| |||
| |||
| Names | |||
|---|---|---|---|
|
IUPAC name
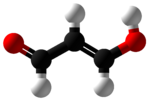
propanedial
| |||
| Other names
Malonic aldehyde; Malonodialdehyde; Propanedial; 1,3-Propanedial ; Malonaldehyde ; Malonyldialdehyde
| |||
| Identifiers | |||
3D model (
JSmol)
|
| ||
| Abbreviations | MDA | ||
| ChemSpider | |||
| KEGG | |||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H4O2 | |||
| Molar mass | 72.063 g·mol−1 | ||
| Appearance | Needle-like solid [1] | ||
| Density | 0.991 g/mL | ||
| Melting point | 72 °C (162 °F; 345 K) | ||
| Boiling point | 108 °C (226 °F; 381 K) | ||
| Hazards | |||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
none [1] | ||
REL (Recommended)
|
Ca [1] | ||
IDLH (Immediate danger)
|
Ca [N.D.] [1] | ||
| Related compounds | |||
Related alkenals
|
Glucic acid | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
Malondialdehyde belong to the class of β-dicarbonyls. A colorless liquid, malondialdehyde is a highly reactive compound that occurs as the enol. [2] It is a physiological metabolite, and a marker for oxidative stress.
Structure and synthesis
Malondialdehyde mainly exists as its enol, hydroxyacrolein: [2]
- CH2(CHO)2 → HOC(H)=CH-CHO
In organic solvents, the cis-isomer is favored, whereas in water the trans-isomer predominates. The equilibrium is rapid and is inconsequential for many purposes.
In the laboratory it can be generated in situ by hydrolysis of its acetal 1,1,3,3-tetramethoxypropane, which is commercially available and shelf-stable, unlike malondialdehyde. [2] Malondialdehyde is easily deprotonated to give the sodium salt of the enolate (m.p. 245 °C).
Biosynthesis and reactivity
Malondialdehyde results from lipid peroxidation of polyunsaturated fatty acids. [3] It is a prominent product in thromboxane A2 synthesis wherein cyclooxygenase 1 or cycloxygenase 2 metabolizes arachidonic acid to prostaglandin H2 by platelets and a wide array of other cell types and tissues. This product is further metabolized by thromboxane synthase to thromboxane A2, 12-hydroxyheptadecatrienoic acid, and malonyldialdehyde. Alternatively, it may rearrange non-enzymatically to a mixture of 8-cis and 8-trans isomers of 12-hydroxyeicosaheptaenoic acid plus malonyldialdehyde (see 12-Hydroxyheptadecatrienoic acid). [4] The degree of lipid peroxidation can be estimated by the amount of malondialdehyde in tissues. [3]
Reactive oxygen species degrade polyunsaturated lipids, forming malondialdehyde. [5] This compound is a reactive aldehyde and is one of the many reactive electrophile species that cause toxic stress in cells and form covalent protein adducts referred to as "advanced lipoxidation end-products" (ALE), in analogy to advanced glycation end-products (AGE). [6] The production of this aldehyde is used as a biomarker to measure the level of oxidative stress in an organism. [7] [8]
Malondialdehyde reacts with deoxyadenosine and deoxyguanosine in DNA, forming DNA adducts, the primary one being M1G, which is mutagenic. [9] The guanidine group of arginine residues condense with malondialdehyde to give 2-aminopyrimidines.
Human ALDH1A1 aldehyde dehydrogenase is capable of oxidizing malondialdehyde.
