Physics. 70 (4): 1039–1263. Bibcode: 1998RvMP...70.1039I. doi: 10.1103/RevModPhys.70.1039. ISSN 0034-6861.
- ^ Ogitsu, Tadashi; Schwegler, Eric; Galli, Giulia (2013). "β-Rhombohedral Boron: At the Crossroads of the Chemistry of Boron and the Physics of Frustration". Chemical Reviews. 113 (5): 3425–3449. doi: 10.1021/cr300356t. ISSN 0009-2665. OSTI 1227014. PMID 23472640.
- ^ Reihl, B.; Bednorz, J. G.; Müller, K. A.; Jugnet, Y.; Landgren, G.; Morar, J. F. (1984). "Electronic structure of strontium titanate". Physical Review B. 30 (2): 803–806. Bibcode: 1984PhRvB..30..803R. doi: 10.1103/PhysRevB.30.803. ISSN 0163-1829.
- ^ Höchst, H.; Bringans, R. D.; Steiner, P.; Wolf, Th. (1982). "Photoemission study of the electronic structure of stoichiometric and substoichiometric TiN and ZrN". Physical Review B. 25 (12): 7183–7191. Bibcode: 1982PhRvB..25.7183H. doi: 10.1103/PhysRevB.25.7183. ISSN 0163-1829.
- ^ Kim, Sangtae; Lee, Miso; Hong, Changho; Yoon, Youngchae; An, Hyungmin; Lee, Dongheon; Jeong, Wonseok; Yoo, Dongsun; Kang, Youngho; Youn, Yong; Han, Seungwu (2020). "A band-gap database for semiconducting inorganic materials calculated with hybrid functional". Scientific Data. 7 (1): 387. Bibcode: 2020NatSD...7..387K. doi: 10.1038/s41597-020-00723-8. ISSN 2052-4463. PMC 7658987. PMID 33177500.
- ^ Cottrell, Alan (1985). An introduction to metallurgy: SI units (2. ed., repr ed.). London: Arnold. ISBN 978-0-7131-2510-8.
- ^ DeGarmo, E. Paul, ed. (2003). Materials and processes in manufacturing (9th ed.). Hoboken, N.J: Wiley. ISBN 978-0-471-65653-1.
- ^ Waltman, R. J.; Bargon, J. (1986). "Electrically conducting polymers: a review of the electropolymerization reaction, of the effects of chemical structure on polymer film properties, and of applications towards technology". Canadian Journal of Chemistry. 64 (1): 76–95. doi: 10.1139/v86-015. ISSN 0008-4042.
- ^ Das, Tapan K.; Prusty, Smita (2012). "Review on Conducting Polymers and Their Applications". Polymer-Plastics Technology and Engineering. 51 (14): 1487–1500. doi: 10.1080/03602559.2012.710697. ISSN 0360-2559.
- ^ Wolff, P. A. (1962). "Theory of the Band Structure of Very Degenerate Semiconductors". Physical Review. 126 (2): 405–412. Bibcode: 1962PhRv..126..405W. doi: 10.1103/PhysRev.126.405. ISSN 0031-899X.
- ^ a b Steudel, Ralf (2020). Chemistry of the Non-Metals: Syntheses - Structures - Bonding - Applications. De Gruyter. p. 4. doi: 10.1515/9783110578065. ISBN 978-3-11-057806-5.
- ^ "1.8: Introduction to the Periodic Table". Chemistry LibreTexts. 2015. Retrieved 2024-06-16.
- ^ Worstall, Tim (2015). "'The Scientific Method' Is A Myth, Long Live The Scientific Method". Forbes. Archived from the original on 2015-11-12. Retrieved 2024-06-27.
- ^ Worstall, Tim (2015). "To Prove Econ 101 Is Wrong You Do Need To Understand Econ 101". Forbes. Retrieved 2024-06-27.
- ^ Perepezko, John H. (2009). "The Hotter the Engine, the Better". Science. 326 (5956): 1068–1069. Bibcode: 2009Sci...326.1068P. doi: 10.1126/science.1179327. ISSN 0036-8075. PMID 19965415.
- ^ Ardell, A. J. (1985). "Precipitation hardening". Metallurgical Transactions A. 16 (12): 2131–2165. Bibcode: 1985MTA....16.2131A. doi: 10.1007/BF02670416. ISSN 0360-2133.
- ^ Melvyn C. Usselman: William Hyde Wollaston Encyclopædia Britannica, retrieved 31 March 2013
- ^ William Hyde Wollaston (1802) "A method of examining refractive and dispersive powers, by prismatic reflection," Philosophical Transactions of the Royal Society, 92: 365–380; see especially p. 378.
- ^ Hearnshaw, J.B. (1986). The analysis of starlight. Cambridge: Cambridge University Press. p. 27. ISBN 978-0-521-39916-6.
- ^ Joseph Fraunhofer (1814 - 1815) "Bestimmung des Brechungs- und des Farben-Zerstreuungs - Vermögens verschiedener Glasarten, in Bezug auf die Vervollkommnung achromatischer Fernröhre" (Determination of the refractive and color-dispersing power of different types of glass, in relation to the improvement of achromatic telescopes), Denkschriften der Königlichen Akademie der Wissenschaften zu München (Memoirs of the Royal Academy of Sciences in Munich), 5: 193–226; see especially pages 202–205 and the plate following page 226.
