Mauveine, also known as aniline purple and Perkin's mauve, was one of the first synthetic dyes. [1] [2] It was discovered serendipitously by William Henry Perkin in 1856 while he was attempting to synthesise the phytochemical quinine for the treatment of malaria. [3] It is also among the first chemical dyes to have been mass-produced. [4] [5]
Chemistry
Mauveine is a mixture of four related aromatic compounds differing in number and placement of methyl groups. Its organic synthesis involves dissolving aniline, p-toluidine, and o-toluidine in sulfuric acid and water in a roughly 1:1:2 ratio, then adding potassium dichromate. [6]
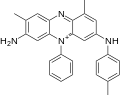
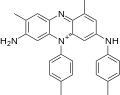
Mauveine A (C26H23N+4X−) incorporates 2 molecules of aniline, one of p-toluidine, and one of o-toluidine. Mauveine B (C27H25N+4X−) incorporates one molecule each of aniline, p-toluidine, and two of o-toluidine. In 1879, Perkin showed mauveine B related to safranines by oxidative/ reductive loss of the p-tolyl group. [7] In fact, safranine is a 2,8-dimethyl phenazinium salt, whereas the parasafranine produced by Perkin is presumed [8] to be the 1,8- (or 2,9-) dimethyl isomer.
The molecular structure of mauveine proved difficult to determine, finally being identified in 1994. [9] In 2007, two more were isolated and identified: mauveine B2, an isomer of mauveine B with methyl on different aryl group, and mauveine C, which has one more p-methyl group than mauveine A. [10]
-
Skeletal formula of mauveine A
-
Skeletal formula of mauveine B
-
Skeletal formula of mauveine B2
-
Skeletal formula of mauveine C
In 2008, additional mauveines and pseudomauveines were discovered, bringing the total number of these compounds up to 12. [11] In 2015 a crystal structure was reported for the first time. [12]
History

Mauveine #8D029B
#8D029B
In 1856, William Henry Perkin, then age 18, was given a challenge by his professor, August Wilhelm von Hofmann, to synthesize quinine. In one attempt, Perkin oxidized aniline using potassium dichromate, whose toluidine impurities reacted with the aniline and yielded a black solid, suggesting a "failed" organic synthesis. Cleaning the flask with alcohol, Perkin noticed purple portions of the solution.
Suitable as a dye of silk and other textiles, it was patented by Perkin, who the next year opened a dyeworks mass-producing it at Greenford on the banks of the Grand Union Canal in Middlesex. [13] It was originally called aniline purple. In 1859, it was named mauve in England via the French name for the mallow flower, and chemists later called it mauveine. [14] Between 1859 and 1861, mauve became a fashion must have. The weekly journal All the Year Round described women wearing the colour as "all flying countryward, like so many migrating birds of purple paradise". [15] Punch magazine published cartoons poking fun at the huge popularity of the colour “The Mauve Measles are spreading to so serious an extent that it is high time to consider by what means [they] may be checked.” [16] [17] [18]
By 1870, demand succumbed to newer synthetic colors in the synthetic dye industry launched by mauveine.
In the early 20th century, the U.S. National Association of Confectioners permitted mauveine as a food coloring with a variety of equivalent names: rosolan, violet paste, chrome violet, anilin violet, anilin purple, Perkin's violet, indisin, phenamin, purpurin and lydin. [19]
Laborers in the aniline dye industry were later found to be at increased risk of bladder cancer, specifically transitional cell carcinoma, yet by the 1950s, the synthetic dye industry had helped transform medicine, including cancer treatment. [20] [21] [22]
References
- ^ Hubner (2006). "History – 150 Years of mauveine". Chemie in unserer Zeit. 40 (4): 274–275. doi: 10.1002/ciuz.200690054.
- ^ Anthony S. Travis (1990). "Perkin's Mauve: Ancestor of the Organic Chemical Industry". Technology and Culture. 31 (1): 51–82. doi: 10.2307/3105760. JSTOR 3105760. S2CID 112031120.
- ^ St. Clair, Kassia (2016). The Secret Lives of Colour. London: John Murray. p. 21. ISBN 9781473630819. OCLC 936144129.
