| Marine habitats |
|---|
 |
| Coastal habitats |
| Ocean surface |
| Open ocean |
| Sea floor |
A marine coastal ecosystem is a marine ecosystem which occurs where the land meets the ocean. Marine coastal ecosystems include many very different types of marine habitats, each with their own characteristics and species composition. They are characterized by high levels of biodiversity and productivity.
For example, estuaries are areas where freshwater rivers meet the saltwater of the ocean, creating an environment that is home to a wide variety of species, including fish, shellfish, and birds. Salt marshes are coastal wetlands which thrive on low-energy shorelines in temperate and high-latitude areas, populated with salt-tolerant plants such as cordgrass and marsh elder that provide important nursery areas for many species of fish and shellfish. Mangrove forests survive in the intertidal zones of tropical or subtropical coasts, populated by salt-tolerant trees that protect habitat for many marine species, including crabs, shrimp, and fish.
Further examples are coral reefs and seagrass meadows, which are both found in warm, shallow coastal waters. Coral reefs thrive in nutrient-poor waters on high-energy shorelines that are agitated by waves. They are underwater ecosystem made up of colonies of tiny animals called coral polyps. These polyps secrete hard calcium carbonate skeletons that builds up over time, creating complex and diverse underwater structures. These structures function as some of the most biodiverse ecosystems on the planet, providing habitat and food for a huge range of marine organisms. Seagrass meadows can be adjacent to coral reefs. These meadows are underwater grasslands populated by marine flowering plants that provide nursery habitats and food sources for many fish species, crabs and sea turtles, as well as dugongs. In slightly deeper waters are kelp forests, underwater ecosystems found in cold, nutrient-rich waters, primarily in temperate regions. These are dominated by a large brown algae called kelp, a type of seaweed that grows several meters tall, creating dense and complex underwater forests. Kelp forests provide important habitats for many fish species, sea otters and sea urchins.
Directly and indirectly, marine coastal ecosystems provide vast arrays of ecosystem services for humans, such as cycling nutrients and elements, and purifying water by filtering pollutants. They sequester carbon as a cushion against climate change. They protect coasts by reducing the impacts of storms, reducing coastal erosion and moderating extreme events. They provide essential nurseries and fishing grounds for commercial fisheries. They provide recreational services and support tourism.
These ecosystems are vulnerable to various anthropogenic and natural disturbances, such as pollution, overfishing, and coastal development, which have significant impacts on their ecological functioning and the services they provide. Climate change is impacting coastal ecosystems with sea level rises, ocean acidification, and increased storm frequency and intensity. When marine coastal ecosystems are damaged or destroyed, there can be serious consequences for the marine species that depend on them, as well as for the overall health of the ocean ecosystem. Some conservation efforts are underway to protect and restore marine coastal ecosystems, such as establishing marine protected areas and developing sustainable fishing practices.
Overview


Coastal seas are highly productive systems, providing an array of ecosystem services to humankind, such as processing of nutrient effluents from land and climate regulation. [1] However, coastal ecosystems are threatened by human-induced pressures such as climate change and eutrophication. In the coastal zone, the fluxes and transformations of nutrients and carbon sustaining coastal ecosystem functions and services are strongly regulated by benthic (that is, occurring at the seafloor) biological and chemical processes. [1]
Coastal systems also contribute to the regulation of climate and nutrient cycles, by efficiently processing anthropogenic emissions from land before they reach the ocean. [2] [3] [4] [5] The high value of these ecosystem services is obvious considering that a large proportion of the world population lives close to the coast. [6] [7] [1]
Currently, coastal seas around the world are undergoing major ecological changes driven by human-induced pressures, such as climate change, anthropogenic nutrient inputs, overfishing and the spread of invasive species. [8] [9] In many cases, the changes alter underlying ecological functions to such an extent that new states are achieved and baselines are shifted. [10] [11] [1]
In 2015, the United Nations established 17 Sustainable Development Goals with the aim of achieving certain targets by 2030. Their mission statement for their 14th goal, Life below water, is to "conserve and sustainably use the oceans, seas and marine resources for sustainable development". [12] The United Nations has also declared 2021–2030 the UN Decade on Ecosystem Restoration, but restoration of coastal ecosystems is not receiving appropriate attention. [13]
Coastal habitats
Intertidal zone

Intertidal zones are the areas that are visible and exposed to air during low tide and covered up by saltwater during high tide. [14] There are four physical divisions of the intertidal zone with each one having its distinct characteristics and wildlife. These divisions are the Spray zone, High intertidal zone, Middle Intertidal zone, and Low intertidal zone. The Spray zone is a damp area that is usually only reached by the ocean and submerged only under high tides or storms. The high intertidal zone is submerged at high tide but remains dry for long periods between high tides. [14] Due to the large variance of conditions possible in this region, it is inhabited by resilient wildlife that can withstand these changes such as barnacles, marine snails, mussels and hermit crabs. [14] Tides flow over the middle intertidal zone two times a day and this zone has a larger variety of wildlife. [14] The low intertidal zone is submerged nearly all the time except during the lowest tides and life is more abundant here due to the protection that the water gives. [14]
Estuaries

Estuaries occur where there is a noticeable change in salinity between saltwater and freshwater sources. This is typically found where rivers meet the ocean or sea. The wildlife found within estuaries is unique as the water in these areas is brackish - a mix of freshwater flowing to the ocean and salty seawater. [15] Other types of estuaries also exist and have similar characteristics as traditional brackish estuaries. The Great Lakes are a prime example. There, river water mixes with lake water and creates freshwater estuaries. [15] Estuaries are extremely productive ecosystems that many humans and animal species rely on for various activities. [16] This can be seen as, of the 32 largest cities in the world, 22 are located on estuaries as they provide many environmental and economic benefits such as crucial habitat for many species, and being economic hubs for many coastal communities. [16] Estuaries also provide essential ecosystem services such as water filtration, habitat protection, erosion control, gas regulation nutrient cycling, and it even gives education, recreation and tourism opportunities to people. [17]

Lagoons
Lagoons are areas that are separated from larger water by natural barriers such as coral reefs or sandbars. There are two types of lagoons, coastal and oceanic/atoll lagoons. [18] A coastal lagoon is, as the definition above, simply a body of water that is separated from the ocean by a barrier. An atoll lagoon is a circular coral reef or several coral islands that surround a lagoon. Atoll lagoons are often much deeper than coastal lagoons. [19] Most lagoons are very shallow meaning that they are greatly affected by changed in precipitation, evaporation and wind. This means that salinity and temperature are widely varied in lagoons and that they can have water that ranges from fresh to hypersaline. [19] Lagoons can be found in on coasts all over the world, on every continent except Antarctica and is an extremely diverse habitat being home to a wide array of species including birds, fish, crabs, plankton and more. [19] Lagoons are also important to the economy as they provide a wide array of ecosystem services in addition to being the home of so many different species. Some of these services include fisheries, nutrient cycling, flood protection, water filtration, and even human tradition. [19]
Reefs
Coral reefs
Coral reefs are one of the most well-known marine ecosystems in the world, with the largest being the Great Barrier Reef. These reefs are composed of large coral colonies of a variety of species living together. The corals from multiple symbiotic relationships with the organisms around them. [20] Coral reefs are being heavily affected by global warming. They are one of the most vulnerable marine ecosystems. Due to marine heatwaves that have high warming levels coral reefs are at risk of a great decline, loss of its important structures, and exposure to higher frequency of marine heatwaves. [21]
-
Coral reef
-
Global distribution of coral, mangrove, and seagrass diversity
Bivalve reefs

Bivalve reefs provide coastal protection through erosion control and shoreline stabilization, and modify the physical landscape by ecosystem engineering, thereby providing habitat for species by facilitative interactions with other habitats such as tidal flat benthic communities, seagrasses and marshes. [23]
Vegetated
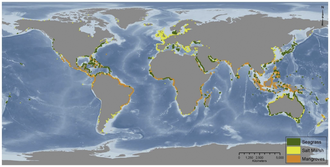
Vegetated coastal ecosystems occur throughout the world, as illustrated in the diagram on the right. Seagrass beds are found from cold polar waters to the tropics. Mangrove forests are confined to tropical and sub-tropical areas, while tidal marshes are found in all regions, but most commonly in temperate areas. Combined, these ecosystems cover about 50 million hectares and provide a diverse array of ecosystem services such as fishery production, coastline protection, pollution buffering, as well as high rates of carbon sequestration. [25] [24]
Rapid loss of vegetated coastal ecosystems through land-use change has occurred for centuries, and has accelerated in recent decades. Causes of habitat conversion vary globally and include conversion to aquaculture, agriculture, forest over-exploitation, industrial use, upstream dams, dredging, eutrophication of overlying waters, urban development, and conversion to open water due to accelerated sea-level rise and subsidence. [25] [24]
Vegetated coastal ecosystems typically reside over organic-rich sediments that may be several meters deep and effectively lock up carbon due to low-oxygen conditions and other factors that inhibit decomposition at depth. [26] These carbon stocks can exceed those of terrestrial ecosystems, including forests, by several times. [27] [28] When coastal habitats are degraded or converted to other land uses, the sediment carbon is destabilised or exposed to oxygen, and subsequent increased microbial activity releases large amounts of greenhouse gasses to the atmosphere or water column. [29] [26] [30] [31] [32] [33] The potential economic impacts that come from releasing stored coastal blue carbon to the atmosphere are felt worldwide. Economic impacts of greenhouse gas emissions in general stem from associated increases in droughts, sea level, and frequency of extreme weather events. [34] [24]
-
Conceptual diagram of estuarine vegetation
Coastal wetlands

Coastal wetlands are among the most productive ecosystems on Earth and generate vital services that benefit human societies around the world. Sediment-stabilization by wetlands such as salt marshes and mangroves serves to protect coastal communities from storm-waves, flooding, and land erosion. [39] Coastal wetlands also reduce pollution from human waste, [40] [41] remove excess nutrients from the water column, [42] trap pollutants, [43] and sequester carbon. [44] Further, near-shore wetlands act as both essential nursery habitats and feeding grounds for game fish, supporting a diverse group of economically important species. [45] [46] [47] [48] [49]
Mangrove forests
Mangroves are trees or shrubs that grow in low-oxygen soil near coastlines in tropical or subtropical latitudes. [50] They are an extremely productive and complex ecosystem that connects the land and sea. Mangroves consist of species that are not necessarily related to each other and are often grouped for the characteristics they share rather than genetic similarity. [51] Because of their proximity to the coast, they have all developed adaptions such as salt excretion and root aeration to live in salty, oxygen-depleted water. [51] Mangroves can often be recognized by their dense tangle of roots that act to protect the coast by reducing erosion from storm surges, currents, wave, and tides. [50] The mangrove ecosystem is also an important source of food for many species as well as excellent at sequestering carbon dioxide from the atmosphere with global mangrove carbon storage is estimated at 34 million metric tons per year. [51]
Salt marshes
Salt marshes are a transition from the ocean to the land, where fresh and saltwater mix. [52] The soil in these marshes is often made up of mud and a layer of organic material called peat. Peat is characterized as waterlogged and root-filled decomposing plant matter that often causes low oxygen levels (hypoxia). These hypoxic conditions causes growth of the bacteria that also gives salt marshes the sulfurous smell they are often known for. [53] Salt marshes exist around the world and are needed for healthy ecosystems and a healthy economy. They are extremely productive ecosystems and they provide essential services for more than 75 percent of fishery species and protect shorelines from erosion and flooding. [53] Salt marshes can be generally divided into the high marsh, low marsh, and the upland border. The low marsh is closer to the ocean, with it being flooded at nearly every tide except low tide. [52] The high marsh is located between the low marsh and the upland border and it usually only flooded when higher than usual tides are present. [52] The upland border is the freshwater edge of the marsh and is usually located at elevations slightly higher than the high marsh. This region is usually only flooded under extreme weather conditions and experiences much less waterlogged conditions and salt stress than other areas of the marsh. [52]
Seagrass meadows
Seagrasses form dense underwater meadows which are among the most productive ecosystems in the world. They provide habitats and food for a diversity of marine life comparable to coral reefs. This includes invertebrates like shrimp and crabs, cod and flatfish, marine mammals and birds. They provide refuges for endangered species such as seahorses, turtles, and dugongs. They function as nursery habitats for shrimps, scallops and many commercial fish species. Seagrass meadows provide coastal storm protection by the way their leaves absorb energy from waves as they hit the coast. They keep coastal waters healthy by absorbing bacteria and nutrients, and slow the speed of climate change by sequestering carbon dioxide into the sediment of the ocean floor.
Seagrasses evolved from marine algae which colonized land and became land plants, and then returned to the ocean about 100 million years ago. However, today seagrass meadows are being damaged by human activities such as pollution from land runoff, fishing boats that drag dredges or trawls across the meadows uprooting the grass, and overfishing which unbalances the ecosystem. Seagrass meadows are currently being destroyed at a rate of about two football fields every hour.
-
Seagrass meadow
-
Kelp forest
| External videos | |
|---|---|
Kelp forests
Kelp forests occur worldwide throughout temperate and polar coastal oceans. [54] In 2007, kelp forests were also discovered in tropical waters near Ecuador. [55]
Physically formed by brown macroalgae, kelp forests provide a unique habitat for marine organisms [56] and are a source for understanding many ecological processes. Over the last century, they have been the focus of extensive research, particularly in trophic ecology, and continue to provoke important ideas that are relevant beyond this unique ecosystem. For example, kelp forests can influence coastal oceanographic patterns [57] and provide many ecosystem services. [58]
However, the influence of humans has often contributed to kelp forest degradation. Of particular concern are the effects of overfishing nearshore ecosystems, which can release herbivores from their normal population regulation and result in the overgrazing of kelp and other algae. [59] This can rapidly result in transitions to barren landscapes where relatively few species persist. [60] [61] Already due to the combined effects of overfishing and climate change, kelp forests have all but disappeared in many especially vulnerable places, such as Tasmania's east coast and the coast of Northern California. [62] [63] The implementation of marine protected areas is one management strategy useful for addressing such issues, since it may limit the impacts of fishing and buffer the ecosystem from additive effects of other environmental stressors.
Coastal ecology
Coastal food webs
| External videos | |
|---|---|
– David Attenborough, full episode, Netflix |
Coastal waters include the waters in estuaries and over continental shelves. They occupy about 8 percent of the total ocean area [64] and account for about half of all the ocean productivity. The key nutrients determining eutrophication are nitrogen in coastal waters and phosphorus in lakes. Both are found in high concentrations in guano (seabird feces), which acts as a fertilizer for the surrounding ocean or an adjacent lake. Uric acid is the dominant nitrogen compound, and during its mineralization different nitrogen forms are produced. [65]
Ecosystems, even those with seemingly distinct borders, rarely function independently of other adjacent systems. [66] Ecologists are increasingly recognizing the important effects that cross-ecosystem transport of energy and nutrients have on plant and animal populations and communities. [67] [68] A well known example of this is how seabirds concentrate marine-derived nutrients on breeding islands in the form of feces (guano) which contains ~15–20% nitrogen (N), as well as 10% phosphorus. [69] [70] [71] These nutrients dramatically alter terrestrial ecosystem functioning and dynamics and can support increased primary and secondary productivity. [72] [73] However, although many studies have demonstrated nitrogen enrichment of terrestrial components due to guano deposition across various taxonomic groups, [72] [74] [75] [76] only a few have studied its retroaction on marine ecosystems and most of these studies were restricted to temperate regions and high nutrient waters. [69] [77] [78] [79] In the tropics, coral reefs can be found adjacent to islands with large populations of breeding seabirds, and could be potentially affected by local nutrient enrichment due to the transport of seabird-derived nutrients in surrounding waters. Studies on the influence of guano on tropical marine ecosystems suggest nitrogen from guano enriches seawater and reef primary producers. [77] [80] [81]
Reef building corals have essential nitrogen needs and, thriving in nutrient-poor tropical waters [82] where nitrogen is a major limiting nutrient for primary productivity, [83] they have developed specific adaptations for conserving this element. Their establishment and maintenance are partly due to their symbiosis with unicellular dinoflagellates, Symbiodinium spp. (zooxanthellae), that can take up and retain dissolved inorganic nitrogen (ammonium and nitrate) from the surrounding waters. [84] [85] [86] These zooxanthellae can also recycle the animal wastes and subsequently transfer them back to the coral host as amino acids, [87] ammonium or urea. [88] Corals are also able to ingest nitrogen-rich sediment particles [89] [90] and plankton. [91] [92] Coastal eutrophication and excess nutrient supply can have strong impacts on corals, leading to a decrease in skeletal growth, [85] [93] [94] [95] [81]
-
Pathways for guano-derived nitrogen to enter marine food webs [81]
-
Seabird colonies are nutrient hot spots, especially, for nitrogen and phosphorus [65]
Coastal predators

Food web theory predicts that current global declines in marine predators could generate unwanted consequences for many marine ecosystems. In coastal plant communities, such as kelp, seagrass meadows, mangrove forests and salt marshes, several studies have documented the far-reaching effects of changing predator populations. Across coastal ecosystems, the loss of marine predators appears to negatively affect coastal plant communities and the ecosystem services they provide. [96]
The green world hypothesis predicts loss of predator control on herbivores could result in runaway consumption that would eventually denude a landscape or seascape of vegetation. [97] Since the inception of the green world hypothesis, ecologists have tried to understand the prevalence of indirect and alternating effects of predators on lower trophic levels ( trophic cascades), and their overall impact on ecosystems. [98] Multiple lines of evidence now suggest that top predators are key to the persistence of some ecosystems. [98] [96]
With an estimated habitat loss greater than 50 percent, coastal plant communities are among the world’s most endangered ecosystems. [99] [100] [101] As bleak as this number is, the predators that patrol coastal systems have fared far worse. Several predatory taxa including species of marine mammals, elasmobranchs, and seabirds have declined by 90 to 100 percent compared to historical populations. [10] [102] Predator declines pre-date habitat declines, [10] suggesting alterations to predator populations may be a major driver of change for coastal systems. [103] [104] [96]
There is little doubt that collapsing marine predator populations results from overharvesting by humans. Localized declines and extinctions of coastal predators by humans began over 40,000 years ago with subsistence harvesting. [105] However, for most large bodied, marine predators ( toothed whales, large pelagic fish, sea birds, pinnipeds, and otters) the beginning of their sharp global declines occurred over the last century, coinciding with the expansion of coastal human populations and advances in industrial fishing. [10] [106] Following global declines in marine predators, evidence of trophic cascades in coastal ecosystems started to emerge, [107] [108] [109] [110] with the disturbing realisation that they affected more than just populations of lower trophic levels. [98] [96]
Understanding the importance of predators in coastal plant communities has been bolstered by their documented ability to influence ecosystem services. Multiple examples have shown that changes to the strength or direction of predator effects on lower trophic levels can influence coastal erosion, [111] carbon sequestration, [112] [113] and ecosystem resilience. [114] The idea that the extirpation of predators can have far-reaching effects on the persistence of coastal plants and their ecosystem services has become a major motivation for their conservation in coastal systems. [98] [113] [96]
Seascape ecology