- ^ Jenkins, Francis A.; White, Harvey E. (1981). Fundamentals of Optics (4th ed.). McGraw-Hill. p. 18. ISBN 978-0-07-256191-3.
-
^ See:
- Gustav Kirchhoff (1859) "Ueber die Fraunhofer'schen Linien" (On Fraunhofer's lines), Monatsbericht der Königlichen Preussische Akademie der Wissenschaften zu Berlin (Monthly report of the Royal Prussian Academy of Sciences in Berlin), 662–665.
- Gustav Kirchhoff (1859) "Ueber das Sonnenspektrum" (On the sun's spectrum), Verhandlungen des naturhistorisch-medizinischen Vereins zu Heidelberg (Proceedings of the Natural History / Medical Association in Heidelberg), 1 (7) : 251–255.
- ^ G. Kirchhoff (1860). "Ueber die Fraunhofer'schen Linien". Annalen der Physik. 185 (1): 148–150. Bibcode: 1860AnP...185..148K. doi: 10.1002/andp.18601850115.
- ^ G. Kirchhoff (1860). "Ueber das Verhältniss zwischen dem Emissionsvermögen und dem Absorptionsvermögen der Körper für Wärme und Licht" [On the relation between the emissive power and the absorptive power of bodies towards heat and light]. Annalen der Physik. 185 (2): 275–301. Bibcode: 1860AnP...185..275K. doi: 10.1002/andp.18601850205.
- ^ "Kirchhoff and Bunsen on Spectroscopy". www.chemteam.info. Retrieved 2024-07-02.
- ^ Jaschek, C; Jascheck, M (1990). The Classification of Stars. Cambridge: Cambridge University Press. p. 22. ISBN 978-0-521-26773-1.
- ^ Shao, Zewei; Cao, Xun; Luo, Hongjie; Jin, Ping (2018). "Recent progress in the phase-transition mechanism and modulation of vanadium dioxide materials". NPG Asia Materials. 10 (7): 581–605. Bibcode: 2018npgAM..10..581S. doi: 10.1038/s41427-018-0061-2. ISSN 1884-4049.
- ^ Wigner, E.; Huntington, H. B. (1935). "On the Possibility of a Metallic Modification of Hydrogen". The Journal of Chemical Physics. 3 (12): 764–770. Bibcode: 1935JChPh...3..764W. doi: 10.1063/1.1749590. ISSN 0021-9606.
- ^ Kushwah, D. S. (1975). "Field Effect Transistors-A review of their growth and the state of the art". IETE Journal of Education. 16 (3): 126–132. doi: 10.1080/09747338.1975.11450118. ISSN 0974-7338.
- ^ Zhang, Shubo (2020). "Review of Modern Field Effect Transistor Technologies for Scaling". Journal of Physics: Conference Series. 1617 (1): 012054. Bibcode: 2020JPhCS1617a2054Z. doi: 10.1088/1742-6596/1617/1/012054. ISSN 1742-6588.
- ^ Vazirisereshk, Mohammad R.; Martini, Ashlie; Strubbe, David A.; Baykara, Mehmet Z. (2019). "Solid Lubrication with MoS2: A Review". Lubricants. 7 (7): 57. arXiv: 1906.05854. doi: 10.3390/lubricants7070057. ISSN 2075-4442.
- ^ Griffiths, David J. (2017). Introduction to Electrodynamics (4 ed.). Cambridge University Press. doi: 10.1017/9781108333511.008. ISBN 978-1-108-33351-1.
- ^ "Dielectric | Definition, Properties, & Polarization | Britannica". 2021. Archived from the original on 2021-04-27. Retrieved 2024-06-16.
- ^ a b "Electrostriction | Piezoelectricity, Ferroelectricity, Magnetostriction | Britannica". www.britannica.com. Retrieved 2024-06-16.
- ^ Zubko, Pavlo; Catalan, Gustau; Tagantsev, Alexander K. (2013). "Flexoelectric Effect in Solids". Annual Review of Materials Research. 43 (1): 387–421. Bibcode: 2013AnRMS..43..387Z. doi: 10.1146/annurev-matsci-071312-121634. hdl: 10261/99362. ISSN 1531-7331.
- ^ Mizzi, C. A.; Lin, A. Y. W.; Marks, L. D. (2019). "Does Flexoelectricity Drive Triboelectricity?". Physical Review Letters. 123 (11): 116103. arXiv: 1904.10383. Bibcode: 2019PhRvL.123k6103M. doi: 10.1103/PhysRevLett.123.116103. ISSN 0031-9007. PMID 31573269.
- ^ Kasap, Safa; Koughia, Cyril; Ruda, Harry E. (2017), Kasap, Safa; Capper, Peter (eds.), "Electrical Conduction in Metals and Semiconductors", Springer Handbook of Electronic and Photonic Materials, Cham: Springer International Publishing, p. 1, doi: 10.1007/978-3-319-48933-9_2, ISBN 978-3-319-48933-9, retrieved 2024-06-30
- ^ Rogers, Alan (2009). Essentials of Photonics (2nd ed.). CRC Press. doi: 10.1201/9781315222042. ISBN 9781315222042.
- ^ Jackson, John David (2009). Classical electrodynamics (3rd ed.). Hoboken, NY: Wiley. ISBN 978-0-471-30932-1.