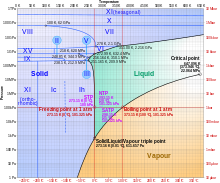
Ice VI is a form of ice that exists at high pressure at the order of about 1 GPa (= 10 000 bar) and temperatures ranging from 130 up to 355 Kelvin (−143 °C up to 82 °C); see also the phase diagram of water. Its discovery and the discovery of other high-pressure forms of water were published by P.W. Bridgman in January 1912. [1]
It is part of one of the inner layers of Titan. [2]
Properties
Ice VI has a density of 1.31 g/cm3 and a tetragonal crystal system with the space group P42/nmc; its unit cell contains 10 water molecules and has the dimensions a=6.27 Å and c=5.79 Å. [3] The triple point of ice VI with ice VII and liquid water is at about 82 °C and 2.22 GPa and its triple point with ice V and liquid water is at 0.16 °C and 0.6324 GPa = 6324 bar. [4]
Ice VI undergoes phase transitions into ices XV [5] and XIX [6] upon cooling depending on pressure as hydrochloric acid is doped.
See also
References
- ^ Water, in the Liquid and Five Solid Forms, under Pressure P.W. Bridgman (1912), www.jstor.org, retrieved 3 October 2019
- ^ "Titan - in Depth".
- ^ Reports: Structure of Ice VI science.sciencemag.org, B. Kamb, 8 October 1965.
- ^ Water Phase Diagram Archived 2016-03-14 at the Wayback Machine www1.lsbu.ac.uk, version of 9 September 2019, retrieved 3 October 2019
-
^ Salzmann CG, Radaelli PG, Mayer E, Finney JL (2009).
"Ice XV: a new thermodynamically stable phase of ice". Phys Rev Lett. 103 (10): 105701.
arXiv:
0906.2489.
Bibcode:
2009PhRvL.103j5701S.
doi:
10.1103/PhysRevLett.103.105701.
PMID
19792330.
S2CID
13999983.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ Yamane R, Komatsu K, Gouchi J, Uwatoko Y, Machida S, Hattori T, Kagi H; et al. (2021).
"Experimental evidence for the existence of a second partially-ordered phase of ice VI". Nat Commun. 12 (1): 1129.
Bibcode:
2021NatCo..12.1129Y.
doi:
10.1038/s41467-021-21351-9.
PMC
7893076.
PMID
33602936.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link)
External links
- Physik des Eises (PDF in German, iktp.tu-dresden.de)
- Ice phases (www.idc-online.com)