Seascape ecology is the marine and coastal version of landscape ecology. [117] It is currently emerging as an interdisciplinary and spatially explicit ecological science with relevance to marine management, biodiversity conservation, and restoration. [116] Seascapes are complex ocean spaces, shaped by dynamic and interconnected patterns and processes operating across a range of spatial and temporal scales. [118] [119] [120] Rapid advances in geospatial technologies and the proliferation of sensors, both above and below the ocean surface, have revealed intricate and scientifically intriguing ecological patterns and processes, [121] [122] [123] some of which are the result of human activities. [124] [125] Despite progress in the collecting, mapping, and sharing of ocean data, the gap between technological advances and the ability to generate ecological insights for marine management and conservation practice remains substantial. [126] [127] For instance, fundamental gaps exist in the understanding of multidimensional spatial structure in the sea, [123] [120] [128] and the implications for planetary health and human wellbeing. [127] Deeper understanding of the multi-scale linkages between ecological structure, function, and change will better support the design of whole-system strategies for biodiversity preservation and reduce uncertainty around the consequences of human activity. For example, in the design and evaluation of marine protected areas (MPAs) and habitat restoration, it is important to understand the influence of spatial context, configuration, and connectivity, and to consider effects of scale. [129] [130] [131] [132] [116]
-
Fish migrations between coral reef, macroalgae, seagrass and mangrove habitats: [133] (a) diel and tidal foraging migrations, (b) ontogenetic migration of juvenile coral reef fish.
-
The ecosystem cascade to structure the stock-taking and scenario analysis steps in the marine spatial planning process. [134]
Interactions between ecosystems

The diagram on the right shows the principal interactions between mangroves, seagrass, and coral reefs. [135] Coral reefs, seagrasses, and mangroves buffer habitats further inland from storms and wave damage as well as participate in a tri-system exchange of mobile fish and invertebrates. Mangroves and seagrasses are critical in regulating sediment, freshwater, and nutrient flows to coral reefs. [135]
The diagram immediately below shows locations where mangroves, coral reefs, and seagrass beds exist within one km of each other. Buffered intersection between the three systems provides relative co-occurrence rates on a global scale. Regions where systems strongly intersect include Central America (Belize), the Caribbean, the Red Sea, the Coral Triangle (particularly Malaysia), Madagascar, and the Great Barrier Reef. [135]

The diagram at the right graphically illustrates the ecosystem service synergies between mangroves, seagrasses, and coral reefs. The ecosystem services provided by intact reefs, seagrasses, and mangroves are both highly valuable and mutually enhance each other. Coastal protection (storm/wave attenuation) maintains the structure of adjacent ecosystems, and associated ecosystem services, in an offshore-to-onshore direction. Fisheries are characterized by migratory species, and therefore, protecting fisheries in one ecosystem increases fish biomass in others. Tourism benefits from coastal protection and healthy fisheries from multiple ecosystems. Here, we do not draw within-ecosystem connections in order to better emphasise synergies between systems. [135]
Network ecology
To compound things, removal of biomass from the ocean occurs simultaneously with multiple other stressors associated to climate change that compromise the capacity of these socio-ecological systems to respond to perturbations. [137] [138] [139] Besides sea surface temperature, climate change also affects many other physical–chemical characteristics of marine coastal waters (stratification, acidification, ventilation) [140] [141] as well as the wind regimes that control surface water productivity along the productive coastal upwelling ecosystems. [142] [143] [144] [145] [146] Changes in the productivity of the oceans are reflected in changes of plankton biomass. Plankton contributes approximately half of the global primary production, supports marine food webs, influences the biogeochemical process in the ocean, and strongly affects commercial fisheries. [147] [148] [149] Indeed, an overall decrease in marine plankton productivity is expected over global scales. [141] [147] [150] Long-term increases and decreases in plankton productivity have already occurred over the past two decades [151] [152] along extensive regions of the Humboldt upwelling ecosystem off Chile, and are expected to propagate up the pelagic and benthic food webs. [136]
Network ecology has advanced understanding of ecosystems by providing a powerful framework to analyse biological communities. [153] Previous studies used this framework to assess food web robustness against species extinctions, defined as the fraction of initial species that remain present in the ecosystem after a primary extinction. [154] [155] [156] [157] [158] [159] [160] [161] These studies showed the importance for food web persistence of highly connected species (independent of trophic position), [154] [157] [162] basal species, [155] and highly connected species that, at the same time, trophically support other highly connected species. [158] Most of these studies used a static approach, which stems from network theory and analyzes the impacts of structural changes on food webs represented by nodes (species) and links (interactions) that connect nodes, but ignores interaction strengths and population dynamics of interacting species. [154] Other studies used a dynamic approach, which considers not only the structure and intensity of interactions in a food web, but also the changes in species biomasses through time and the indirect effects that these changes have on other species. [155] [156] [163] [164] [165] [136]
Coastal biogeochemistry


Globally, eutrophication is one of the major environmental problems in coastal ecosystems. Over the last century the annual riverine inputs of nitrogen and phosphorus to the oceans have increased from 19 to 37 megatonnes of nitrogen and from 2 to 4 megatonnes of phosphorus. [166] Regionally, these increases were even more substantial as observed in the United States, Europe and China. In the Baltic Sea nitrogen and phosphorus loads increased by roughly a factor of three and six, respectively. [167] The riverine nitrogen flux has increased by an order of magnitude to coastal waters of China within thirty years, while phosphorus export has tripled between 1970 and 2000. [168] [169] [1]
Efforts to mitigate eutrophication through nutrient load reductions are hampered by the effects of climate change. [9] Changes in precipitation increase the runoff of N, P and carbon (C) from land, which together with warming and increased CO2 dissolution alter the coupled marine nutrient and carbon cycles. [170] [171] [1]
In contrast to the open ocean where biogeochemical cycling is largely dominated by pelagic processes driven primarily by ocean circulation, in the coastal zone, pelagic and benthic processes interact strongly and are driven by a complex and dynamic physical environment. [172] Eutrophication in coastal areas leads to shifts toward rapidly growing opportunistic algae, and generally to a decline in benthic macrovegetation because of decreased light penetration, substrate change and more reducing sediments. [173] [174] Increased production and warming waters have caused expanding hypoxia at the seafloor with a consequent loss of benthic fauna. [175] [176] Hypoxic systems tend to lose many long-lived higher organisms and biogeochemical cycles typically become dominated by benthic bacterial processes and rapid pelagic turnover. [177] However, if hypoxia does not occur, benthic fauna tends to increase in biomass with eutrophication. [178] [179] [180] [1]
Changes in benthic biota have far-reaching impacts on biogeochemical cycles in the coastal zone and beyond. In the illuminated zone, benthic microphytes and macrophytes mediate biogeochemical fluxes through primary production, nutrient storage and sediment stabilization and act as a habitat and food source for a variety of animals, as shown in the diagram on the left above. Benthic animals contribute to biogeochemical transformations and fluxes between water and sediments both directly through their metabolism and indirectly by physically reworking the sediments and their porewaters and stimulating bacterial processes. Grazing on pelagic organic matter and biodeposition of feces and pseudofeces by suspension-feeding fauna increases organic matter sedimentation rates. [181] [182] In addition, nutrients and carbon are retained in biomass and transformed from organic to inorganic forms through metabolic processes. [183] [180] [184] Bioturbation, including sediment reworking and burrow ventilation activities ( bioirrigation), redistributes particles and solutes within the sediment and enhances sediment-water fluxes of solutes. [185] [186] Bioturbation can also enhance resuspension of particles, a phenomenon termed "bioresuspension". [187] Together, all these processes affect physical and chemical conditions at the sediment-water interface, [188] and strongly influence organic matter degradation. [189] When up-scaled to the ecosystem level, such modified conditions can significantly alter the functioning of coastal ecosystems and ultimately, the role of the coastal zone in filtering and transforming nutrients and carbon. [1]
Artisan fisheries

Artisanal fisheries use simple fishing gears and small vessels. [136] Their activities tend to be confined to coastal areas. In general, top-down and bottom-up forces determine ecosystem functioning and dynamics. Fisheries as a top-down force can shorten and destabilise food webs, while effects driven by climate change can alter the bottom-up forces of primary productivity. [136]
Direct human impacts and the full suite of drivers of global change are the main cause of species extinctions in Anthropocene ecosystems, [190] [105] with detrimental consequences on ecosystem functioning and their services to human societies. [191] [192] The world fisheries crisis is among those consequences, which cuts across fishing strategies, oceanic regions, species, and includes countries that have little regulation and those that have implemented rights-based co-management strategies to reduce overharvesting. [193] [194] [195] [196] [136]
Chile has been one of the countries implementing Territorial Use Rights (TURFs) [197] [198] over an unprecedented geographic scale to manage the diverse coastal benthic resources using a co-management strategy. [199] [200] These TURFS are used for artisanal fisheries. Over 60 coastal benthic species are actively harvested by these artisanal fisheries, [201] with species that are extracted from intertidal and shallow subtidal habitats. [202] [203] The Chilean TURFs system brought significant improvements in sustainability of this complex socio-ecological system, helping to rebuild benthic fish stocks, [201] [199] improving fishers’ perception towards sustainability and increasing compliance9, as well as showing positive ancillary effects on conservation of biodiversity. [204] [205] However, the situation of most artisanal fisheries is still far from sustainable, and many fish stocks and coastal ecosystems show signs of overexploitation and ecosystem degradation, a consequence of the low levels of cooperation and low enforcement of TURF regulations, which leads to high levels of free-riding and illegal fishing. [206] [207] [208] It is imperative to improve understanding of the effects of these multi-species artisanal fisheries which simultaneously harvest species at all trophic levels from kelp primary producers to top carnivores. [203] [209] [136]
Remote sensing
Coastal zones are among the most populated areas on the planet. [212] [213] As the population continues to increase, economic development must expand to support human welfare. However, this development may damage the ability of the coastal environment to continue supporting human welfare for current and future generations. [214] The management of complex coastal and marine social-ecological systems requires tools that provide frameworks with the capability of responding to current and emergent issues. [215] [210] Remote data collection technologies include satellite-based remote sensing, aerial remote sensing, unmanned aerial vehicles, unmanned surface vehicles, unmanned underwater vehicles, and static sensors. [210]
Frameworks have been developed that attempt to address and integrate these complex issues, such as the Millennium Ecosystem Assessment framework which links drivers, ecosystem services, and human welfare [216] [210] However, obtaining the environmental data that is necessary to use such frameworks is difficult, especially in countries where access to reliable data and their dissemination are limited or non-existent [217] and even thwarted. [210] Traditional techniques of point sampling and observation in the environment do deliver high information content, [218] but they are expensive and often do not provide adequate spatial and temporal coverage, while remote sensing can provide cost-effective solutions, as well as data for locations where there is no or only limited information. [219] [210]
Coastal observing systems are typically nationally funded and built around national priorities. As a result, there are presently significant differences between countries in terms of sustainability, observing capacity and technologies, as well as methods and research priorities. [211] Ocean observing systems in coastal areas need to move toward integrated, multidisciplinary and multiscale systems, where heterogeneity can be exploited to deliver fit-for-purpose answers. [211] Essential elements of such distributed observation systems are the use of machine-to-machine communication, data fusion and processing applying recent technological developments for the Internet of Things (IoT) toward a common cyberinfrastructure. [211] It has been argued that the standardisation that IoT brings to wireless sensing will revolutionise areas like this. [220]
Coastal areas are the most dynamic and productive parts of the oceans, which makes them a significant source of human resources and services. Coastal waters are located immediately in contact with human populations and exposed to anthropogenic disturbances, placing these resources and services under threat. [221] These concerns explain why, in several coastal regions, a rapidly increasing number of observing systems have been implemented in the last decade. [222] Expansion of coherent and sustained coastal observations has been fragmented and driven by national and regional policies and is often undertaken through short-term research projects. [223] This results in significant differences between countries both in terms of sustainability and observing technologies, methods and research priorities. [211]
Unlike the open ocean, where challenges are rather well-defined and stakeholders are fewer and well-identified, coastal processes are complex, acting on several spatial and temporal scales, with numerous and diversified users and stakeholders, often with conflicting interests. To adapt to such complexity coastal ocean observing system must be an integrated, multidisciplinary and multiscale system of systems. [224] [211]
Regime shifts

Marine ecosystems are affected by diverse pressures and consequently may undergo significant changes that can be interpreted as regime shifts. [225] Marine ecosystems worldwide are affected by increasing natural and anthropogenic pressures and consequently undergo significant changes at unprecedented rates. Affected by these changes, ecosystems can reorganise and still maintain the same function, structure, and identity. [226] However, under some circumstances, the ecosystem may undergo changes that modify the system’s structure and function and this process can be described as a shift to a new regime. [226] [227] [228] [225]
Usually, a regime shift is triggered by large-scale climate-induced variations, [229] intense fishing exploitation [230] or both. [231] Criteria used to define regime shifts vary and the changes that have to occur in order to consider that a system has undergone a regime shift are not well-defined. [232] Normally, regime shifts are defined as high amplitude, low-frequency and often abrupt changes in species abundance and community composition that are observed at multiple trophic levels (TLs). [233] These changes are expected to occur on a large spatial scale and take place concurrently with physical changes in the climate system. [233] [228] [234] [235] [236] [237] [232] [225]
Regime shifts have been described in several marine ecosystems including Northern Benguela, [238] the North Sea, [239] and the Baltic Sea. [240] In large upwelling ecosystems, it is common to observe decadal fluctuations in species abundance and their replacements. [241] These fluctuations might be irreversible and might be an indicator of the new regime, as was the case in the Northern Benguela ecosystem. [238] However, changes in the upwelling systems might be interpreted as fluctuations within the limits of natural variability for an ecosystem, and not as an indicator of the regime shift. [234] The Portuguese continental shelf ecosystem (PCSE) constitutes the northernmost part of the Canary Current Upwelling System and is characterised by seasonal upwelling that occurs during the spring and summer as a result of steady northerly winds. [242] [243] It has recently changed in the abundance of coastal pelagic species such as sardine, chub mackerel, horse mackerel, blue jack mackerel and anchovy. [244] [245] [246] [247] Moreover, in the last decades, an increase in higher trophic level species has been documented. [248] The causes underlying changes in the pelagic community are not clear but it has been suggested that they result from a complex interplay between environmental variability, species interactions and fishing pressure. [249] [250] [251] [225]
There is evidence, that changes in the intensity of the Iberian coastal upwelling (resulting from the strengthening or weakening northern winds) had occurred in the last decades. However, the character of these changes is contradictory where some authors observed intensification of upwelling-favourable winds [252] [253] while others documented their weakening. [254] [255] A 2019 review of upwelling rate and intensity along the Portuguese coast documented a successive weakening of the upwelling since 1950 that lasted till mid/late 1970s in the north-west and south-west and till 1994 in the south coast. [256] An increase in upwelling index over the period 1985–2009 was documented in all studied regions while additionally upwelling intensification were observed in the south. [256] A continuous increase in water temperature, ranging from 0.1 to 0.2 °C per decade has also been documented. [257] [225]
Threats and decline

Many marine fauna utilise coastal habitats as critical nursery areas, for shelter and feeding, yet these habitats are increasingly at risk from agriculture, aquaculture, industry and urban expansion. [258] Indeed, these systems are subject to what may be called "a triple whammy" of increasing industrialisation and urbanisation, an increased loss of biological and physical resources (fish, water, energy, space), and a decreased resilience to the consequences of a warming climate and sea level rise. [259] This has given rise to the complete loss, modification or disconnection of natural coastal ecosystems globally. For example, almost 10% of the entire Great Barrier Reef coastline in Australia (2,300 km) has been replaced with urban infrastructure (e.g., rock seawalls, jetties, marinas), causing massive loss and fragmentation of sensitive coastal ecosystems. [260] Global loss of seagrass reached around 7% of seagrasses area per year by the end of the twentieth century. [261] A global analysis of tidal wetlands ( mangroves, tidal flats, and tidal marshes) published in 2022 estimated global losses of 13,700 km2 (5,300 sq mi) from 1999-2019, however, this study also estimated that these losses were largely offset by the establishment of 9,700 km2 (3,700 sq mi) of new tidal wetlands that were not present in 1999. [262] Approximately three-quarters of the 4,000 km2 (1,500 sq mi) net decrease between 1999 and 2019 occurred in Asia (74.1%), with 68.6% concentrated in three countries: Indonesia (36%), China (20.6%), and Myanmar (12%). [262] Of these global tidal wetland losses and gains, 39% of losses and 14% of gains were attributed to direct human activities. [262]
Approximately 40% of the global mangrove has been lost since the 1950's [263] with more than 9,736 km2 of the world's mangroves continuing to be degraded in the 20 years period between 1996 and 2016. [264] Saltmarshes are drained when coastal land is claimed for agriculture, and deforestation is an increasing threat to shoreline vegetation (such as mangroves) when coastal land is appropriated for urban and industrial development, [263] both of which may result in the degradation of blue carbon storages and increasing greenhouse gas emissions. [265]
These accumulating pressures and impacts on coastal ecosystems are neither isolated nor independent, rather they are synergistic, with feedbacks and interactions that cause individual effects to be greater than their sums. [266] In the year before the ecosystem restoration Decade commences, there is a critical knowledge deficit inhibiting an appreciation of the complexity of coastal ecosystems that hampers the development of responses to mitigate continuing impacts—not to mention uncertainty on projected losses of coastal systems for some of the worst-case future climate change scenarios. [267]
Restoration

