From Wikipedia, the free encyclopedia
(Redirected from
Naphthoylindole)
Class of chemical compounds
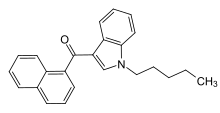
Naphthoylindoles are a class of synthetic cannabinoids. [1]
Pharmacology
Behaving similarly in vivo to endocannabinoids such as anandamide or 2-arachidonoylglycerol (2-AG), Naphthoylindoles can bind to endocannabinoid receptors in animals, presenting as CB1 and/or CB2 partial/ full agonists.
History
They have gained notoriety over the years for illicit usage and distribution in Europe and North America, typically marketed as "herbal incense blends." [2]
See also
- Structural scheduling of synthetic cannabinoids
- List of JWH cannabinoids, includes many naphthoylindoles
- Naphthoyl, an acyl group, derived, in this case, from 1-naphthoic acid
- Indole
References
- ^ Manera C, Tuccinardi T, Martinelli A (2008). "Indoles and related compounds as cannabinoid ligands". Mini Reviews in Medicinal Chemistry. 8 (4): 370–87. doi: 10.2174/138955708783955935. PMID 18473928.
- ^ "Synthetic cannabinoids drug profile". emcdda.europa.eu. EMCDDA. Retrieved 21 May 2023.
Retrieved from "
https://en.wikipedia.org/?title=Naphthoylindoles&oldid=1205945337"