- ^ Hicks, Jan (2017-08-25). "William Henry Perkin and the world's first synthetic dye". Science and Industry Museum blog. Retrieved 2019-10-07.
- ^ "The color purple: How an accidental discovery changed fashion forever". CNN. 12 March 2018.
- ^ A Microscale Synthesis of Mauve Scaccia, Rhonda L.; Coughlin, David; Ball, David W. J. Chem. Educ. 1998 75 769 Abstract
- ^ Perkin, W. H. (January 1879). "On mauveine and allied colouring matters". J. Chem. Soc. Trans. 1879: 717–732. doi: 10.1039/CT8793500717.
- ^ Website source: ch.ic.ac.uk Link
- ^ Meth-Cohn, O.; Smith, M. (1994). "What did W. H. Perkin actually make when he oxidised aniline to obtain mauveine?". Journal of the Chemical Society, Perkin Transactions 1. 1994: 5–7. doi: 10.1039/P19940000005.
- ^ Seixas De Melo, J.; Takato, S.; Sousa, M.; Melo, M. J.; Parola, A. J. (2007). "Revisiting Perkin's dye(s): The spectroscopy and photophysics of two new mauveine compounds (B2 and C)". Chemical Communications (25): 2624–6. doi: 10.1039/b618926a. PMID 17579759.
- ^ Sousa, Micaela M.; Melo, Maria J.; Parola, A. Jorge; Morris, Peter J. T.; Rzepa, Henry S.; De Melo, J. Sérgio Seixas (2008). "A Study in Mauve: Unveiling Perkin's Dye in Historic Samples". Chemistry - A European Journal. 14 (28): 8507–8513. doi: 10.1002/chem.200800718. hdl: 10316/8229. PMID 18671308.
- ^ Plater, M. John; Harrison, William T. A.; Rzepa, Henry S. (2015). "Syntheses and Structures of Pseudo-Mauveine Picrate and 3-Phenylamino-5-(2-Methylphenyl)-7-Amino-8-Methylphenazinium Picrate Ethanol Mono-Solvate: The First Crystal Structures of a Mauveine Chromophore and a Synthetic Derivative". Journal of Chemical Research. 39 (12): 711–718. doi: 10.3184/174751915X14474318419130. hdl: 2164/5423.
- ^ Google Earth location: Download
-
^ Matthew, H.C.G.; Howard Harrison, Brian (2004).
Oxford Dictionary of National Biography: In Association with the British Academy.
Oxford University Press.
ISBN
0-19-861393-8.
perkins tyrian.purple.
- ^ Garfield, Simon (2000-09-21). "Simon Garfield on mauve". The Guardian. ISSN 0261-3077. Retrieved 2020-05-27.
- ^ Blakemore, Erin. "How Malaria Gave Us Mauve". Smithsonian Magazine. Retrieved 2020-05-27.
- ^ Jackson, Shelley. "Colors / Mauve | Shelley Jackson". cabinetmagazine.org. Retrieved 2020-05-27.
- ^ "Day dress | V&A Search the Collections". V and A Collections. 2020-05-27. Retrieved 2020-05-27.
-
^ Leffmann, Henry; William Beam (1901).
Select Methods in Food Analysis. Philadelphia: P. Blakiston's Son & Co. p.
77.
perkins tyrian.purple.
- ^ Cartwright, R.A. (1983). "Historical and modern epidemiological studies on populations exposed to N-substituted aryl compounds". Environmental Health Perspectives. 49: 13–19. doi: 10.1289/ehp.834913. PMC 1569142. PMID 6339220.
- ^ John E Lesch, The First Miracle Drugs: How the Sulfa Drugs Transformed Medicine (New York: Oxford University Press, 2007), pp 202–3
- ^ D J Th Wagener, The History of Oncology (Houten: Springer, 2009), pp 150–1.
Further reading
- Simon Garfield (2002). Mauve: How One Man Invented a Color That Changed the World. W. W. Norton & Company. ISBN 978-0393323139.
External links
- Perkin anniversary website Archived 2006-11-11 at the Wayback Machine
- Rotatable 3D models of mauveine are available using Jmol