The United Nations has declared 2021–2030 the UN Decade on Ecosystem Restoration. This call to action has the purpose of recognising the need to massively accelerate global restoration of degraded ecosystems, to fight the climate heating crisis, enhance food security, provide clean water and protect biodiversity on the planet. The scale of restoration will be key. For example, the Bonn Challenge has the goal to restore 350 million km2, about the size of India, of degraded terrestrial ecosystems by 2030. However, international support for restoration of blue coastal ecosystems, which provide an impressive array of benefits to people, has lagged.
The diagram on the right shows the current state of modified and impacted coastal ecosystems and the expected state following the decade of restoration. [267] Also, shown is the uncertainty in the success of past restoration efforts, current state of altered systems, climate variability, and restoration actions that are available now or on the horizon. This could mean that delivering the Decade on Ecosystem Restoration for coastal systems needs to be viewed as a means of getting things going where the benefits might take longer than a decade. [267]
Only the Global Mangrove Alliance [268] comes close to the Bonn Challenge, with the aim of increasing the global area of mangroves by 20% by 2030. [267] However, mangrove scientists have reservations about this target, voicing concerns that it is unrealistic and may prompt inappropriate practices in attempting to reach this target. [269] [267]
Conservation and connectivity
There has recently been a perceptual shift away from habitat representation as the sole or primary focus of conservation prioritisation, towards consideration of ecological processes that shape the distribution and abundance of biodiversity features. [270] [271] [272] [273] In marine ecosystems, connectivity processes are paramount, [274] and designing systems of marine protected areas that maintain connectivity between habitat patches has long been considered an objective of conservation planning. [270] [275] Two forms of connectivity are critical to structuring coral reef fish populations: [276] dispersal of larvae in the pelagic environment, [277] and post-settlement migration by individuals across the seascape. [278] Whilst a growing literature has described approaches for considering larval connectivity in conservation prioritisation, [279] [280] [281] relatively less attention has been directed towards developing and applying methods for considering post-settlement connectivity [274] [282] [283]
Seascape connectivity (connectedness among different habitats in a seascape, c.f. among patches of the same habitat type [131] is essential for species that utilise more than one habitat, either during diurnal movements or at different stages in their life history. Mangroves, seagrass beds, and lagoon reefs provide nursery areas for many commercially and ecologically important fish species that subsequently make ontogenetic shifts to adult populations on coral reefs. [284] [285] [286] These back-reef habitats are often overlooked for conservation or management in favour of coral reefs that support greater adult biomass, yet they can be equally if not more at risk from habitat degradation and loss. [287] [46] [288] Even where juveniles are not targeted by fishers, they can be vulnerable to habitat degradation, for example from sedimentation caused by poor land-use practices. [289] [283]
There is clear empirical evidence that proximity to nursery habitats can enhance the effectiveness (i.e. increasing the abundance, density, or biomass of fish species) of marine protected areas on coral reefs. [131] [290] [291] [292] [293] For example, at study sites across the western Pacific, the abundance of harvested fish species was significantly greater on protected reefs close to mangroves, but not on protected reefs isolated from mangroves. [292] The functional role of herbivorous fish species that perform ontogenetic migrations may also enhance the resilience of coral reefs close to mangroves. [294] [295] Despite this evidence, and widespread calls to account for connectivity among habitats in the design of spatial management, [285] [292] [293] there remain few examples where seascape connectivity is explicitly considered in spatial conservation prioritisation (the analytical process of identifying priority areas for conservation or management actions). [283]
See also
References
- ^
a
b
c
d
e
f
g
h
i
j Ehrnsten, Eva; Sun, Xiaole; Humborg, Christoph; Norkko, Alf; Savchuk, Oleg P.; Slomp, Caroline P.; Timmermann, Karen; Gustafsson, Bo G. (2020).
"Understanding Environmental Changes in Temperate Coastal Seas: Linking Models of Benthic Fauna to Carbon and Nutrient Fluxes". Frontiers in Marine Science. 7.
doi:
10.3389/fmars.2020.00450.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^ Tappin, A.D. (2002). "An Examination of the Fluxes of Nitrogen and Phosphorus in Temperate and Tropical Estuaries: Current Estimates and Uncertainties". Estuarine, Coastal and Shelf Science. 55 (6): 885–901. Bibcode: 2002ECSS...55..885T. doi: 10.1006/ecss.2002.1034.
- ^ Laruelle, G. G.; Roubeix, V.; Sferratore, A.; Brodherr, B.; Ciuffa, D.; Conley, D. J.; Dürr, H. H.; Garnier, J.; Lancelot, C.; Le Thi Phuong, Q.; Meunier, J.-D.; Meybeck, M.; Michalopoulos, P.; Moriceau, B.; Ní Longphuirt, S.; Loucaides, S.; Papush, L.; Presti, M.; Ragueneau, O.; Regnier, P.; Saccone, L.; Slomp, C. P.; Spiteri, C.; Van Cappellen, P. (2009). "Anthropogenic perturbations of the silicon cycle at the global scale: Key role of the land-ocean transition". Global Biogeochemical Cycles. 23 (4): n/a. Bibcode: 2009GBioC..23.4031L. doi: 10.1029/2008GB003267. S2CID 130818402.
- ^ Regnier, Pierre; Friedlingstein, Pierre; Ciais, Philippe; MacKenzie, Fred T.; Gruber, Nicolas; Janssens, Ivan A.; Laruelle, Goulven G.; Lauerwald, Ronny; Luyssaert, Sebastiaan; Andersson, Andreas J.; Arndt, Sandra; Arnosti, Carol; Borges, Alberto V.; Dale, Andrew W.; Gallego-Sala, Angela; Goddéris, Yves; Goossens, Nicolas; Hartmann, Jens; Heinze, Christoph; Ilyina, Tatiana; Joos, Fortunat; Larowe, Douglas E.; Leifeld, Jens; Meysman, Filip J. R.; Munhoven, Guy; Raymond, Peter A.; Spahni, Renato; Suntharalingam, Parvadha; Thullner, Martin (2013). "Anthropogenic perturbation of the carbon fluxes from land to ocean". Nature Geoscience. 6 (8): 597–607. Bibcode: 2013NatGe...6..597R. doi: 10.1038/ngeo1830. hdl: 10871/18939. S2CID 53418968.
- ^ Ramesh, R.; Chen, Z.; Cummins, V.; Day, J.; d'Elia, C.; Dennison, B.; Forbes, D.L.; Glaeser, B.; Glaser, M.; Glavovic, B.; Kremer, H.; Lange, M.; Larsen, J.N.; Le Tissier, M.; Newton, A.; Pelling, M.; Purvaja, R.; Wolanski, E. (2015). "Land–Ocean Interactions in the Coastal Zone: Past, present & future". Anthropocene. 12: 85–98. Bibcode: 2015Anthr..12...85R. doi: 10.1016/j.ancene.2016.01.005. hdl: 20.500.11815/1762.
- ^ Costanza, Robert; d'Arge, Ralph; De Groot, Rudolf; Farber, Stephen; Grasso, Monica; Hannon, Bruce; Limburg, Karin; Naeem, Shahid; O'Neill, Robert V.; Paruelo, Jose; Raskin, Robert G.; Sutton, Paul; Van Den Belt, Marjan (1997). "The value of the world's ecosystem services and natural capital". Nature. 387 (6630): 253–260. Bibcode: 1997Natur.387..253C. doi: 10.1038/387253a0. S2CID 672256.
- ^ Costanza, Robert; De Groot, Rudolf; Sutton, Paul; Van Der Ploeg, Sander; Anderson, Sharolyn J.; Kubiszewski, Ida; Farber, Stephen; Turner, R. Kerry (2014). "Changes in the global value of ecosystem services". Global Environmental Change. 26: 152–158. doi: 10.1016/j.gloenvcha.2014.04.002. S2CID 15215236.
- ^ Halpern, Benjamin S.; Walbridge, Shaun; Selkoe, Kimberly A.; Kappel, Carrie V.; Micheli, Fiorenza; d'Agrosa, Caterina; Bruno, John F.; Casey, Kenneth S.; Ebert, Colin; Fox, Helen E.; Fujita, Rod; Heinemann, Dennis; Lenihan, Hunter S.; Madin, Elizabeth M. P.; Perry, Matthew T.; Selig, Elizabeth R.; Spalding, Mark; Steneck, Robert; Watson, Reg (2008). "A Global Map of Human Impact on Marine Ecosystems". Science. 319 (5865): 948–952. Bibcode: 2008Sci...319..948H. doi: 10.1126/science.1149345. PMID 18276889. S2CID 26206024.
- ^ a b Cloern, James E.; Abreu, Paulo C.; Carstensen, Jacob; Chauvaud, Laurent; Elmgren, Ragnar; Grall, Jacques; Greening, Holly; Johansson, John Olov Roger; Kahru, Mati; Sherwood, Edward T.; Xu, Jie; Yin, Kedong (2016). "Human activities and climate variability drive fast-paced change across the world's estuarine-coastal ecosystems". Global Change Biology. 22 (2): 513–529. Bibcode: 2016GCBio..22..513C. doi: 10.1111/gcb.13059. PMID 26242490. S2CID 35848588.
- ^ a b c d Lotze, Heike K.; Lenihan, Hunter S.; Bourque, Bruce J.; Bradbury, Roger H.; Cooke, Richard G.; Kay, Matthew C.; Kidwell, Susan M.; Kirby, Michael X.; Peterson, Charles H.; Jackson, Jeremy B. C. (2006). "Depletion, Degradation, and Recovery Potential of Estuaries and Coastal Seas". Science. 312 (5781): 1806–1809. Bibcode: 2006Sci...312.1806L. doi: 10.1126/science.1128035. PMID 16794081. S2CID 12703389.
- ^ Kemp, W. M.; Testa, J. M.; Conley, D. J.; Gilbert, D.; Hagy, J. D. (2009). "Temporal responses of coastal hypoxia to nutrient loading and physical controls". Biogeosciences. 6 (12): 2985–3008. Bibcode: 2009BGeo....6.2985K. doi: 10.5194/bg-6-2985-2009.
- ^ United Nations (2017) Resolution adopted by the General Assembly on 6 July 2017, Work of the Statistical Commission pertaining to the 2030 Agenda for Sustainable Development ( A/RES/71/313)
- ^ Waltham, Nathan J.; Elliott, Michael; Lee, Shing Yip; Lovelock, Catherine; Duarte, Carlos M.; Buelow, Christina; Simenstad, Charles; Nagelkerken, Ivan; Claassens, Louw; Wen, Colin K-C; Barletta, Mario (2020). "UN Decade on Ecosystem Restoration 2021–2030—What Chance for Success in Restoring Coastal Ecosystems?". Frontiers in Marine Science. 7: 71. doi: 10.3389/fmars.2020.00071. hdl: 2440/123896. ISSN 2296-7745.
- ^ a b c d e US Department of Commerce, National Oceanic and Atmospheric Administration. "What is the intertidal zone?". oceanservice.noaa.gov. Retrieved 2019-03-21.
- ^ a b US Department of Commerce, National Oceanic and Atmospheric Administration. "What is an estuary?". oceanservice.noaa.gov. Retrieved 2019-03-22.
- ^ a b US Department of Commerce, National Oceanic and Atmospheric Administration. "Estuaries, NOS Education Offering". oceanservice.noaa.gov. Retrieved 2019-03-22.
- ^ "Estuaries". www.crd.bc.ca. 2013-11-14. Retrieved 2019-03-24.
- ^ US Department of Commerce, National Oceanic and Atmospheric Administration. "What is a lagoon?". oceanservice.noaa.gov. Retrieved 2019-03-24.
- ^ a b c d Miththapala, Sriyanie (2013). "Lagoons and Estuaries" (PDF). IUCN, International Union for Conservation of Nature. Archived from the original (PDF) on 2016-11-23. Retrieved 2021-10-31.
- ^ "Corals and Coral Reefs". Ocean Portal | Smithsonian. 2012-09-12. Retrieved 2018-03-27.
- ^ IPCC. Chapter 3: Oceans and Coastal Ecosystems and their Services. IPCC Sixth Assessment Report. 1 October 2021. https://report.ipcc.ch/ar6wg2/pdf/IPCC_AR6_WGII_FinalDraft_Chapter03.pdf.
- ^ Harman, Amanda (1997). Manatees & dugongs (in Lithuanian). New York: Benchmark Books. p. 7. ISBN 0-7614-0294-2. OCLC 34319364.
- ^ Ysebaert T., Walles B., Haner J., Hancock B. (2019) "Habitat Modification and Coastal Protection by Ecosystem-Engineering Reef-Building Bivalves". In: Smaal A., Ferreira J., Grant J., Petersen J., Strand Ø. (eds) Goods and Services of Marine Bivalves. Springer. doi: 10.1007/978-3-319-96776-9_13
- ^
a
b
c
d Pendleton, Linwood; Donato, Daniel C.; Murray, Brian C.; Crooks, Stephen; Jenkins, W. Aaron; Sifleet, Samantha; Craft, Christopher; Fourqurean, James W.; Kauffman, J. Boone; Marbà, Núria; Megonigal, Patrick; Pidgeon, Emily; Herr, Dorothee; Gordon, David; Baldera, Alexis (2012).
"Estimating Global "Blue Carbon" Emissions from Conversion and Degradation of Vegetated Coastal Ecosystems". PLOS ONE. 7 (9): e43542.
Bibcode:
2012PLoSO...743542P.
doi:
10.1371/journal.pone.0043542.
PMC
3433453.
PMID
22962585.
 Material was copied from this source, which is available under a
Creative Commons Attribution 0.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 0.0 International License.
- ^ a b Pendleton, Linwood; Donato, Daniel C.; Murray, Brian C.; Crooks, Stephen; Jenkins, W. Aaron; Sifleet, Samantha; Craft, Christopher; Fourqurean, James W.; Kauffman, J. Boone; Marbà, Núria; Megonigal, Patrick; Pidgeon, Emily; Herr, Dorothee; Gordon, David; Baldera, Alexis (2012). "Estimating Global "Blue Carbon" Emissions from Conversion and Degradation of Vegetated Coastal Ecosystems". PLOS ONE. 7 (9): e43542. Bibcode: 2012PLoSO...743542P. doi: 10.1371/journal.pone.0043542. PMC 3433453. PMID 22962585.
- ^ a b Kristensen, Erik; Bouillon, Steven; Dittmar, Thorsten; Marchand, Cyril (2008). "Organic carbon dynamics in mangrove ecosystems: A review". Aquatic Botany. 89 (2): 201–219. doi: 10.1016/j.aquabot.2007.12.005.
- ^ Donato, Daniel C.; Kauffman, J. Boone; Murdiyarso, Daniel; Kurnianto, Sofyan; Stidham, Melanie; Kanninen, Markku (2011). "Mangroves among the most carbon-rich forests in the tropics". Nature Geoscience. 4 (5): 293–297. Bibcode: 2011NatGe...4..293D. doi: 10.1038/ngeo1123.
- ^ Donato, D.C.; Kauffman, J.B.; MacKenzie, R.A.; Ainsworth, A.; Pfleeger, A.Z. (2012). "Whole-island carbon stocks in the tropical Pacific: Implications for mangrove conservation and upland restoration". Journal of Environmental Management. 97: 89–96. doi: 10.1016/j.jenvman.2011.12.004. PMID 22325586.
- ^ Eong, Ong Jin (1993). "Mangroves - a carbon source and sink". Chemosphere. 27 (6): 1097–1107. Bibcode: 1993Chmsp..27.1097E. doi: 10.1016/0045-6535(93)90070-L.
- ^ Granek, Elise; Ruttenberg, Benjamin I. (2008). "Changes in biotic and abiotic processes following mangrove clearing". Estuarine, Coastal and Shelf Science. 80 (4): 555–562. Bibcode: 2008ECSS...80..555G. doi: 10.1016/j.ecss.2008.09.012.
- ^ Sjöling, Sara; Mohammed, Salim M.; Lyimo, Thomas J.; Kyaruzi, Jasson J. (2005). "Benthic bacterial diversity and nutrient processes in mangroves: Impact of deforestation". Estuarine, Coastal and Shelf Science. 63 (3): 397–406. Bibcode: 2005ECSS...63..397S. doi: 10.1016/j.ecss.2004.12.002.
- ^ Strangmann, Antje; Bashan, Yoav; Giani, Luise (2008). "Methane in pristine and impaired mangrove soils and its possible effect on establishment of mangrove seedlings". Biology and Fertility of Soils. 44 (3): 511–519. doi: 10.1007/s00374-007-0233-7. S2CID 18477012.
- ^ Sweetman, A. K.; Middelburg, J. J.; Berle, A. M.; Bernardino, A. F.; Schander, C.; Demopoulos, A. W. J.; Smith, C. R. (2010). "Impacts of exotic mangrove forests and mangrove deforestation on carbon remineralization and ecosystem functioning in marine sediments". Biogeosciences. 7 (7): 2129–2145. Bibcode: 2010BGeo....7.2129S. doi: 10.5194/bg-7-2129-2010.
- ^ Lovelock, Catherine E.; Ruess, Roger W.; Feller, Ilka C. (2011). "CO2 Efflux from Cleared Mangrove Peat". PLOS ONE. 6 (6): e21279. Bibcode: 2011PLoSO...621279L. doi: 10.1371/journal.pone.0021279. PMC 3126811. PMID 21738628.
- ^
a
b Gaylard, Sam; Waycott, Michelle; Lavery, Paul (19 June 2020).
"Review of Coast and Marine Ecosystems in Temperate Australia Demonstrates a Wealth of Ecosystem Services". Frontiers in Marine Science. 7. Frontiers Media SA.
doi:
10.3389/fmars.2020.00453.
ISSN
2296-7745.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^ d'Odorico, Paolo; He, Yufei; Collins, Scott; De Wekker, Stephan F. J.; Engel, Vic; Fuentes, Jose D. (2013). "Vegetation-microclimate feedbacks in woodland-grassland ecotones". Global Ecology and Biogeography. 22 (4): 364–379. doi: 10.1111/geb.12000.
- ^ Lee, Jeom-Sook; Kim, Jong-Wook (2018). "Dynamics of zonal halophyte communities in salt marshes in the world". Journal of Marine and Island Cultures. 7. doi: 10.21463/JMIC.2018.07.1.06. S2CID 133926655.
- ^ Cavanaugh, K. C.; Kellner, J. R.; Forde, A. J.; Gruner, D. S.; Parker, J. D.; Rodriguez, W.; Feller, I. C. (2014). "Poleward expansion of mangroves is a threshold response to decreased frequency of extreme cold events". Proceedings of the National Academy of Sciences. 111 (2): 723–727. Bibcode: 2014PNAS..111..723C. doi: 10.1073/pnas.1315800111. PMC 3896164. PMID 24379379.
- ^ Gedan, Keryn B.; Kirwan, Matthew L.; Wolanski, Eric; Barbier, Edward B.; Silliman, Brian R. (2011). "The present and future role of coastal wetland vegetation in protecting shorelines: Answering recent challenges to the paradigm". Climatic Change. 106 (1): 7–29. Bibcode: 2011ClCh..106....7G. doi: 10.1007/s10584-010-0003-7. S2CID 17867808.
-
^ Kadlec, Robert (2009).
Treatment wetlands. Boca Raton, FL.
ISBN
978-1-4200-1251-4.
OCLC
311307374.
{{ cite book}}: CS1 maint: location missing publisher ( link) - ^ Yang, Q.; Tam, N.F.Y.; Wong, Y.S.; Luan, T.G.; Su, W.S.; Lan, C.Y.; Shin, P.K.S.; Cheung, S.G. (2008). "Potential use of mangroves as constructed wetland for municipal sewage treatment in Futian, Shenzhen, China". Marine Pollution Bulletin. 57 (6–12): 735–743. Bibcode: 2008MarPB..57..735Y. doi: 10.1016/j.marpolbul.2008.01.037. PMID 18342338.
- ^ Ouyang, Xiaoguang; Guo, Fen (2016). "Paradigms of mangroves in treatment of anthropogenic wastewater pollution". Science of the Total Environment. 544: 971–979. Bibcode: 2016ScTEn.544..971O. doi: 10.1016/j.scitotenv.2015.12.013. PMID 26706768.
- ^ Gambrell, R. P. (1994). "Trace and Toxic Metals in Wetlands-A Review". Journal of Environmental Quality. 23 (5): 883–891. doi: 10.2134/jeq1994.00472425002300050005x. PMID 34872228.
- ^ McLeod, Elizabeth; Chmura, Gail L.; Bouillon, Steven; Salm, Rodney; Björk, Mats; Duarte, Carlos M.; Lovelock, Catherine E.; Schlesinger, William H.; Silliman, Brian R. (2011). "A blueprint for blue carbon: Toward an improved understanding of the role of vegetated coastal habitats in sequestering CO 2". Frontiers in Ecology and the Environment. 9 (10): 552–560. doi: 10.1890/110004.
