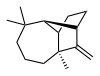
| |
| Names | |
|---|---|
|
IUPAC name
(1R,2S,7S,9S)- 3,3,7-trimethyl- 8-methylenetricyclo- [5.4.0.02,9]undecane
| |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| 5731712 2044263 4663756 | |
| ChEBI |
|
| ChemSpider | |
| ECHA InfoCard | 100.006.812 |
| EC Number |
|
PubChem
CID
|
|
| UNII |
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C15H24 | |
| Molar mass | 204.36 g/mol |
| Density | 0.928 g/cm3 |
| Boiling point | 254 °C (489 °F; 527 K) (706 mm Hg) |
| Hazards | |
| GHS labelling: | |



| |
| Danger | |
| H304, H317, H410 | |
| P261, P272, P273, P280, P301+P310, P302+P352, P321, P331, P333+P313, P363, P391, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Longifolene is the common (or trivial) chemical name of a naturally occurring, oily liquid hydrocarbon found primarily in the high-boiling fraction of certain pine resins. The name is derived from that of a pine species from which the compound was isolated, [1] Chemically, longifolene is a tricyclic sesquiterpene. This molecule is chiral, and the enantiomer commonly found in pines and other higher plants exhibits a positive optical rotation of +42.73°. The other enantiomer (optical rotation −42.73°) is found in small amounts in certain fungi and liverworts.
Longifolene is also one of two most abundant aroma constituents of lapsang souchong tea, because the tea is smoked over pinewood fires. [2]
Occurrence and syntheses
Terpentine obtained from Pinus longifolia (obsolete name for Pinus roxburghii Sarg.) contains as much as 20% of longifolene. [3]
The laboratory synthesis of longifolene has attracted much syntheses. [4] [5] [6] [7] [8] [9] [10]
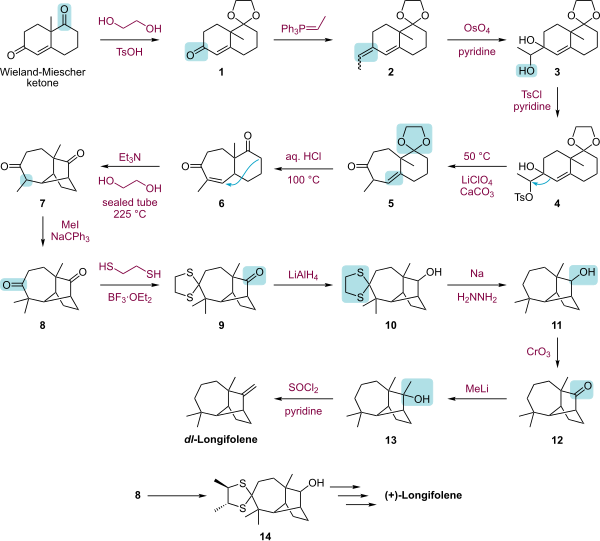 |
| Longifolene total synthesis by Corey.svg |
|---|
Biosynthesis
The biosynthesis of longifolene begins with farnesyl diphosphate (1) (also called farnesyl pyrophosphate) by means of a cationic polycyclization cascade. Loss of the pyrophosphate group and cyclization by the distal alkene gives intermediate 3, which by means of a 1,3-hydride shift gives intermediate 4. After two additional cyclizations, intermediate 6 produces longifolene by a 1,2-alkyl migration.

Reactions
It reacts with borane to give the derivative dilongifolylborane, which is a chiral hydroborating agent. [11]
References
- ^ Naffa, P.; Ourisson, G. Bulletin de la Société chimique de France, 1954, 1410.
- ^ Shan-Shan Yao; Wen-Fei Guo; YI Lu; Yuan-Xun Jiang, "Flavor Characteristics of Lapsang Souchong and Smoked Lapsang Souchong,a Special Chinese Black Tea with Pine Smoking Process", Journal of Agricultural and Food Chemistry, Vol. 53, No.22, (2005)[ permanent dead link]
- ^ Gscheidmeier, Manfred; Fleig, Helmut (2000). "Turpentines". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.a27_267. ISBN 978-3527306732.
- ^ Corey, E. J.; Ohno, Masaji.; Mitra, Rajat B.; Vatakencherry, Paul A. (February 1964). "Total Synthesis of Longifolene". Journal of the American Chemical Society. 86 (3): 478–485. doi: 10.1021/ja01057a039.
- ^ McMurry, John E.; Isser, Stephen J. (October 1972). "Total synthesis of longifolene". Journal of the American Chemical Society. 94 (20): 7132–7137. doi: 10.1021/ja00775a044.
- ^ Volkmann, Robert A.; Andrews, Glenn C.; Johnson, William S. (August 1975). "Novel Synthesis of Longifolene". Journal of the American Chemical Society. 97 (16): 4777–4779. doi: 10.1021/ja00849a062.
- ^ Oppolzer, Wolfgang; Godel, Thierry (April 1978). "A New and Efficient Total Synthesis of (.+-.)-longifolene". Journal of the American Chemical Society. 100 (8): 2583–2584. doi: 10.1021/ja00476a071.
- ^ Schultz, Arthur G.; Puig, Salvador (March 1985). "The Intramolecular Diene-Carbene Cycloaddition Equivalence and an Enantioselective Birch Reduction-Alkylation by the Chiral Auxiliary Approach. Total Synthesis of (.+-.)- and (−)-Longifolene". The Journal of Organic Chemistry. 50 (6): 915–916. doi: 10.1021/jo00206a049.
- ^ Bo, Lei; Fallis, Alex G. (May 1990). "Direct total synthesis of (+)-longifolene via an intramolecular Diels-Alder strategy". Journal of the American Chemical Society. 112 (11): 4609–4610. doi: 10.1021/ja00167a105.
- ^ Ho, Gregory J. Org. Chem. 2005, 70, 5139 -5143.
- ^ Dev, Sukh (1981). "Aspects of Longifolene chemistry. An example of another Facet of natural products chemistry". Accounts of Chemical Research. 14 (3): 82–88. doi: 10.1021/ar00063a004.