Analysis
Malondialdehyde and other thiobarbituric reactive substances (TBARS) condense with two equivalents of thiobarbituric acid to give a fluorescent red derivative that can be assayed spectrophotometrically. [2] [10] 1-Methyl-2-phenylindole is an alternative more selective reagent. [2]
Hazards and pathology
Malondialdehyde is reactive and potentially mutagenic. [11] It has been found in heated edible oils such as sunflower and palm oils. [12]
Corneas of patients with keratoconus and bullous keratopathy have increased levels of malondialdehyde, according to one study. [13] MDA also can be found in tissue sections of joints from patients with osteoarthritis. [14]
Levels of malondialdehyde can be also considered (as a marker of lipid peroxidation) to assess the membrane damage in spermatozoa; this is crucial because oxidative stress affects sperm function by altering membrane fluidity, permeability and impairing sperm functional competence. [15]
See also
References
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0377". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b c d e V. Nair, C. L. O'Neil, P. G. Wang "Malondialdehyde", Encyclopedia of Reagents for Organic Synthesis, 2008, John Wiley & Sons, New York. doi: 10.1002/047084289X.rm013.pub2 Article Online Posting Date: March 14, 2008
- ^
a
b Davey MW1, Stals E, Panis B, Keulemans J, Swennen RL (2005). "High-Throughput Determination of Malondialdehyde in Plant Tissues".
Analytical Biochemistry. 347 (2): 201–207.
doi:
10.1016/j.ab.2005.09.041.
PMID
16289006.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) CS1 maint: numeric names: authors list ( link) - ^ Rouzer, Carol A.; Marnett, Lawrence J. (2003). "Mechanism of Free Radical Oxygenation of Polyunsaturated Fatty Acids by Cyclooxygenases". Chemical Reviews. 103 (6): 2239–2304. doi: 10.1021/cr000068x. PMID 12797830.
- ^ Pryor WA, Stanley JP (1975). "Letter: A suggested mechanism for the production of malondialdehyde during the autoxidation of polyunsaturated fatty acids. Nonenzymatic production of prostaglandin endoperoxides during autoxidation". J. Org. Chem. 40 (24): 3615–7. doi: 10.1021/jo00912a038. PMID 1185332.
- ^ Farmer EE, Davoine C (2007). "Reactive electrophile species". Curr. Opin. Plant Biol. 10 (4): 380–6. doi: 10.1016/j.pbi.2007.04.019. PMID 17646124.
- ^ Moore K, Roberts LJ (1998). "Measurement of lipid peroxidation". Free Radic. Res. 28 (6): 659–71. doi: 10.3109/10715769809065821. PMID 9736317.
- ^ Del Rio D, Stewart AJ, Pellegrini N (2005). "A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress". Nutr Metab Cardiovasc Dis. 15 (4): 316–28. doi: 10.1016/j.numecd.2005.05.003. PMID 16054557.
- ^ Marnett LJ (1999). "Lipid peroxidation-DNA damage by malondialdehyde". Mutat. Res. 424 (1–2): 83–95. doi: 10.1016/S0027-5107(99)00010-X. PMID 10064852.
- ^ "Thiobarbituric acid reactive substances (TBARS) Assay". www.amdcc.org. Archived from the original on 14 September 2006.
- ^ Hartman PE, Putative mutagens and carcinogens in foods. IV. Malonaldehyde (malondialdehyde) Environ Mutagen. 1983;5(4):603-7
- ^ Dourerdjou, P.; Koner, B. C. (2008), Effect of Different Cooking Vessels on Heat-Induced Lipid Peroxidation of Different Edible Oils" Journal of Food Biochemistry, 32: 740–751. doi: 10.1111/j.1745-4514.2008.00195.x
- ^ Buddi R, Lin B, Atilano SR, Zorapapel NC, Kenney MC, Brown DJ (March 2002). "Evidence of oxidative stress in human corneal diseases". J. Histochem. Cytochem. 50 (3): 341–51. doi: 10.1177/002215540205000306. PMID 11850437.
- ^ Tiku ML, Narla H, Jain M, Yalamanchili P (2007). "Glucosamine prevents in vitro collagen degradation in chondrocytes by inhibiting advanced lipoxidation reactions and protein oxidation". Arthritis Research & Therapy. 9 (4): R76. doi: 10.1186/ar2274. PMC 2206377. PMID 17686167.
- ^ Collodel, G.; Moretti, E.; Micheli, L.; Menchiari, A.; Moltoni, L.; Cerretani, D. (March 2015). "Semen characteristics and malondialdehyde levels in men with different reproductive problems". Andrology. 3 (2): 280–286. doi: 10.1111/andr.297. PMID 25331426. S2CID 28027300.