- ^ Lipcius, Romuald N.; Seitz, Rochelle D.; Seebo, Michael S.; Colón-Carrión, Duamed (2005). "Density, abundance and survival of the blue crab in seagrass and unstructured salt marsh nurseries of Chesapeake Bay". Journal of Experimental Marine Biology and Ecology. 319 (1–2): 69–80. doi: 10.1016/j.jembe.2004.12.034.
- ^ a b Mumby, Peter J. (2006). "Connectivity of reef fish between mangroves and coral reefs: Algorithms for the design of marine reserves at seascape scales". Biological Conservation. 128 (2): 215–222. doi: 10.1016/j.biocon.2005.09.042.
- ^ Aburto-Oropeza, O.; Ezcurra, E.; Danemann, G.; Valdez, V.; Murray, J.; Sala, E. (2008). "Mangroves in the Gulf of California increase fishery yields". Proceedings of the National Academy of Sciences. 105 (30): 10456–10459. Bibcode: 2008PNAS..10510456A. doi: 10.1073/pnas.0804601105. PMC 2492483. PMID 18645185.
- ^ Nagelkerken, I.; Blaber, S.J.M.; Bouillon, S.; Green, P.; Haywood, M.; Kirton, L.G.; Meynecke, J.-O.; Pawlik, J.; Penrose, H.M.; Sasekumar, A.; Somerfield, P.J. (2008). "The habitat function of mangroves for terrestrial and marine fauna: A review". Aquatic Botany. 89 (2): 155–185. doi: 10.1016/j.aquabot.2007.12.007.
-
^ Renzi, Julianna J.; He, Qiang; Silliman, Brian R. (2019).
"Harnessing Positive Species Interactions to Enhance Coastal Wetland Restoration". Frontiers in Ecology and Evolution. 7.
doi:
10.3389/fevo.2019.00131.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^ a b US Department of Commerce, National Oceanic and Atmospheric Administration. "What is a mangrove forest?". oceanservice.noaa.gov. Retrieved 2019-03-21.
- ^ a b c "Mangroves". Smithsonian Ocean. 30 April 2018. Retrieved 2019-03-21.
- ^ a b c d "What is a Salt Marsh?" (PDF). New Hampshire Department of Environmental Services. 2004. Archived from the original (PDF) on 2020-10-21. Retrieved 2021-10-31.
- ^ a b US Department of Commerce, National Oceanic and Atmospheric Administration. "What is a salt marsh?". oceanservice.noaa.gov. Retrieved 2019-03-20.
- ^ Mann, K.H. 1973. Seaweeds: their productivity and strategy for growth. Science 182: 975-981.
- ^ Graham, M.H., B.P. Kinlan, L.D. Druehl, L.E. Garske, and S. Banks. 2007. Deep-water kelp refugia as potential hotspots of tropical marine diversity and productivity. Proceedings of the National Academy of Sciences 104: 16576-16580.
- ^ Christie, H., Jørgensen, N.M., Norderhaug, K.M., Waage-Nielsen, E., 2003. Species distribution and habitat exploitation of fauna associated with kelp (Laminaria hyperborea) along the Norwegian coast. Journal of the Marine Biological Association of the UK 83, 687-699.
- ^ Jackson, G.A. and C.D. Winant. 1983. Effect of a kelp forest on coastal currents. Continental Shelf Report 2: 75-80.
- ^ Steneck, R.S., M.H. Graham, B.J. Bourque, D. Corbett, J.M. Erlandson, J.A. Estes and M.J. Tegner. 2002. Kelp forest ecosystems: biodiversity, stability, resilience and future. Environmental Conservation 29: 436-459.
- ^ Sala, E., C.F. Bourdouresque and M. Harmelin-Vivien. 1998. Fishing, trophic cascades, and the structure of algal assemblages: evaluation of an old but untested paradigm. Oikos 82: 425-439.
- ^ Dayton, P.K. 1985a. Ecology of kelp communities. Annual Review of Ecology and Systematics 16: 215-245.
- ^ Norderhaug, K.M., Christie, H., 2009. Sea urchin grazing and kelp re-vegetation in the NE Atlantic. Marine Biology Research 5, 515-528
- ^ Morton, Adam; Cordell, Marni; Fanner, David; Ball, Andy; Evershed, Nick. "The dead sea: Tasmania's underwater forests disappearing in our lifetime". the Guardian. Retrieved 2020-10-22.
- ^ Steinbauer, James. "What Will It Take to Bring Back the Kelp Forest? - Bay Nature Magazine". Bay Nature. Retrieved 2020-10-22.
- ^ Harris, P.T.; Macmillan-Lawler, M.; Rupp, J.; Baker, E.K. (2014). "Geomorphology of the oceans". Marine Geology. 352: 4–24. Bibcode: 2014MGeol.352....4H. doi: 10.1016/j.margeo.2014.01.011.
- ^ a b Otero, X.L., De La Peña-Lastra, S., Pérez-Alberti, A., Ferreira, T.O. and Huerta-Diaz, M.A. (2018) "Seabird colonies as important global drivers in the nitrogen and phosphorus cycles". Nature communications, 9(1): 1–8. doi: 10.1038/s41467-017-02446-8. Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
- ^ Barrett, Kyle; Anderson, Wendy B.; Wait, D. Alexander; Grismer, L. Lee; Polis†, Gary A.; Rose†, Michael D. (2005). "Marine subsidies alter the diet and abundance of insular and coastal lizard populations". Oikos. 109: 145–153. doi: 10.1111/j.0030-1299.2005.13728.x.
- ^ Polis, Gary A.; Hurd, Stephen D. (1996). "Linking Marine and Terrestrial Food Webs: Allochthonous Input from the Ocean Supports High Secondary Productivity on Small Islands and Coastal Land Communities". The American Naturalist. 147 (3): 396–423. doi: 10.1086/285858. S2CID 84701185.
- ^ Gende, Scott M.; Edwards, Richard T.; Willson, Mary F.; Wipfli, Mark S. (2002). "Pacific Salmon in Aquatic and Terrestrial Ecosystems". BioScience. 52 (10): 917. doi: 10.1641/0006-3568(2002)052[0917:PSIAAT]2.0.CO;2. ISSN 0006-3568.
- ^ a b Gagnon, Karine; Rothäusler, Eva; Syrjänen, Anneli; Yli-Renko, Maria; Jormalainen, Veijo (2013). "Seabird Guano Fertilizes Baltic Sea Littoral Food Webs". PLOS ONE. 8 (4): e61284. Bibcode: 2013PLoSO...861284G. doi: 10.1371/journal.pone.0061284. PMC 3623859. PMID 23593452.
- ^ Mizota, Chitoshi; Noborio, Kosuke; Mori, Yoshiaki (2012). "The Great Cormorant (Phalacrocorax carbo) colony as a "hot spot" of nitrous oxide (N2O) emission in central Japan". Atmospheric Environment. 57: 29–34. Bibcode: 2012AtmEn..57...29M. doi: 10.1016/j.atmosenv.2012.02.007.
- ^ Bird, Michael I.; Tait, Elaine; Wurster, Christopher M.; Furness, Robert W. (2008). "Stable carbon and nitrogen isotope analysis of avian uric acid". Rapid Communications in Mass Spectrometry. 22 (21): 3393–3400. Bibcode: 2008RCMS...22.3393B. doi: 10.1002/rcm.3739. PMID 18837063.
- ^ a b Caut, Stéphane; Angulo, Elena; Pisanu, Benoit; Ruffino, Lise; Faulquier, Lucie; Lorvelec, Olivier; Chapuis, Jean-Louis; Pascal, Michel; Vidal, Eric; Courchamp, Franck (2012). "Seabird Modulations of Isotopic Nitrogen on Islands". PLOS ONE. 7 (6): e39125. Bibcode: 2012PLoSO...739125C. doi: 10.1371/journal.pone.0039125. PMC 3377609. PMID 22723945.
- ^ Mulder, Christa P. H.; Anderson, Wendy B.; Towns, David R.; Bellingham, Peter J. (8 September 2011). Seabird Islands: Ecology, Invasion, and Restoration. Oup USA. ISBN 9780199735693.
- ^ McFadden, Tyler Neal; Kauffman, J. Boone; Bhomia, Rupesh K. (2016). "Effects of nesting waterbirds on nutrient levels in mangroves, Gulf of Fonseca, Honduras". Wetlands Ecology and Management. 24 (2): 217–229. doi: 10.1007/s11273-016-9480-4. S2CID 6021420.
- ^ Zwolicki, Adrian; Zmudczyńska-Skarbek, Katarzyna Małgorzata; Iliszko, Lech; Stempniewicz, Lech (2013). "Guano deposition and nutrient enrichment in the vicinity of planktivorous and piscivorous seabird colonies in Spitsbergen". Polar Biology. 36 (3): 363–372. doi: 10.1007/s00300-012-1265-5. S2CID 12110520.
- ^ Doughty, Christopher E.; Roman, Joe; Faurby, Søren; Wolf, Adam; Haque, Alifa; Bakker, Elisabeth S.; Malhi, Yadvinder; Dunning, John B.; Svenning, Jens-Christian (2016). "Global nutrient transport in a world of giants". Proceedings of the National Academy of Sciences. 113 (4): 868–873. Bibcode: 2016PNAS..113..868D. doi: 10.1073/pnas.1502549112. PMC 4743783. PMID 26504209.
- ^ a b Honig, Susanna E.; Mahoney, Brenna (2016). "Evidence of seabird guano enrichment on a coral reef in Oahu, Hawaii". Marine Biology. 163 (2). doi: 10.1007/s00227-015-2808-4. S2CID 87850538.
- ^ Kolb, GS; Ekholm, J.; Hambäck, PA (2010). "Effects of seabird nesting colonies on algae and aquatic invertebrates in coastal waters". Marine Ecology Progress Series. 417: 287–300. Bibcode: 2010MEPS..417..287K. doi: 10.3354/meps08791.
- ^ Wainright, S. C.; Haney, J. C.; Kerr, C.; Golovkin, A. N.; Flint, M. V. (1998). "Utilization of nitrogen derived from seabird guano by terrestrial and marine plants at St. Paul, Pribilof Islands, Bering Sea, Alaska". Marine Biology. 131: 63–71. doi: 10.1007/s002270050297. S2CID 83734364.
- ^ Staunton Smith, J.; Johnson, CR (1995). "Nutrient inputs from seabirds and humans on a populated coral cay". Marine Ecology Progress Series. 124: 189–200. Bibcode: 1995MEPS..124..189S. doi: 10.3354/meps124189.
- ^
a
b
c Lorrain, Anne; Houlbrèque, Fanny; Benzoni, Francesca; Barjon, Lucie; Tremblay-Boyer, Laura; Menkes, Christophe; Gillikin, David P.; Payri, Claude; Jourdan, Hervé; Boussarie, Germain; Verheyden, Anouk; Vidal, Eric (2017).
"Seabirds supply nitrogen to reef-building corals on remote Pacific islets". Scientific Reports. 7 (1): 3721.
Bibcode:
2017NatSR...7.3721L.
doi:
10.1038/s41598-017-03781-y.
PMC
5473863.
PMID
28623288.
S2CID
6539261.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^ Connell, Des W. (4 May 2018). Pollution in Tropical Aquatic Systems. CRC Press. ISBN 9781351092777.
- ^ Hatcher, Bruce Gordon (1990). "Coral reef primary productivity. A hierarchy of pattern and process". Trends in Ecology & Evolution. 5 (5): 149–155. doi: 10.1016/0169-5347(90)90221-X. PMID 21232343.
- ^ Falkowski, Paul G.; Dubinsky, Zvy; Muscatine, Leonard; McCloskey, Lawrence (1993). "Population Control in Symbiotic Corals". BioScience. 43 (9): 606–611. doi: 10.2307/1312147. JSTOR 1312147.
- ^ a b Marubini, F.; Davies, P. S. (1996). "Nitrate increases zooxanthellae population density and reduces skeletogenesis in corals". Marine Biology. 127 (2): 319–328. doi: 10.1007/BF00942117. S2CID 85085823.
- ^ Muscatine, L. (1990) "The role of symbiotic algae in carbon and energy flux in reef corals", Ecosystem World, 25: 75–87.
- ^ Ferrier, M. Drew (1991). "Net uptake of dissolved free amino acids by four scleractinian corals". Coral Reefs. 10 (4): 183–187. Bibcode: 1991CorRe..10..183F. doi: 10.1007/BF00336772. S2CID 25973061.
- ^ Furla, P.; Allemand, D.; Shick, J. M.; Ferrier-Pagès, C.; Richier, S.; Plantivaux, A.; Merle, P. L.; Tambutté, S. (2005). "The Symbiotic Anthozoan: A Physiological Chimera between Alga and Animal". Integrative and Comparative Biology. 45 (4): 595–604. doi: 10.1093/icb/45.4.595. PMID 21676806.
- ^ Mills, Matthew M.; Lipschultz, Fredric; Sebens, Kenneth P. (2004). "Particulate matter ingestion and associated nitrogen uptake by four species of scleractinian corals". Coral Reefs. 23 (3): 311–323. doi: 10.1007/s00338-004-0380-3. S2CID 13212636.
- ^ Mills, M. M.; Sebens, K. P. (2004). "Ingestion and assimilation of nitrogen from benthic sediments by three species of coral". Marine Biology. 145 (6): 1097–1106. doi: 10.1007/s00227-004-1398-3. S2CID 84698653.
- ^ Houlbrèque, F.; Tambutté, E.; Richard, C.; Ferrier-Pagès, C. (2004). "Importance of a micro-diet for scleractinian corals". Marine Ecology Progress Series. 282: 151–160. Bibcode: 2004MEPS..282..151H. doi: 10.3354/meps282151.
- ^ Ferrier-Pagès, C.; Witting, J.; Tambutté, E.; Sebens, K. P. (2003). "Effect of natural zooplankton feeding on the tissue and skeletal growth of the scleractinian coral Stylophora pistillata". Coral Reefs. 22 (3): 229–240. doi: 10.1007/s00338-003-0312-7. S2CID 44869188.
- ^ Marubini, Francesca; Thake, Brenda (1999). "Bicarbonate addition promotes coral growth". Limnology and Oceanography. 44 (3): 716–720. Bibcode: 1999LimOc..44..716M. doi: 10.4319/lo.1999.44.3.0716. S2CID 83654833.
- ^ Ferrier-Pagès, C.; Leclercq, N.; Jaubert, J.; Pelegrí, SP (2000). "Enhancement of pico- and nanoplankton growth by coral exudates". Aquatic Microbial Ecology. 21: 203–209. doi: 10.3354/ame021203.
- ^ Renegar, DA; Riegl, BM (2005). "Effect of nutrient enrichment and elevated CO2 partial pressure on growth rate of Atlantic scleractinian coral Acropora cervicornis". Marine Ecology Progress Series. 293: 69–76. Bibcode: 2005MEPS..293...69R. doi: 10.3354/meps293069.
- ^
a
b
c
d
e
f Atwood, Trisha B.; Hammill, Edd (2018).
"The Importance of Marine Predators in the Provisioning of Ecosystem Services by Coastal Plant Communities". Frontiers in Plant Science. 9: 1289.
doi:
10.3389/fpls.2018.01289.
PMC
6129962.
PMID
30233626.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^ Hairston, Nelson G.; Smith, Frederick E.; Slobodkin, Lawrence B. (1960). "Community Structure, Population Control, and Competition". The American Naturalist. 94 (879). University of Chicago Press: 421–425. doi: 10.1086/282146. ISSN 0003-0147. S2CID 84548124.
- ^ a b c d Estes, James A.; Terborgh, John; Brashares, Justin S.; Power, Mary E.; Berger, Joel; Bond, William J.; Carpenter, Stephen R.; Essington, Timothy E.; Holt, Robert D.; Jackson, Jeremy B. C.; Marquis, Robert J.; Oksanen, Lauri; Oksanen, Tarja; Paine, Robert T.; Pikitch, Ellen K.; Ripple, William J.; Sandin, Stuart A.; Scheffer, Marten; Schoener, Thomas W.; Shurin, Jonathan B.; Sinclair, Anthony R. E.; Soulé, Michael E.; Virtanen, Risto; Wardle, David A. (2011). "Trophic Downgrading of Planet Earth". Science. 333 (6040): 301–306. Bibcode: 2011Sci...333..301E. doi: 10.1126/science.1205106. PMID 21764740. S2CID 7752940.
- ^ Zedler, Joy B.; Kercher, Suzanne (2005). "WETLAND RESOURCES: Status, Trends, Ecosystem Services, and Restorability". Annual Review of Environment and Resources. 30: 39–74. doi: 10.1146/annurev.energy.30.050504.144248.
- ^ Waycott, M.; Duarte, C. M.; Carruthers, T. J. B.; Orth, R. J.; Dennison, W. C.; Olyarnik, S.; Calladine, A.; Fourqurean, J. W.; Heck, K. L.; Hughes, A. R.; Kendrick, G. A.; Kenworthy, W. J.; Short, F. T.; Williams, S. L. (2009). "Accelerating loss of seagrasses across the globe threatens coastal ecosystems". Proceedings of the National Academy of Sciences. 106 (30): 12377–12381. Bibcode: 2009PNAS..10612377W. doi: 10.1073/pnas.0905620106. PMC 2707273. PMID 19587236.
- ^ Duarte, Carlos M.; Losada, Iñigo J.; Hendriks, Iris E.; Mazarrasa, Inés; Marbà, Núria (2013). "The role of coastal plant communities for climate change mitigation and adaptation". Nature Climate Change. 3 (11): 961–968. Bibcode: 2013NatCC...3..961D. doi: 10.1038/nclimate1970.
- ^ Paleczny, Michelle; Hammill, Edd; Karpouzi, Vasiliki; Pauly, Daniel (2015). "Population Trend of the World's Monitored Seabirds, 1950-2010". PLOS ONE. 10 (6): e0129342. Bibcode: 2015PLoSO..1029342P. doi: 10.1371/journal.pone.0129342. PMC 4461279. PMID 26058068.
- ^ Jackson, J. B. C. (8 May 2001). "What was natural in the coastal oceans?". Proceedings of the National Academy of Sciences. 98 (10): 5411–5418. Bibcode: 2001PNAS...98.5411J. doi: 10.1073/pnas.091092898. ISSN 0027-8424. PMC 33227. PMID 11344287.
- ^ Jackson, Jeremy B. C.; Kirby, Michael X.; Berger, Wolfgang H.; Bjorndal, Karen A.; Botsford, Louis W.; Bourque, Bruce J.; Bradbury, Roger H.; Cooke, Richard; Erlandson, Jon; Estes, James A.; Hughes, Terence P.; Kidwell, Susan; Lange, Carina B.; Lenihan, Hunter S.; Pandolfi, John M.; Peterson, Charles H.; Steneck, Robert S.; Tegner, Mia J.; Warner, Robert R. (27 July 2001). "Historical Overfishing and the Recent Collapse of Coastal Ecosystems". Science. 293 (5530). American Association for the Advancement of Science (AAAS): 629–637. doi: 10.1126/science.1059199. ISSN 0036-8075. PMID 11474098. S2CID 1459898.
- ^ a b McCauley, Douglas J.; Pinsky, Malin L.; Palumbi, Stephen R.; Estes, James A.; Joyce, Francis H.; Warner, Robert R. (2015). "Marine defaunation: Animal loss in the global ocean". Science. 347 (6219). doi: 10.1126/science.1255641. PMID 25593191. S2CID 2500224.
- ^ Lotze, Heike K.; Worm, Boris (2009). "Historical baselines for large marine animals". Trends in Ecology & Evolution. 24 (5): 254–262. doi: 10.1016/j.tree.2008.12.004. PMID 19251340.
- ^ Estes, James A.; Palmisano, John F. (1974). "Sea Otters: Their Role in Structuring Nearshore Communities". Science. 185 (4156): 1058–1060. Bibcode: 1974Sci...185.1058E. doi: 10.1126/science.185.4156.1058. PMID 17738247. S2CID 35892592.
- ^ Sala, E.; Zabala, M. (1996). "Fish predation and the structure of the sea urchin Paracentrotus lividus populations in the NW Mediterranean". Marine Ecology Progress Series. 140: 71–81. Bibcode: 1996MEPS..140...71S. doi: 10.3354/meps140071.
- ^ Myers, Ransom A.; Baum, Julia K.; Shepherd, Travis D.; Powers, Sean P.; Peterson, Charles H. (2007). "Cascading Effects of the Loss of Apex Predatory Sharks from a Coastal Ocean". Science. 315 (5820): 1846–1850. Bibcode: 2007Sci...315.1846M. doi: 10.1126/science.1138657. PMID 17395829. S2CID 22332630.
- ^ Heithaus, Michael R.; Alcoverro, Teresa; Arthur, Rohan; Burkholder, Derek A.; Coates, Kathryn A.; Christianen, Marjolijn J. A.; Kelkar, Nachiket; Manuel, Sarah A.; Wirsing, Aaron J.; Kenworthy, W. Judson; Fourqurean, James W. (2014). "Seagrasses in the age of sea turtle conservation and shark overfishing". Frontiers in Marine Science. 1. doi: 10.3389/fmars.2014.00028. hdl: 10261/102715.
- ^ Coverdale, Tyler C.; Brisson, Caitlin P.; Young, Eric W.; Yin, Stephanie F.; Donnelly, Jeffrey P.; Bertness, Mark D. (2014). "Indirect Human Impacts Reverse Centuries of Carbon Sequestration and Salt Marsh Accretion". PLOS ONE. 9 (3): e93296. Bibcode: 2014PLoSO...993296C. doi: 10.1371/journal.pone.0093296. PMC 3968132. PMID 24675669.
- ^ Wilmers, Christopher C.; Estes, James A.; Edwards, Matthew; Laidre, Kristin L.; Konar, Brenda (2012). "Do trophic cascades affect the storage and flux of atmospheric carbon? An analysis of sea otters and kelp forests". Frontiers in Ecology and the Environment. 10 (8): 409–415. doi: 10.1890/110176.
- ^ a b Atwood, Trisha B.; Connolly, Rod M.; Ritchie, Euan G.; Lovelock, Catherine E.; Heithaus, Michael R.; Hays, Graeme C.; Fourqurean, James W.; MacReadie, Peter I. (2015). "Predators help protect carbon stocks in blue carbon ecosystems". Nature Climate Change. 5 (12): 1038–1045. Bibcode: 2015NatCC...5.1038A. doi: 10.1038/NCLIMATE2763.
- ^ Hughes, Brent B.; Hammerstrom, Kamille K.; Grant, Nora E.; Hoshijima, Umi; Eby, Ron; Wasson, Kerstin (11 May 2016). "Trophic cascades on the edge: fostering seagrass resilience via a novel pathway". Oecologia. 182 (1). Springer Science and Business Media LLC: 231–241. Bibcode: 2016Oecol.182..231H. doi: 10.1007/s00442-016-3652-z. ISSN 0029-8549. PMID 27167224. S2CID 15168162.
- ^ Silvestri, Silvia; Kershaw, Francine (2010). Framing the Flow: Innovative Approaches to Understand, Protect and Value Ecosystem Services Across Linked Habitats. University of New England Press. ISBN 9789280730654.
- ^
a
b
c Pittman, SJ; et al. (2021).
"Seascape ecology: Identifying research priorities for an emerging ocean sustainability science". Marine Ecology Progress Series. 663: 1–29.
Bibcode:
2021MEPS..663....1P.
doi:
10.3354/meps13661.
hdl:
10536/DRO/DU:30151612.
ISSN
0171-8630.
S2CID
233453217.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^ Pittman, Simon (2018). Seascape ecology. Hoboken, NJ, USA: John Wiley & Sons, Inc. ISBN 978-1-119-08443-3. OCLC 993642256.
- ^ Steele, John H. (1978). "Some Comments on Plankton Patches". Spatial Pattern in Plankton Communities. pp. 1–20. doi: 10.1007/978-1-4899-2195-6_1. ISBN 978-1-4899-2197-0.
- ^ Levin, Simon A. (1992). "The Problem of Pattern and Scale in Ecology: The Robert H. MacArthur Award Lecture". Ecology. 73 (6): 1943–1967. doi: 10.2307/1941447. JSTOR 1941447.
- ^ a b Pittman, Simon (2018). Seascape ecology. Hoboken, NJ, USA: John Wiley & Sons, Inc. ISBN 978-1-119-08443-3. OCLC 993642256.
- ^ Thrush, S.F; Schneider, D.C; Legendre, P.; Whitlatch, R.B; Dayton, P.K; Hewitt, J.E; Hines, A.H; Cummings, V.J; Lawrie, S.M; Grant, J.; Pridmore, R.D; Turner, S.J; McArdle, B.H (1997). "Scaling-up from experiments to complex ecological systems: Where to next?". Journal of Experimental Marine Biology and Ecology. 216 (1–2): 243–254. doi: 10.1016/S0022-0981(97)00099-3.
- ^ Schneider, David C. (2001). "The Rise of the Concept of Scale in Ecology". BioScience. 51 (7): 545. doi: 10.1641/0006-3568(2001)051[0545:TROTCO]2.0.CO;2. ISSN 0006-3568. S2CID 27798897.
- ^ a b Boström, C.; Pittman, SJ; Simenstad, C.; Kneib, RT (2011). "Seascape ecology of coastal biogenic habitats: Advances, gaps, and challenges". Marine Ecology Progress Series. 427: 191–217. Bibcode: 2011MEPS..427..191B. doi: 10.3354/meps09051. hdl: 1834/30670.
- ^ Bishop, Melanie J.; Mayer-Pinto, Mariana; Airoldi, Laura; Firth, Louise B.; Morris, Rebecca L.; Loke, Lynette H.L.; Hawkins, Stephen J.; Naylor, Larissa A.; Coleman, Ross A.; Chee, Su Yin; Dafforn, Katherine A. (2017). "Effects of ocean sprawl on ecological connectivity: Impacts and solutions". Journal of Experimental Marine Biology and Ecology. 492: 7–30. doi: 10.1016/j.jembe.2017.01.021. hdl: 10026.1/9695.
- ^ Halpern, Benjamin S.; Frazier, Melanie; Afflerbach, Jamie; Lowndes, Julia S.; Micheli, Fiorenza; o'Hara, Casey; Scarborough, Courtney; Selkoe, Kimberly A. (2019). "Recent pace of change in human impact on the world's ocean". Scientific Reports. 9 (1): 11609. Bibcode: 2019NatSR...911609H. doi: 10.1038/s41598-019-47201-9. PMC 6691109. PMID 31406130.
- ^ Borja, Angel; Andersen, Jesper H.; Arvanitidis, Christos D.; Basset, Alberto; Buhl-Mortensen, Lene; Carvalho, Susana; Dafforn, Katherine A.; Devlin, Michelle J.; Escobar-Briones, Elva G.; Grenz, Christian; Harder, Tilmann; Katsanevakis, Stelios; Liu, Dongyan; Metaxas, Anna; Morán, Xosé Anxelu G.; Newton, Alice; Piroddi, Chiara; Pochon, Xavier; Queirós, Ana M.; Snelgrove, Paul V. R.; Solidoro, Cosimo; St. John, Michael A.; Teixeira, Heliana (2020). "Past and Future Grand Challenges in Marine Ecosystem Ecology". Frontiers in Marine Science. 7. doi: 10.3389/fmars.2020.00362. hdl: 10400.1/14027.
- ^ a b Claudet, Joachim; et al. (2020). "A Roadmap for Using the UN Decade of Ocean Science for Sustainable Development in Support of Science, Policy, and Action". One Earth. 2 (1): 34–42. Bibcode: 2020OEart...2...34C. doi: 10.1016/j.oneear.2019.10.012. S2CID 211352061.
- ^ d'Urban Jackson, Tim; Williams, Gareth J.; Walker-Springett, Guy; Davies, Andrew J. (2020). "Three-dimensional digital mapping of ecosystems: A new era in spatial ecology". Proceedings of the Royal Society B: Biological Sciences. 287 (1920). doi: 10.1098/rspb.2019.2383. PMC 7031661. PMID 32075534.
- ^ García-Charton, J. A.; Pérez-Ruzafa, Á.; Sánchez-Jerez, P.; Bayle-Sempere, J. T.; Reñones, O.; Moreno, D. (2004). "Multi-scale spatial heterogeneity, habitat structure, and the effect of marine reserves on Western Mediterranean rocky reef fish assemblages". Marine Biology. 144: 161–182. doi: 10.1007/s00227-003-1170-0. S2CID 85630092.
- ^ Huntington, Brittany E.; Karnauskas, Mandy; Babcock, Elizabeth A.; Lirman, Diego (2010). "Untangling Natural Seascape Variation from Marine Reserve Effects Using a Landscape Approach". PLOS ONE. 5 (8): e12327. Bibcode: 2010PLoSO...512327H. doi: 10.1371/journal.pone.0012327. PMC 2924891. PMID 20808833.
- ^ a b c Olds, Andrew D.; Connolly, Rod M.; Pitt, Kylie A.; Pittman, Simon J.; Maxwell, Paul S.; Huijbers, Chantal M.; Moore, Brad R.; Albert, Simon; Rissik, David; Babcock, Russell C.; Schlacher, Thomas A. (2016). "Quantifying the conservation value of seascape connectivity: A global synthesis". Global Ecology and Biogeography. 25: 3–15. doi: 10.1111/geb.12388.
- ^ Proudfoot, Beatrice; Devillers, Rodolphe; Brown, Craig J. (2020). "Integrating fine‐scale seafloor mapping and spatial pattern metrics into marine conservation prioritization". Aquatic Conservation: Marine and Freshwater Ecosystems. 30 (8): 1613–1625. doi: 10.1002/aqc.3360. S2CID 219767109.
-
^ Berkström, Charlotte; Papadopoulos, Myron; Jiddawi, Narriman Saleh; Nordlund, Lina Mtwana (2019).
"Fishers' Local Ecological Knowledge (LEK) on Connectivity and Seascape Management". Frontiers in Marine Science. 6.
doi:
10.3389/fmars.2019.00130.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
-
^ von Thenen, Miriam; Frederiksen, Pia; Hansen, Henning Sten; Schiele, Kerstin S. (2020).
"A structured indicator pool to operationalize expert-based ecosystem service assessments for marine spatial planning". Ocean & Coastal Management. 187: 105071.
Bibcode:
2020OCM...18705071V.
doi:
10.1016/j.ocecoaman.2019.105071.
S2CID
212792922.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^
a
b
c
d
e Carlson, Rachel R.; Evans, Luke J.; Foo, Shawna A.; Grady, Bryant W.; Li, Jiwei; Seeley, Megan; Xu, Yaping; Asner, Gregory P. (2021).
"Synergistic benefits of conserving land-sea ecosystems". Global Ecology and Conservation. 28. Elsevier BV: e01684.
doi:
10.1016/j.gecco.2021.e01684.
ISSN
2351-9894.
S2CID
236255580.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^
a
b
c
d
e
f
g Ávila-Thieme, M. Isidora; Corcoran, Derek; Pérez-Matus, Alejandro; Wieters, Evie A.; Navarrete, Sergio A.; Marquet, Pablo A.; Valdovinos, Fernanda S. (2021).
"Alteration of coastal productivity and artisanal fisheries interact to affect a marine food web". Scientific Reports. 11 (1): 1765.
Bibcode:
2021NatSR..11.1765A.
doi:
10.1038/s41598-021-81392-4.
PMC
7815714.
PMID
33469119.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^ Somero, G. N. (2010). "The physiology of climate change: How potentials for acclimatization and genetic adaptation will determine 'winners' and 'losers'". Journal of Experimental Biology. 213 (6): 912–920. doi: 10.1242/jeb.037473. PMID 20190116. S2CID 16838841.
- ^ Hoegh-Guldberg, Ove; Bruno, John F. (2010). "The Impact of Climate Change on the World's Marine Ecosystems". Science. 328 (5985): 1523–1528. Bibcode: 2010Sci...328.1523H. doi: 10.1126/science.1189930. PMID 20558709. S2CID 206526700.
- ^ Brose, Ulrich; Dunne, Jennifer A.; Montoya, Jose M.; Petchey, Owen L.; Schneider, Florian D.; Jacob, Ute (2012). "Climate change in size-structured ecosystems". Philosophical Transactions of the Royal Society B: Biological Sciences. 367 (1605): 2903–2912. doi: 10.1098/rstb.2012.0232. PMC 3479741. PMID 23007078.
- ^ Doney, Scott C.; Ruckelshaus, Mary; Emmett Duffy, J.; Barry, James P.; Chan, Francis; English, Chad A.; Galindo, Heather M.; Grebmeier, Jacqueline M.; Hollowed, Anne B.; Knowlton, Nancy; Polovina, Jeffrey; Rabalais, Nancy N.; Sydeman, William J.; Talley, Lynne D. (2012). "Climate Change Impacts on Marine Ecosystems". Annual Review of Marine Science. 4: 11–37. Bibcode: 2012ARMS....4...11D. doi: 10.1146/annurev-marine-041911-111611. PMID 22457967.
- ^ a b Kwiatkowski, Lester; Aumont, Olivier; Bopp, Laurent (2019). "Consistent trophic amplification of marine biomass declines under climate change". Global Change Biology. 25 (1): 218–229. Bibcode: 2019GCBio..25..218K. doi: 10.1111/gcb.14468. PMID 30295401. S2CID 52930402.
- ^ Bakun, Andrew (1990). "Global Climate Change and Intensification of Coastal Ocean Upwelling". Science. 247 (4939): 198–201. Bibcode: 1990Sci...247..198B. doi: 10.1126/science.247.4939.198. PMID 17813287. S2CID 32516158.
- ^ Bakun, Andrew; Field, David B.; Redondo-Rodriguez, ANA; Weeks, Scarla J. (2010). "Greenhouse gas, upwelling-favorable winds, and the future of coastal ocean upwelling ecosystems". Global Change Biology. 16 (4): 1213–1228. Bibcode: 2010GCBio..16.1213B. doi: 10.1111/j.1365-2486.2009.02094.x. S2CID 85126141.
- ^ Thiel, M., Castilla, J.C., Fernández, M. and Navarrete, S., (2007). "The Humboldt current system of northern and central Chile". Oceanography and Marine Biology: An Annual Review, 45: 195-344
- ^ Morales, Carmen; Hormazabal, Samuel; Andrade, Isabel; Correa-Ramirez, Marco (2013). "Time-Space Variability of Chlorophyll-a and Associated Physical Variables within the Region off Central-Southern Chile". Remote Sensing. 5 (11): 5550–5571. Bibcode: 2013RemS....5.5550M. doi: 10.3390/rs5115550.
- ^ Aiken, C. M.; Navarrete, S. A.; Pelegrí, J. L. (2011). "Potential changes in larval dispersal and alongshore connectivity on the central Chilean coast due to an altered wind climate". Journal of Geophysical Research. 116 (G4). Bibcode: 2011JGRG..116.4026A. doi: 10.1029/2011JG001731. hdl: 10261/73010.
- ^ a b Blanchard, Julia L.; Jennings, Simon; Holmes, Robert; Harle, James; Merino, Gorka; Allen, J. Icarus; Holt, Jason; Dulvy, Nicholas K.; Barange, Manuel (2012). "Potential consequences of climate change for primary production and fish production in large marine ecosystems". Philosophical Transactions of the Royal Society B: Biological Sciences. 367 (1605): 2979–2989. doi: 10.1098/rstb.2012.0231. PMC 3479740. PMID 23007086.
- ^ Testa, Giovanni; Masotti, Italo; Farías, Laura (2018). "Temporal Variability in Net Primary Production in an Upwelling Area off Central Chile (36°S)". Frontiers in Marine Science. 5. doi: 10.3389/fmars.2018.00179.
- ^ Batten, Sonia D.; Abu-Alhaija, Rana; Chiba, Sanae; Edwards, Martin; Graham, George; Jyothibabu, R.; Kitchener, John A.; Koubbi, Philippe; McQuatters-Gollop, Abigail; Muxagata, Erik; Ostle, Clare; Richardson, Anthony J.; Robinson, Karen V.; Takahashi, Kunio T.; Verheye, Hans M.; Wilson, Willie (2019). "A Global Plankton Diversity Monitoring Program". Frontiers in Marine Science. 6. doi: 10.3389/fmars.2019.00321.
- ^ Chust, Guillem; Allen, J. Icarus; Bopp, Laurent; Schrum, Corinna; Holt, Jason; Tsiaras, Kostas; Zavatarelli, Marco; Chifflet, Marina; Cannaby, Heather; Dadou, Isabelle; Daewel, Ute; Wakelin, Sarah L.; Machu, Eric; Pushpadas, Dhanya; Butenschon, Momme; Artioli, Yuri; Petihakis, George; Smith, Chris; Garçon, Veronique; Goubanova, Katerina; Le Vu, Briac; Fach, Bettina A.; Salihoglu, Baris; Clementi, Emanuela; Irigoien, Xabier (2014). "Biomass changes and trophic amplification of plankton in a warmer ocean" (PDF). Global Change Biology. 20 (7): 2124–2139. Bibcode: 2014GCBio..20.2124C. doi: 10.1111/gcb.12562. hdl: 11511/31726. PMID 24604761. S2CID 12403222.
- ^ Weidberg, Nicolas; Ospina-Alvarez, Andres; Bonicelli, Jessica; Barahona, Mario; Aiken, Christopher M.; Broitman, Bernardo R.; Navarrete, Sergio A. (2020). "Spatial shifts in productivity of the coastal ocean over the past two decades induced by migration of the Pacific Anticyclone and Bakun's effect in the Humboldt Upwelling Ecosystem". Global and Planetary Change. 193: 103259. arXiv: 2104.11698. Bibcode: 2020GPC...19303259W. doi: 10.1016/j.gloplacha.2020.103259. S2CID 224872806.
- ^ Aguirre, Catalina; García-Loyola, Sebastián; Testa, Giovanni; Silva, Diego; Farías, Laura (2018). "Insight into anthropogenic forcing on coastal upwelling off south-central Chile". Elementa: Science of the Anthropocene. 6: 59. Bibcode: 2018EleSA...6...59A. doi: 10.1525/elementa.314.
- ^ Pascual, Mercedes (2006). Ecological networks : linking structure to dynamics in food webs. Oxford New York: Oxford University Press. ISBN 978-0-19-518816-5. OCLC 727944896.
- ^ a b c Dunne, Jennifer A.; Williams, Richard J.; Martinez, Neo D. (2002). "Network structure and biodiversity loss in food webs: Robustness increases with connectance". Ecology Letters. 5 (4): 558–567. doi: 10.1046/j.1461-0248.2002.00354.x. S2CID 2114852.
- ^ a b c Curtsdotter, Alva; Binzer, Amrei; Brose, Ulrich; De Castro, Francisco; Ebenman, Bo; Eklöf, Anna; Riede, Jens O.; Thierry, Aaron; Rall, Björn C. (2011). "Robustness to secondary extinctions: Comparing trait-based sequential deletions in static and dynamic food webs". Basic and Applied Ecology. 12 (7): 571–580. doi: 10.1016/j.baae.2011.09.008.
- ^ a b Ramos-Jiliberto, Rodrigo; Valdovinos, Fernanda S.; Moisset De Espanés, Pablo; Flores, José D. (2012). "Topological plasticity increases robustness of mutualistic networks". Journal of Animal Ecology. 81 (4): 896–904. doi: 10.1111/j.1365-2656.2012.01960.x. PMID 22313043.
- ^ a b Valdovinos, Fernanda S.; Moisset De Espanés, Pablo; Flores, José D.; Ramos-Jiliberto, Rodrigo (2013). "Adaptive foraging allows the maintenance of biodiversity of pollination networks". Oikos. 122 (6): 907–917. doi: 10.1111/j.1600-0706.2012.20830.x.
- ^ a b Allesina, Stefano; Pascual, Mercedes (2009). "Googling Food Webs: Can an Eigenvector Measure Species' Importance for Coextinctions?". PLOS Computational Biology. 5 (9): e1000494. Bibcode: 2009PLSCB...5E0494A. doi: 10.1371/journal.pcbi.1000494. PMC 2725316. PMID 19730676.
- ^ De Santana, CN; Rozenfeld, AF; Marquet, PA; Duarte, CM (2013). "Topological properties of polar food webs". Marine Ecology Progress Series. 474: 15–26. Bibcode: 2013MEPS..474...15D. doi: 10.3354/meps10073. hdl: 11336/7024.
- ^ Eklöf, Anna; Tang, Si; Allesina, Stefano (2013). "Secondary extinctions in food webs: A Bayesian network approach". Methods in Ecology and Evolution. 4 (8): 760–770. doi: 10.1111/2041-210X.12062. S2CID 84592425.
- ^ Staniczenko, Phillip P. A.; Lewis, Owen T.; Jones, Nick S.; Reed-Tsochas, Felix (2010). "Structural dynamics and robustness of food webs". Ecology Letters. 13 (7): 891–899. doi: 10.1111/j.1461-0248.2010.01485.x. PMID 20482578.
- ^ Ives, Anthony R.; Cardinale, Bradley J. (2004). "Food-web interactions govern the resistance of communities after non-random extinctions". Nature. 429 (6988): 174–177. Bibcode: 2004Natur.429..174I. doi: 10.1038/nature02515. PMID 15141210. S2CID 4351240.
- ^ Rebolledo, Rolando; Navarrete, Sergio A.; Kéfi, Sonia; Rojas, Sergio; Marquet, Pablo A. (2019). "An Open-System Approach to Complex Biological Networks". SIAM Journal on Applied Mathematics. 79 (2): 619–640. doi: 10.1137/17M1153431. S2CID 146077530.
- ^ McCann, Kevin Shear (2000). "The diversity–stability debate". Nature. 405 (6783): 228–233. doi: 10.1038/35012234. PMID 10821283. S2CID 4319289.
- ^ Glaum, Paul; Cocco, Valentin; Valdovinos, Fernanda S. (2020). "Integrating economic dynamics into ecological networks: The case of fishery sustainability". Science Advances. 6 (45). Bibcode: 2020SciA....6.4891G. doi: 10.1126/sciadv.aaz4891. PMC 7673689. PMID 33148659.
- ^ Beusen, Arthur H. W.; Bouwman, Alexander F.; Van Beek, Ludovicus P. H.; Mogollón, José M.; Middelburg, Jack J. (2016). "Global riverine N and P transport to ocean increased during the 20th century despite increased retention along the aquatic continuum". Biogeosciences. 13 (8): 2441–2451. Bibcode: 2016BGeo...13.2441B. doi: 10.5194/bg-13-2441-2016. hdl: 1887/80781. S2CID 54722438.
- ^ Gustafsson, Bo G.; Schenk, Frederik; Blenckner, Thorsten; Eilola, Kari; Meier, H. E. Markus; Müller-Karulis, Bärbel; Neumann, Thomas; Ruoho-Airola, Tuija; Savchuk, Oleg P.; Zorita, Eduardo (2012). "Reconstructing the Development of Baltic Sea Eutrophication 1850–2006". Ambio. 41 (6): 534–548. doi: 10.1007/s13280-012-0318-x. PMC 3428479. PMID 22926877.
- ^ Qu, Hong Juan; Kroeze, Carolien (4 August 2011). "Nutrient export by rivers to the coastal waters of China: management strategies and future trends". Regional Environmental Change. 12 (1). Springer Science and Business Media LLC: 153–167. doi: 10.1007/s10113-011-0248-3. ISSN 1436-3798. S2CID 55439201.
- ^ Cui, S.; Shi, Y.; Groffman, P. M.; Schlesinger, W. H.; Zhu, Y.-G. (2013). "Centennial-scale analysis of the creation and fate of reactive nitrogen in China (1910-2010)". Proceedings of the National Academy of Sciences. 110 (6): 2052–2057. Bibcode: 2013PNAS..110.2052C. doi: 10.1073/pnas.1221638110. PMC 3568337. PMID 23341613.
- ^ Falkowski, P.; Scholes, R. J.; Boyle, E.; Canadell, J.; Canfield, D.; Elser, J.; Gruber, N.; Hibbard, K.; Högberg, P.; Linder, S.; MacKenzie, F. T.; Moore Iii, B.; Pedersen, T.; Rosenthal, Y.; Seitzinger, S.; Smetacek, V.; Steffen, W. (2000). "The Global Carbon Cycle: A Test of Our Knowledge of Earth as a System". Science. 290 (5490): 291–296. Bibcode: 2000Sci...290..291F. doi: 10.1126/science.290.5490.291. PMID 11030643.
- ^ Gruber, Nicolas; Galloway, James N. (2008). "An Earth-system perspective of the global nitrogen cycle". Nature. 451 (7176): 293–296. Bibcode: 2008Natur.451..293G. doi: 10.1038/nature06592. PMID 18202647. S2CID 4318396.
- ^ Statham, Peter J. (2012). "Nutrients in estuaries — an overview and the potential impacts of climate change". Science of the Total Environment. 434: 213–227. Bibcode: 2012ScTEn.434..213S. doi: 10.1016/j.scitotenv.2011.09.088. PMID 22119025.
- ^ Cloern, JE (2001). "Our evolving conceptual model of the coastal eutrophication problem". Marine Ecology Progress Series. 210: 223–253. Bibcode: 2001MEPS..210..223C. doi: 10.3354/meps210223.
- ^ Grall, Jacques; Chauvaud, Laurent (2002). "Marine eutrophication and benthos: The need for new approaches and concepts". Global Change Biology. 8 (9): 813–830. Bibcode: 2002GCBio...8..813G. doi: 10.1046/j.1365-2486.2002.00519.x. S2CID 83974188.
- ^ Diaz, Robert J.; Rosenberg, Rutger (2008). "Spreading Dead Zones and Consequences for Marine Ecosystems". Science. 321 (5891): 926–929. Bibcode: 2008Sci...321..926D. doi: 10.1126/science.1156401. PMID 18703733. S2CID 32818786.
- ^ Breitburg, Denise; Levin, Lisa A.; Oschlies, Andreas; Grégoire, Marilaure; Chavez, Francisco P.; Conley, Daniel J.; Garçon, Véronique; Gilbert, Denis; Gutiérrez, Dimitri; Isensee, Kirsten; Jacinto, Gil S.; Limburg, Karin E.; Montes, Ivonne; Naqvi, S. W. A.; Pitcher, Grant C.; Rabalais, Nancy N.; Roman, Michael R.; Rose, Kenneth A.; Seibel, Brad A.; Telszewski, Maciej; Yasuhara, Moriaki; Zhang, Jing (2018). "Declining oxygen in the global ocean and coastal waters". Science. 359 (6371). Bibcode: 2018Sci...359M7240B. doi: 10.1126/science.aam7240. PMID 29301986. S2CID 206657115.
- ^ Griffiths, Jennifer R.; Kadin, Martina; Nascimento, Francisco J. A.; Tamelander, Tobias; Törnroos, Anna; Bonaglia, Stefano; Bonsdorff, Erik; Brüchert, Volker; Gårdmark, Anna; Järnström, Marie; Kotta, Jonne; Lindegren, Martin; Nordström, Marie C.; Norkko, Alf; Olsson, Jens; Weigel, Benjamin; Žydelis, Ramunas; Blenckner, Thorsten; Niiranen, Susa; Winder, Monika (2017). "The importance of benthic-pelagic coupling for marine ecosystem functioning in a changing world". Global Change Biology. 23 (6): 2179–2196. Bibcode: 2017GCBio..23.2179G. doi: 10.1111/gcb.13642. PMID 28132408. S2CID 13874905.
- ^ Cederwall, H. and Elmgren, R. (1980) "Biomass increase of benthic macrofauna demonstrates eutrophication of the Baltic Sea". In: Symposium of the Baltic Marine Biologists: relationship and exchange between the pelagic and benthic biota.
- ^ Pearson, T. H., and Rosenberg, R. (1987). “Feast and famine: Structuring factors in marine benthic communities”, in Organization Of Communities:The 27th Symposium of the British Ecological Society, Aberystwyth 1986, Eds: P. S. Giller and J. H. R. Gee (Oxford: Blackwell), 373–395.
- ^ a b Josefson, A.B.; Rasmussen, B. (2000). "Nutrient Retention by Benthic Macrofaunal Biomass of Danish Estuaries: Importance of Nutrient Load and Residence Time". Estuarine, Coastal and Shelf Science. 50 (2): 205–216. Bibcode: 2000ECSS...50..205J. doi: 10.1006/ecss.1999.0562.
- ^ Kautsky, N.; Evans, S. (1987). "Role of biodeposition by Mytilus edulis in the circulation of matter and nutrients in a Baltic coastal ecosystem". Marine Ecology Progress Series. 38: 201–212. Bibcode: 1987MEPS...38..201K. doi: 10.3354/meps038201.
- ^ Newell R.I. (2004) "Ecosystem influences of natural and cultivated populations of suspension-feeding bivalve molluscs: a review". Journal of Shellfish Research, 23(1): 51–62.
- ^ Herman, P.M.J.; Middelburg, J.J.; Van De Koppel, J.; Heip, C.H.R. (1999). "Ecology of Estuarine Macrobenthos". Estuaries. Advances in Ecological Research. Vol. 29. pp. 195–240. doi: 10.1016/S0065-2504(08)60194-4. ISBN 9780120139293.
- ^ Ehrnsten, Eva; Norkko, Alf; Timmermann, Karen; Gustafsson, Bo G. (2019). "Benthic-pelagic coupling in coastal seas – Modelling macrofaunal biomass and carbon processing in response to organic matter supply". Journal of Marine Systems. 196: 36–47. Bibcode: 2019JMS...196...36E. doi: 10.1016/j.jmarsys.2019.04.003. S2CID 155821874.
- ^ Kristensen, E.; Penha-Lopes, G.; Delefosse, M.; Valdemarsen, T.; Quintana, CO; Banta, GT (2012). "What is bioturbation? The need for a precise definition for fauna in aquatic sciences". Marine Ecology Progress Series. 446: 285–302. Bibcode: 2012MEPS..446..285K. doi: 10.3354/meps09506.
- ^ Villnäs, Anna; Norkko, Joanna; Hietanen, Susanna; Josefson, Alf B.; Lukkari, Kaarina; Norkko, Alf (2013). "The role of recurrent disturbances for ecosystem multifunctionality". Ecology. 94 (10): 2275–2287. doi: 10.1890/12-1716.1. hdl: 10138/233616. PMID 24358713.
- ^ Le Hir, P.; Monbet, Y.; Orvain, F. (2007). "Sediment erodability in sediment transport modelling: Can we account for biota effects?". Continental Shelf Research. 27 (8): 1116–1142. Bibcode: 2007CSR....27.1116L. doi: 10.1016/j.csr.2005.11.016.
- ^ Stief, P. (2013). "Stimulation of microbial nitrogen cycling in aquatic ecosystems by benthic macrofauna: Mechanisms and environmental implications". Biogeosciences. 10 (12): 7829–7846. Bibcode: 2013BGeo...10.7829S. doi: 10.5194/bg-10-7829-2013. hdl: 21.11116/0000-0001-C75E-6.
- ^ Aller, Robert C. (1982). "Carbonate Dissolution in Nearshore Terrigenous Muds: The Role of Physical and Biological Reworking". The Journal of Geology. 90 (1). University of Chicago Press: 79–95. Bibcode: 1982JG.....90...79A. doi: 10.1086/628652. ISSN 0022-1376. S2CID 129291436.
- ^ Barnosky, Anthony D.; Matzke, Nicholas; Tomiya, Susumu; Wogan, Guinevere O. U.; Swartz, Brian; Quental, Tiago B.; Marshall, Charles; McGuire, Jenny L.; Lindsey, Emily L.; Maguire, Kaitlin C.; Mersey, Ben; Ferrer, Elizabeth A. (2011). "Has the Earth's sixth mass extinction already arrived?". Nature. 471 (7336): 51–57. Bibcode: 2011Natur.471...51B. doi: 10.1038/nature09678. PMID 21368823. S2CID 4424650.
- ^ Chapin Iii, F. Stuart; Zavaleta, Erika S.; Eviner, Valerie T.; Naylor, Rosamond L.; Vitousek, Peter M.; Reynolds, Heather L.; Hooper, David U.; Lavorel, Sandra; Sala, Osvaldo E.; Hobbie, Sarah E.; Mack, Michelle C.; Díaz, Sandra (2000). "Consequences of changing biodiversity". Nature. 405 (6783): 234–242. doi: 10.1038/35012241. hdl: 11336/37401. PMID 10821284. S2CID 205006508.
- ^ Díaz, Sandra; Fargione, Joseph; Chapin, F. Stuart; Tilman, David (2006). "Biodiversity Loss Threatens Human Well-Being". PLOS Biology. 4 (8): e277. doi: 10.1371/journal.pbio.0040277. PMC 1543691. PMID 16895442.
- ^ Worm, Boris; Hilborn, Ray; Baum, Julia K.; Branch, Trevor A.; Collie, Jeremy S.; Costello, Christopher; Fogarty, Michael J.; Fulton, Elizabeth A.; Hutchings, Jeffrey A.; Jennings, Simon; Jensen, Olaf P.; Lotze, Heike K.; Mace, Pamela M.; McClanahan, Tim R.; Minto, Cóilín; Palumbi, Stephen R.; Parma, Ana M.; Ricard, Daniel; Rosenberg, Andrew A.; Watson, Reg; Zeller, Dirk (2009). "Rebuilding Global Fisheries". Science. 325 (5940): 578–585. Bibcode: 2009Sci...325..578W. doi: 10.1126/science.1173146. hdl: 11336/100063. PMID 19644114. S2CID 2805799.
- ^ Defeo, Omar; Castilla, Juan Carlos (2005). "More than One Bag for the World Fishery Crisis and Keys for Co-management Successes in Selected Artisanal Latin American Shellfisheries". Reviews in Fish Biology and Fisheries. 15 (3): 265–283. doi: 10.1007/s11160-005-4865-0. S2CID 6912211.
- ^ Pauly, Daniel; Zeller, Dirk (2016). "Catch reconstructions reveal that global marine fisheries catches are higher than reported and declining". Nature Communications. 7: 10244. Bibcode: 2016NatCo...710244P. doi: 10.1038/ncomms10244. PMC 4735634. PMID 26784963.
- ^ Defeo, Omar; Castrejón, Mauricio; Pérez-Castañeda, Roberto; Castilla, Juan C.; Gutiérrez, Nicolás L.; Essington, Timothy E.; Folke, Carl (2016). "Co-management in Latin American small-scale shellfisheries: Assessment from long-term case studies". Fish and Fisheries. 17: 176–192. doi: 10.1111/faf.12101.
- ^ Sustainable fisheries: Territorial Use Rights for Fishing (TURF) Programs Fishery Solutions Center, Environmental Defense Fund. Retrieved: 2 November 2021.
- ^ Christy, F.T. (1982) "Territorial use rights in marine fisheries: definitions and conditions", Food and Agriculture Organisation, Fishing Technical Paper 227. United Nations, Rome.
- ^ a b Gelcich, S.; Hughes, T. P.; Olsson, P.; Folke, C.; Defeo, O.; Fernandez, M.; Foale, S.; Gunderson, L. H.; Rodriguez-Sickert, C.; Scheffer, M.; Steneck, R. S.; Castilla, J. C. (2010). "Navigating transformations in governance of Chilean marine coastal resources". Proceedings of the National Academy of Sciences. 107 (39): 16794–16799. doi: 10.1073/pnas.1012021107. PMC 2947917. PMID 20837530.
- ^ Gelcich, S.; Cinner, J.; Donlan, CJ; Tapia-Lewin, S.; Godoy, N.; Castilla, JC (2017). "Fishers' perceptions on the Chilean coastal TURF system after two decades: Problems, benefits, and emerging needs". Bulletin of Marine Science. 93: 53–67. doi: 10.5343/bms.2015.1082.
- ^ a b Castilla, Juan Carlos; Gelcich, Stefan; Defeo, Omar (2007). "Successes, Lessons, and Projections from Experience in Marine Benthic Invertebrate Artisanal Fisheries in Chile". Fisheries Management. pp. 23–42. doi: 10.1002/9780470996072.ch2. ISBN 9780470996072.
- ^ Kéfi, Sonia; Berlow, Eric L.; Wieters, Evie A.; Joppa, Lucas N.; Wood, Spencer A.; Brose, Ulrich; Navarrete, Sergio A. (2015). "Network structure beyond food webs: Mapping non‐trophic and trophic interactions on Chilean rocky shores". Ecology. 96 (1): 291–303. doi: 10.1890/13-1424.1. PMID 26236914.
- ^ a b Pérez-Matus, A.; Ospina-Alvarez, A.; Camus, PA; Carrasco, SA; Fernandez, M.; Gelcich, S.; Godoy, N.; Ojeda, FP; Pardo, LM; Rozbaczylo, N.; Subida, MD; Thiel, M.; Wieters, EA; Navarrete, SA (2017). "Temperate rocky subtidal reef community reveals human impacts across the entire food web". Marine Ecology Progress Series. 567: 1–16. Bibcode: 2017MEPS..567....1P. doi: 10.3354/meps12057.
- ^ Pérez-Matus, Alejandro; Carrasco, Sergio A.; Gelcich, Stefan; Fernandez, Miriam; Wieters, Evie A. (2017). "Exploring the effects of fishing pressure and upwelling intensity over subtidal kelp forest communities in Central Chile". Ecosphere. 8 (5): e01808. doi: 10.1002/ecs2.1808.
- ^ Gelcich, Stefan; Fernández, Miriam; Godoy, Natalio; Canepa, Antonio; Prado, Luis; Castilla, Juan Carlos (2012). "Territorial User Rights for Fisheries as Ancillary Instruments for Marine Coastal Conservation in Chile". Conservation Biology. 26 (6): 1005–1015. doi: 10.1111/j.1523-1739.2012.01928.x. PMID 22971114. S2CID 12693228.
- ^ Oyanedel, Rodrigo; Keim, Andres; Castilla, Juan Carlos; Gelcich, Stefan (2018). "Illegal fishing and territorial user rights in Chile". Conservation Biology. 32 (3): 619–627. doi: 10.1111/cobi.13048. PMID 29114934. S2CID 3703022.
- ^ Donlan, C. Josh; Wilcox, Chris; Luque, Gloria M.; Gelcich, Stefan (2020). "Estimating illegal fishing from enforcement officers". Scientific Reports. 10 (1): 12478. Bibcode: 2020NatSR..1012478D. doi: 10.1038/s41598-020-69311-5. PMC 7385102. PMID 32719385.
- ^ Andreu-Cazenave, Miguel; Subida, Maria Dulce; Fernandez, Miriam (2017). "Exploitation rates of two benthic resources across management regimes in central Chile: Evidence of illegal fishing in artisanal fisheries operating in open access areas". PLOS ONE. 12 (6): e0180012. Bibcode: 2017PLoSO..1280012A. doi: 10.1371/journal.pone.0180012. PMC 5493345. PMID 28666013.
- ^ Castilla, Juan Carlos (1999). "Coastal marine communities: Trends and perspectives from human-exclusion experiments". Trends in Ecology & Evolution. 14 (7): 280–283. doi: 10.1016/s0169-5347(99)01602-x. PMID 10370266.
- ^
a
b
c
d
e
f El Mahrad, Badr; Newton, Alice; Icely, John; Kacimi, Ilias; Abalansa, Samuel; Snoussi, Maria (2020).
"Contribution of Remote Sensing Technologies to a Holistic Coastal and Marine Environmental Management Framework: A Review". Remote Sensing. 12 (14): 2313.
Bibcode:
2020RemS...12.2313E.
doi:
10.3390/rs12142313.
hdl:
10400.1/14723.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^
a
b
c
d
e
f Mariani, Patrizio; Bachmayer, Ralf; Kosta, Sokol; Pietrosemoli, Ermanno; Ardelan, Murat V.; Connelly, Douglas P.; Delory, Eric; Pearlman, Jay S.; Petihakis, George; Thompson, Fletcher; Crise, Alessandro (20 August 2021).
"Collaborative Automation and IoT Technologies for Coastal Ocean Observing Systems". Frontiers in Marine Science. 8. Frontiers Media SA.
doi:
10.3389/fmars.2021.647368.
hdl:
11250/3046028.
ISSN
2296-7745.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^ Small, Christopher and Nicholls, Robert J. (2003) "A global analysis of human settlement in coastal zones". Journal of Coastal Research, 19(3): 584-599
- ^ Balk, D., Montgomery, M.R., McGranahan, G., Kim, D., Mara, V., Todd, M., Buettner, T. and Dorélien, A. (2009) "Mapping urban settlements and the risks of climate change in Africa, Asia and South America". Population dynamics and climate change, 80: 103.
- ^ He, Qiang; Bertness, Mark D.; Bruno, John F.; Li, Bo; Chen, Guoqian; Coverdale, Tyler C.; Altieri, Andrew H.; Bai, Junhong; Sun, Tao; Pennings, Steven C.; Liu, Jianguo; Ehrlich, Paul R.; Cui, Baoshan (2015). "Economic development and coastal ecosystem change in China". Scientific Reports. 4: 5995. doi: 10.1038/srep05995. PMC 4125988. PMID 25104138.
- ^ El Mahrad, Badr; Abalansa, Samuel; Newton, Alice; Icely, John D.; Snoussi, Maria; Kacimi, Ilias (2020). "Social-Environmental Analysis for the Management of Coastal Lagoons in North Africa". Frontiers in Environmental Science. 8. doi: 10.3389/fenvs.2020.00037. hdl: 10400.1/13958.
- ^ Corvalán, C (2005). Ecosystems and human well-being : health synthesis : a report of the millennium ecosystem assessment. Geneva: World Health Organization. ISBN 978-92-4-156309-3. OCLC 560102324.
- ^ Kennedy, Robert E.; Townsend, Philip A.; Gross, John E.; Cohen, Warren B.; Bolstad, Paul; Wang, Y.Q.; Adams, Phyllis (2009). "Remote sensing change detection tools for natural resource managers: Understanding concepts and tradeoffs in the design of landscape monitoring projects". Remote Sensing of Environment. 113 (7): 1382–1396. Bibcode: 2009RSEnv.113.1382K. doi: 10.1016/j.rse.2008.07.018. S2CID 55168956.
- ^ Strong, James Asa; Elliott, Michael (2017). "The value of remote sensing techniques in supporting effective extrapolation across multiple marine spatial scales". Marine Pollution Bulletin. 116 (1–2): 405–419. Bibcode: 2017MarPB.116..405S. doi: 10.1016/j.marpolbul.2017.01.028. PMID 28118970.
- ^ Cristina, Sónia; Icely, John; Costa Goela, Priscila; Angel Delvalls, Tomás; Newton, Alice (2015). "Using remote sensing as a support to the implementation of the European Marine Strategy Framework Directive in SW Portugal". Continental Shelf Research. 108: 169–177. Bibcode: 2015CSR...108..169C. doi: 10.1016/j.csr.2015.03.011. hdl: 10400.1/11857. S2CID 140161355.
- ^ Hart, Jane K.; Martinez, Kirk (1 May 2015). "Toward an environmental Internet of Things". Earth and Space Science. 2 (5): 194–200. Bibcode: 2015E&SS....2..194H. doi: 10.1002/2014EA000044.
- ^ Lynch, Tim P.; Morello, Elisabetta B.; Evans, Karen; Richardson, Anthony J.; Rochester, Wayne; Steinberg, Craig R.; Roughan, Moninya; Thompson, Peter; Middleton, John F.; Feng, Ming; Sherrington, Robert; Brando, Vittorio; Tilbrook, Bronte; Ridgway, Ken; Allen, Simon; Doherty, Peter; Hill, Katherine; Moltmann, Tim C. (17 December 2014). Álvarez, Inés (ed.). "IMOS National Reference Stations: A Continental-Wide Physical, Chemical and Biological Coastal Observing System". PLOS ONE. 9 (12). Public Library of Science (PLoS): e113652. Bibcode: 2014PLoSO...9k3652L. doi: 10.1371/journal.pone.0113652. ISSN 1932-6203. PMC 4269483. PMID 25517905.
- ^ Moltmann, Tim; Turton, Jon; Zhang, Huai-Min; Nolan, Glenn; Gouldman, Carl; Griesbauer, Laura; Willis, Zdenka; Piniella, Ángel Muñiz; Barrell, Sue; Andersson, Erik; Gallage, Champika; Charpentier, Etienne; Belbeoch, Mathieu; Poli, Paul; Rea, Anthony; Burger, Eugene F.; Legler, David M.; Lumpkin, Rick; Meinig, Christian; O’Brien, Kevin; Saha, Korak; Sutton, Adrienne; Zhang, Dongxiao; Zhang, Yongsheng (28 June 2019). "A Global Ocean Observing System (GOOS), Delivered Through Enhanced Collaboration Across Regions, Communities, and New Technologies". Frontiers in Marine Science. 6. Frontiers Media SA. doi: 10.3389/fmars.2019.00291. ISSN 2296-7745.
- ^ Farcy, Patrick; Durand, Dominique; Charria, Guillaume; Painting, Suzanne J.; Tamminen, Timo; Collingridge, Kate; Grémare, Antoine J.; Delauney, Laurent; Puillat, Ingrid (20 September 2019). "Toward a European Coastal Observing Network to Provide Better Answers to Science and to Societal Challenges; The JERICO Research Infrastructure". Frontiers in Marine Science. 6. Frontiers Media SA. doi: 10.3389/fmars.2019.00529. hdl: 10138/348076. ISSN 2296-7745.
- ^ GOOS (2012). "Requirements for Global Implementation of the Strategic Plan for Coastal GOOS". Rep. Glob. Ocean Observ. Syst., 193: 200. Intergovernmental Oceanographic Commission, UNESCO.
- ^
a
b
c
d
e
f Szalaj, Dorota; Silva, Alexandra; Ré, Pedro; Cabral, Henrique (26 March 2021).
"Detecting Regime Shifts in the Portuguese Continental Shelf Ecosystem Within the Last Three Decades". Frontiers in Marine Science. 8. Frontiers Media SA.
doi:
10.3389/fmars.2021.629130.
ISSN
2296-7745.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^ a b Walker, Brian; Holling, C. S.; Carpenter, Stephen R.; Kinzig, Ann P. (2004). "Resilience, Adaptability and Transformability in Social-ecological Systems". Ecology and Society. 9 (2). doi: 10.5751/ES-00650-090205. hdl: 10535/3282.
- ^ May, Robert M. (1977). "Thresholds and breakpoints in ecosystems with a multiplicity of stable states". Nature. 269 (5628): 471–477. Bibcode: 1977Natur.269..471M. doi: 10.1038/269471a0. S2CID 4283750.
- ^ a b Scheffer, Marten; Carpenter, Steve; Foley, Jonathan A.; Folke, Carl; Walker, Brian (2001). "Catastrophic shifts in ecosystems". Nature. 413 (6856): 591–596. Bibcode: 2001Natur.413..591S. doi: 10.1038/35098000. PMID 11595939. S2CID 8001853.
- ^ Parsons, L.S; Lear, W.H (2001). "Climate variability and marine ecosystem impacts: A North Atlantic perspective". Progress in Oceanography. 49 (1–4): 167–188. Bibcode: 2001PrOce..49..167P. doi: 10.1016/S0079-6611(01)00021-0.
- ^ Daskalov, GM (2002). "Overfishing drives a trophic cascade in the Black Sea". Marine Ecology Progress Series. 225: 53–63. Bibcode: 2002MEPS..225...53D. doi: 10.3354/meps225053.
- ^ Llope, Marcos; Daskalov, Georgi M.; Rouyer, Tristan A.; Mihneva, Vesselina; Chan, Kung-SIK; Grishin, Alexander N.; Stenseth, Nils CHR. (2011). "Overfishing of top predators eroded the resilience of the Black Sea system regardless of the climate and anthropogenic conditions". Global Change Biology. 17 (3): 1251–1265. Bibcode: 2011GCBio..17.1251L. doi: 10.1111/j.1365-2486.2010.02331.x. PMC 3597262.
- ^ a b Lees, Kathryn; Pitois, Sophie; Scott, Catherine; Frid, Chris; MacKinson, Steven (2006). "Characterizing regime shifts in the marine environment". Fish and Fisheries. 7 (2): 104–127. doi: 10.1111/j.1467-2979.2006.00215.x.
- ^ a b McKinnell, S.M; Brodeur, R.D; Hanawa, K.; Hollowed, A.B; Polovina, J.J; Zhang, C.-I (2001). "An introduction to the Beyond el Niño conference: Climate variability and marine ecosystem impacts from the tropics to the Arctic". Progress in Oceanography. 49 (1–4): 1–6. Bibcode: 2001PrOce..49....1M. doi: 10.1016/S0079-6611(01)00012-X.
- ^ a b Cury, Philippe; Shannon, Lynne (2004). "Regime shifts in upwelling ecosystems: Observed changes and possible mechanisms in the northern and southern Benguela". Progress in Oceanography. 60 (2–4): 223–243. doi: 10.1016/j.pocean.2004.02.007.
- ^ Collie, Jeremy S.; Richardson, Katherine; Steele, John H. (2004). "Regime shifts: Can ecological theory illuminate the mechanisms?". Progress in Oceanography. 60 (2–4): 281–302. doi: 10.1016/j.pocean.2004.02.013.
- ^ Deyoung, B.; Harris, R.; Alheit, J.; Beaugrand, G.; Mantua, N.; Shannon, L. (2004). "Detecting regime shifts in the ocean: Data considerations". Progress in Oceanography. 60 (2–4): 143–164. doi: 10.1016/j.pocean.2004.02.017.
- ^ Wooster, Warren S.; Zhang, Chang Ik (2004). "Regime shifts in the North Pacific: Early indications of the 1976–1977 event". Progress in Oceanography. 60 (2–4): 183–200. doi: 10.1016/j.pocean.2004.02.005.
- ^ a b Heymans, Johanna J.; Tomczak, Maciej T. (2016). "Regime shifts in the Northern Benguela ecosystem: Challenges for management". Ecological Modelling. 331: 151–159. doi: 10.1016/j.ecolmodel.2015.10.027.
- ^ Weijerman, M.; Lindeboom, H.; Zuur, AF (2005). "Regime shifts in marine ecosystems of the North Sea and Wadden Sea". Marine Ecology Progress Series. 298: 21–39. Bibcode: 2005MEPS..298...21W. doi: 10.3354/meps298021.
- ^ Tomczak, Maciej T.; Heymans, Johanna J.; Yletyinen, Johanna; Niiranen, Susa; Otto, Saskia A.; Blenckner, Thorsten (2013). "Ecological Network Indicators of Ecosystem Status and Change in the Baltic Sea". PLOS ONE. 8 (10): e75439. Bibcode: 2013PLoSO...875439T. doi: 10.1371/journal.pone.0075439. PMC 3792121. PMID 24116045.
- ^ Cury, P. (2000). "Small pelagics in upwelling systems: Patterns of interaction and structural changes in "wasp-waist" ecosystems". ICES Journal of Marine Science. 57 (3): 603–618. Bibcode: 2000ICJMS..57..603C. doi: 10.1006/jmsc.2000.0712.
- ^ Wooster, W. S., Bakun, A., and McLain, R. M. (1976). "Seasonal upwelling cycle along the Eastern boundary of the North Atlantic". J. Mar. Res., 34: 131–141.
- ^ Fiúza, A. F. D., de Macedo, M. E., and Guerreiro, M. R. (1982). "Climatological space and time variation of the Portuguese coastal upwelling". Oceanol. Acta, 5: 31–40.
- ^ ICES (2008). ICES IBTSWG REPORT 2012, Report of the International Bottom Trawl Survey Working Group (IBTSWG). Lisbon: ICES, 27–30.
- ^ ICES (2017). Report of the Working Group on Southern Horse Mackerel, Anchovy and Sardine (WGHANSA), 24–29 June 2017. Bilbao: ICES.
- ^ Martins, Maria Manuel; Skagen, Dankert; Marques, Vítor; Zwolinski, Juan; Silva, Alexandra (2013). "Changes in the abundance and spatial distribution of the Atlantic chub mackerel (Scomber colias) in the pelagic ecosystem and fisheries off Portugal". Scientia Marina. 77 (4): 551–563. doi: 10.3989/scimar.03861.07B.
- ^ Garrido, S.; Silva, A.; Pastor, J.; Dominguez, R.; Silva, AV; Santos, AM (2015). "Trophic ecology of pelagic fish species off the Iberian coast: Diet overlap, cannibalism and intraguild predation". Marine Ecology Progress Series. 539: 271–285. Bibcode: 2015MEPS..539..271G. doi: 10.3354/meps11506. S2CID 86970659.
- ^ Leitão, F. (2015). "Landing profiles of Portuguese fisheries: Assessing the state of stocks". Fisheries Management and Ecology. 22 (2): 152–163. doi: 10.1111/fme.12112.
- ^ Leitão, F.; Alms, V.; Erzini, K. (2014). "A multi-model approach to evaluate the role of environmental variability and fishing pressure in sardine fisheries". Journal of Marine Systems. 139: 128–138. Bibcode: 2014JMS...139..128L. doi: 10.1016/j.jmarsys.2014.05.013. hdl: 10400.1/8855.
- ^ Leitão, Francisco (2015). "Time series analyses reveal environmental and fisheries controls on Atlantic horse mackerel (Trachurus trachurus) catch rates". Continental Shelf Research. 111: 342–352. Bibcode: 2015CSR...111..342L. doi: 10.1016/j.csr.2015.08.026.
- ^ Veiga-Malta, T.; Szalaj, D.; Angélico, MM; Azevedo, M.; Farias, I.; Garrido, S.; Lourenço, S.; Marçalo, A.; Marques, V.; Moreno, A.; Oliveira, PB; Paiva, VH; Prista, N.; Silva, C.; Sobrinho-Gonçalves, L.; Vingada, J.; Silva, A. (2019). "First representation of the trophic structure and functioning of the Portuguese continental shelf ecosystem: Insights into the role of sardine". Marine Ecology Progress Series. 617–618: 323–340. Bibcode: 2019MEPS..617..323V. doi: 10.3354/meps12724. S2CID 92274693.
- ^ Miranda, P. M. A.; Alves, J. M. R.; Serra, N. (2013). "Climate change and upwelling: Response of Iberian upwelling to atmospheric forcing in a regional climate scenario". Climate Dynamics. 40 (11–12): 2813–2824. Bibcode: 2013ClDy...40.2813M. doi: 10.1007/s00382-012-1442-9. S2CID 140670266.
- ^ Pires, Ana Cordeiro; Nolasco, Rita; Rocha, Alfredo; Dubert, Jesus (2013). "Assessing future climate change in the Iberian Upwelling System". Journal of Coastal Research. 165: 1909–1914. doi: 10.2112/SI65-323.1. S2CID 131408682.
- ^ Barton, E.D.; Field, D.B.; Roy, C. (2013). "Canary current upwelling: More or less?". Progress in Oceanography. 116: 167–178. Bibcode: 2013PrOce.116..167B. doi: 10.1016/j.pocean.2013.07.007. hdl: 10261/80552.
- ^ Sydeman, W. J.; García-Reyes, M.; Schoeman, D. S.; Rykaczewski, R. R.; Thompson, S. A.; Black, B. A.; Bograd, S. J. (2014). "Climate change and wind intensification in coastal upwelling ecosystems". Science. 345 (6192): 77–80. Bibcode: 2014Sci...345...77S. doi: 10.1126/science.1251635. PMID 24994651. S2CID 206555669.
- ^ a b Leitão, Francisco; Baptista, Vânia; Vieira, Vasco; Laginha Silva, Patrícia; Relvas, Paulo; Alexandra Teodósio, Maria (2019). "A 60-Year Time Series Analyses of the Upwelling along the Portuguese Coast". Water. 11 (6): 1285. doi: 10.3390/w11061285. hdl: 10400.1/12712.
- ^ Baptista, Vânia; Silva, Patrícia Laginha; Relvas, Paulo; Teodósio, M. Alexandra; Leitão, Francisco (2018). "Sea surface temperature variability along the Portuguese coast since 1950". International Journal of Climatology. 38 (3): 1145–1160. Bibcode: 2018IJCli..38.1145B. doi: 10.1002/joc.5231. S2CID 134746927.
- ^ Seto, Karen C.; Fragkias, Michail; Güneralp, Burak; Reilly, Michael K. (2011). "A Meta-Analysis of Global Urban Land Expansion". PLOS ONE. 6 (8): e23777. Bibcode: 2011PLoSO...623777S. doi: 10.1371/journal.pone.0023777. PMC 3158103. PMID 21876770.
- ^ Elliott, Michael; Mander, Lucas; Mazik, Krysia; Simenstad, Charles; Valesini, Fiona; Whitfield, Alan; Wolanski, Eric (2016). "Ecoengineering with Ecohydrology: Successes and failures in estuarine restoration". Estuarine, Coastal and Shelf Science. 176: 12–35. Bibcode: 2016ECSS..176...12E. doi: 10.1016/j.ecss.2016.04.003.
- ^ Waltham, Nathan J.; Sheaves, Marcus (2015). "Expanding coastal urban and industrial seascape in the Great Barrier Reef World Heritage Area: Critical need for coordinated planning and policy". Marine Policy. 57: 78–84. doi: 10.1016/j.marpol.2015.03.030.
- ^ Waycott, M.; Duarte, C. M.; Carruthers, T. J. B.; Orth, R. J.; Dennison, W. C.; Olyarnik, S.; Calladine, A.; Fourqurean, J. W.; Heck, K. L.; Hughes, A. R.; Kendrick, G. A.; Kenworthy, W. J.; Short, F. T.; Williams, S. L. (2009). "Accelerating loss of seagrasses across the globe threatens coastal ecosystems". Proceedings of the National Academy of Sciences. 106 (30): 12377–12381. Bibcode: 2009PNAS..10612377W. doi: 10.1073/pnas.0905620106. PMC 2707273. PMID 19587236.
- ^ a b c Murray, Nicholas J.; Worthington, Thomas A.; Bunting, Pete; Duce, Stephanie; Hagger, Valerie; Lovelock, Catherine E.; Lucas, Richard; Saunders, Megan I.; Sheaves, Marcus; Spalding, Mark; Waltham, Nathan J.; Lyons, Mitchell B. (13 May 2022). "High-resolution mapping of losses and gains of Earth's tidal wetlands". Science. 376 (6594): 744–749. Bibcode: 2022Sci...376..744M. doi: 10.1126/science.abm9583. hdl: 2160/55fdc0d4-aa3e-433f-8a88-2098b1372ac5. PMID 35549414. S2CID 248749118.
- ^ a b Valiela, Ivan; Bowen, Jennifer L.; York, Joanna K. (2001). "Mangrove Forests: One of the World's Threatened Major Tropical Environments". BioScience. 51 (10): 807. doi: 10.1641/0006-3568(2001)051[0807:MFOOTW]2.0.CO;2. ISSN 0006-3568. S2CID 4848686.
- ^ Thomas, Nathan; Lucas, Richard; Bunting, Peter; Hardy, Andrew; Rosenqvist, Ake; Simard, Marc (2017). "Distribution and drivers of global mangrove forest change, 1996–2010". PLOS ONE. 12 (6): e0179302. Bibcode: 2017PLoSO..1279302T. doi: 10.1371/journal.pone.0179302. PMC 5464653. PMID 28594908.
- ^ Atwood, Trisha B.; Connolly, Rod M.; Almahasheer, Hanan; Carnell, Paul E.; Duarte, Carlos M.; Ewers Lewis, Carolyn J.; Irigoien, Xabier; Kelleway, Jeffrey J.; Lavery, Paul S.; MacReadie, Peter I.; Serrano, Oscar; Sanders, Christian J.; Santos, Isaac; Steven, Andrew D. L.; Lovelock, Catherine E. (2017). "Global patterns in mangrove soil carbon stocks and losses". Nature Climate Change. 7 (7): 523–528. Bibcode: 2017NatCC...7..523A. doi: 10.1038/nclimate3326. hdl: 10072/346256.
- ^ Duarte, Carlos M.; Dennison, William C.; Orth, Robert J. W.; Carruthers, Tim J. B. (2008). "The Charisma of Coastal Ecosystems: Addressing the Imbalance". Estuaries and Coasts. 31 (2): 233–238. doi: 10.1007/s12237-008-9038-7. S2CID 86131712.
- ^
a
b
c
d
e
f Waltham, Nathan J.; Elliott, Michael; Lee, Shing Yip; Lovelock, Catherine; Duarte, Carlos M.; Buelow, Christina; Simenstad, Charles; Nagelkerken, Ivan; Claassens, Louw; Wen, Colin K-C; Barletta, Mario; Connolly, Rod M.; Gillies, Chris; Mitsch, William J.; Ogburn, Matthew B.; Purandare, Jemma; Possingham, Hugh; Sheaves, Marcus (2020).
"UN Decade on Ecosystem Restoration 2021–2030—What Chance for Success in Restoring Coastal Ecosystems?". Frontiers in Marine Science. 7.
doi:
10.3389/fmars.2020.00071.
hdl:
2440/123896.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^ The Global Mangrove Alliance Home page.
- ^ Lee, Shing Yip; Hamilton, Stu; Barbier, Edward B.; Primavera, Jurgenne; Lewis, Roy R. (29 April 2019). "Better restoration policies are needed to conserve mangrove ecosystems". Nature Ecology & Evolution. 3 (6). Springer Science and Business Media LLC: 870–872. doi: 10.1038/s41559-019-0861-y. ISSN 2397-334X. PMID 31036899. S2CID 139106235.
- ^ a b Magris, Rafael A.; Pressey, Robert L.; Weeks, Rebecca; Ban, Natalie C. (2014). "Integrating connectivity and climate change into marine conservation planning". Biological Conservation. 170: 207–221. doi: 10.1016/j.biocon.2013.12.032.
- ^ Álvarez-Romero, Jorge G.; Pressey, Robert L.; Ban, Natalie C.; Vance-Borland, Ken; Willer, Chuck; Klein, Carissa Joy; Gaines, Steven D. (2011). "Integrated Land-Sea Conservation Planning: The Missing Links". Annual Review of Ecology, Evolution, and Systematics. 42: 381–409. doi: 10.1146/annurev-ecolsys-102209-144702.
- ^ Grantham, Hedley S.; Game, Edward T.; Lombard, Amanda T.; Hobday, Alistair J.; Richardson, Anthony J.; Beckley, Lynnath E.; Pressey, Robert L.; Huggett, Jenny A.; Coetzee, Janet C.; Van Der Lingen, Carl D.; Petersen, Samantha L.; Merkle, Dagmar; Possingham, Hugh P. (2011). "Accommodating Dynamic Oceanographic Processes and Pelagic Biodiversity in Marine Conservation Planning". PLOS ONE. 6 (2): e16552. Bibcode: 2011PLoSO...616552G. doi: 10.1371/journal.pone.0016552. PMC 3032775. PMID 21311757.
- ^ Pressey, Robert L.; Cabeza, Mar; Watts, Matthew E.; Cowling, Richard M.; Wilson, Kerrie A. (2007). "Conservation planning in a changing world". Trends in Ecology & Evolution. 22 (11): 583–592. doi: 10.1016/j.tree.2007.10.001. PMID 17981360.
- ^ a b Weeks, Rebecca (2017). "Incorporating seascape connectivity in conservation prioritisation". PLOS ONE. 12 (7): e0182396. Bibcode: 2017PLoSO..1282396W. doi: 10.1371/journal.pone.0182396. PMC 5533427. PMID 28753647.
- ^ Almany, G. R.; Connolly, S. R.; Heath, D. D.; Hogan, J. D.; Jones, G. P.; McCook, L. J.; Mills, M.; Pressey, R. L.; Williamson, D. H. (2009). "Connectivity, biodiversity conservation and the design of marine reserve networks for coral reefs". Coral Reefs. 28 (2): 339–351. Bibcode: 2009CorRe..28..339A. doi: 10.1007/s00338-009-0484-x. S2CID 26332636.
- ^ Brown, Christopher J.; Harborne, Alastair R.; Paris, Claire B.; Mumby, Peter J. (2016). "Uniting paradigms of connectivity in marine ecology" (PDF). Ecology. 97 (9): 2447–2457. doi: 10.1002/ecy.1463. hdl: 10072/173575. PMID 27859092.
- ^ Cowen, Robert K.; Sponaugle, Su (2009). "Larval Dispersal and Marine Population Connectivity". Annual Review of Marine Science. 1: 443–466. Bibcode: 2009ARMS....1..443C. doi: 10.1146/annurev.marine.010908.163757. PMID 21141044.
- ^ Nagelkerken, Ivan (2009). Ecological connectivity among tropical coastal ecosystems. Dordrecht: Springer. ISBN 978-90-481-2406-0. OCLC 489216282.
- ^ Bode, Michael; Williamson, David H.; Weeks, Rebecca; Jones, Geoff P.; Almany, Glenn R.; Harrison, Hugo B.; Hopf, Jess K.; Pressey, Robert L. (2016). "Planning Marine Reserve Networks for Both Feature Representation and Demographic Persistence Using Connectivity Patterns". PLOS ONE. 11 (5): e0154272. Bibcode: 2016PLoSO..1154272B. doi: 10.1371/journal.pone.0154272. PMC 4864080. PMID 27168206.
- ^ Andrello, Marco; Jacobi, Martin Nilsson; Manel, Stéphanie; Thuiller, Wilfried; Mouillot, David (2015). "Extending networks of protected areas to optimize connectivity and population growth rate". Ecography. 38 (3): 273–282. doi: 10.1111/ecog.00975. S2CID 53605848.
- ^ White, JW; Botsford, LW; Hastings, A.; Largier, JL (2010). "Population persistence in marine reserve networks: Incorporating spatial heterogeneities in larval dispersal". Marine Ecology Progress Series. 398: 49–67. Bibcode: 2010MEPS..398...49W. doi: 10.3354/meps08327.
- ^ Grüss, Arnaud; Kaplan, David M.; Guénette, Sylvie; Roberts, Callum M.; Botsford, Louis W. (2011). "Consequences of adult and juvenile movement for marine protected areas". Biological Conservation. 144 (2): 692–702. doi: 10.1016/j.biocon.2010.12.015.
- ^
a
b
c Weeks, Rebecca (2017).
"Incorporating seascape connectivity in conservation prioritisation". PLOS ONE. 12 (7): e0182396.
Bibcode:
2017PLoSO..1282396W.
doi:
10.1371/journal.pone.0182396.
PMC
5533427.
PMID
28753647.
 Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a
Creative Commons Attribution 4.0 International License.
- ^ Adams, AJ; Dahlgren, CP; Kellison, GT; Kendall, MS; Layman, CA; Ley, JA; Nagelkerken, I.; Serafy, JE (2006). "Nursery function of tropical back-reef systems". Marine Ecology Progress Series. 318: 287–301. Bibcode: 2006MEPS..318..287A. doi: 10.3354/meps318287. hdl: 2066/35583. S2CID 55137201.
- ^ a b Ortiz, Delisse M.; Tissot, Brian N. (2012). "Evaluating ontogenetic patterns of habitat use by reef fish in relation to the effectiveness of marine protected areas in West Hawaii". Journal of Experimental Marine Biology and Ecology. 432–433: 83–93. doi: 10.1016/j.jembe.2012.06.005.
- ^ Pittman, Simon J.; Olds, Andrew D. (2015). "Seascape ecology of fishes on coral reefs". In Mora, Camilo (ed.). Ecology of Fishes on Coral Reefs. pp. 274–282. doi: 10.1017/CBO9781316105412.036. ISBN 9781316105412.
- ^ Unsworth, Richard K.F.; Cullen, Leanne C. (2010). "Recognising the necessity for Indo-Pacific seagrass conservation". Conservation Letters. 3 (2): 63–73. doi: 10.1111/j.1755-263X.2010.00101.x. S2CID 86729880.
- ^ Nagelkerken, Ivan; Sheaves, Marcus; Baker, Ronald; Connolly, Rod M. (2015). "The seascape nursery: A novel spatial approach to identify and manage nurseries for coastal marine fauna". Fish and Fisheries. 16 (2): 362–371. doi: 10.1111/faf.12057. hdl: 10072/60422.
- ^ Hamilton, Richard J.; Almany, Glenn R.; Stevens, Don; Bode, Michael; Pita, John; Peterson, Nate A.; Choat, J. Howard (2016). "Hyperstability masks declines in bumphead parrotfish (Bolbometopon muricatum) populations". Coral Reefs. 35 (3): 751–763. Bibcode: 2016CorRe..35..751H. doi: 10.1007/s00338-016-1441-0. S2CID 11931956.
- ^ Martin, TSH; Olds, AD; Pitt, KA; Johnston, AB; Butler, IR; Maxwell, PS; Connolly, RM (2015). "Effective protection of fish on inshore coral reefs depends on the scale of mangrove-reef connectivity". Marine Ecology Progress Series. 527: 157–165. Bibcode: 2015MEPS..527..157M. doi: 10.3354/meps11295. hdl: 10072/124967.
- ^ Olds, Andrew D.; Connolly, Rod M.; Pitt, Kylie A.; Maxwell, Paul S. (2012). "Habitat connectivity improves reserve performance". Conservation Letters. 5: 56–63. doi: 10.1111/j.1755-263X.2011.00204.x. S2CID 3958145.
- ^ a b c Olds, Andrew D.; Albert, Simon; Maxwell, Paul S.; Pitt, Kylie A.; Connolly, Rod M. (2013). "Mangrove-reef connectivity promotes the effectiveness of marine reserves across the western Pacific". Global Ecology and Biogeography. 22 (9): 1040–1049. doi: 10.1111/geb.12072. hdl: 10072/55829.
- ^ a b Nagelkerken, Ivan; Grol, Monique G. G.; Mumby, Peter J. (2012). "Effects of Marine Reserves versus Nursery Habitat Availability on Structure of Reef Fish Communities". PLOS ONE. 7 (6): e36906. Bibcode: 2012PLoSO...736906N. doi: 10.1371/journal.pone.0036906. PMC 3366965. PMID 22675474.
- ^ Olds, Andrew D.; Pitt, Kylie A.; Maxwell, Paul S.; Connolly, Rod M. (2012). "Synergistic effects of reserves and connectivity on ecological resilience". Journal of Applied Ecology. 49 (6): 1195–1203. doi: 10.1111/jpe.12002.
- ^ Lindenmayer, David; et al. (2007). "A checklist for ecological management of landscapes for conservation". Ecology Letters. 11 (1): 071010211025003––. doi: 10.1111/j.1461-0248.2007.01114.x. PMID 17927771.
Further reading
- Horn, Michael (1999). Intertidal fishes : life in two worlds. San Diego: Academic Press. ISBN 978-0-08-053493-0. OCLC 237380300.


![A shy but threatened dugong grazes a seagrass meadow, encouraging regrowth[22]](https://upload.wikimedia.org/wikipedia/commons/thumb/9/91/Dugong_Marsa_Alam.jpg/400px-Dugong_Marsa_Alam.jpg)

![Ecosystem services provided by a vegetated coastal ecosystem.[35] Diagram showing connectivity between a vegetated coastal ecosystem for the Penaeid prawn lifecycle indicating that valuations for harvest areas may overlook critical importance within the lifecycle.[35]](https://upload.wikimedia.org/wikipedia/commons/thumb/f/f1/Ecosystem_services_provided_by_a_vegetated_coastal_ecosystem.jpg/668px-Ecosystem_services_provided_by_a_vegetated_coastal_ecosystem.jpg)




![Pathways for guano-derived nitrogen to enter marine food webs[81]](https://upload.wikimedia.org/wikipedia/commons/thumb/1/12/Pathways_for_guano-derived_nitrogen_to_enter_marine_food_webs.webp/528px-Pathways_for_guano-derived_nitrogen_to_enter_marine_food_webs.webp.png)
![Seabird colonies are nutrient hot spots, especially, for nitrogen and phosphorus[65]](https://upload.wikimedia.org/wikipedia/commons/thumb/6/64/Seabird_ornitheutrophication_coupling.png/565px-Seabird_ornitheutrophication_coupling.png)
![Fish migrations between coral reef, macroalgae, seagrass and mangrove habitats:[133] (a) diel and tidal foraging migrations, (b) ontogenetic migration of juvenile coral reef fish.](https://upload.wikimedia.org/wikipedia/commons/thumb/3/3e/Fish_migrations_between_coral_reef%2C_macroalgae%2C_seagrass%2C_and_mangrove_habitats.jpg/575px-Fish_migrations_between_coral_reef%2C_macroalgae%2C_seagrass%2C_and_mangrove_habitats.jpg)
![The ecosystem cascade to structure the stock-taking and scenario analysis steps in the marine spatial planning process.[134]](https://upload.wikimedia.org/wikipedia/commons/thumb/f/f9/Ecosystem_cascade_for_structuring_marine_spatial_planning.jpg/618px-Ecosystem_cascade_for_structuring_marine_spatial_planning.jpg)