This article has multiple issues. Please help
improve it or discuss these issues on the
talk page. (
Learn how and when to remove these template messages)
|
Geopolymers are inorganic, typically ceramic, materials that form long-range, covalently bonded, non-crystalline ( amorphous) networks. Geopolymers are a sub-class of alkali-activated cements. They are mainly produced by a chemical reaction between a chemically reactive aluminosilicate powder (e.g. metakaolin or other clay-derived powders, natural pozzolan, or suitable glasses), and an aqueous solution ( alkaline or acidic) that causes this powder to react and form into a solid monolith. The most common pathway to produce geopolymers is by the reaction of metakaolin with sodium silicate, which is an alkaline solution, but other processes are also possible.[ citation needed]
Commercially produced geopolymers may be used for fire- and heat-resistant coatings and adhesives, medicinal applications, high-temperature ceramics, new binders for fire-resistant fiber composites, toxic and radioactive waste encapsulation, and as cementing components in making concrete. The properties and uses of geopolymers are being explored in many scientific and industrial disciplines: modern inorganic chemistry, physical chemistry, colloid chemistry, mineralogy, geology, and in other types of engineering process technologies.
The original raw materials used in the synthesis of geopolymers were mainly rock-forming minerals of geological origin, hence the name: geopolymer was coined by Joseph Davidovits in 1978 [1] These materials and associated terminology were then popularized over the following decades via his work with the Institut Géopolymère (Geopolymer Institute).
One can distinguish between two synthesis routes, respectively:
- in alkaline medium (Na+, K+, Li+, Cs+, Ca2+…), and;
- in acidic medium ( phosphoric acid: H3PO4).
The alkaline route is the most important in terms of R&D and commercial applications and will be described below. Details on the acidic route have been published by Wagh in 2004, [2] by Perera et al. in 2008, [3] and by Cao et al. in 2005. [4]
What is a geopolymer?
In the 1950s, Viktor Glukhovsky developed concrete materials originally known under the names "soil silicate concretes" and "soil cements", [5] but since the introduction of the geopolymer concept by Joseph Davidovits, the terminology and definitions of the word geopolymer have become more diverse and often conflicting. The word geopolymer is sometimes used to refer to naturally occurring organic macromolecules; [6] that sense of the word differs from the now-more-common use of this terminology to discuss inorganic materials which can have either cement-like or ceramic-like character.
In the following presentation, a geopolymer is essentially a mineral chemical compound or mixture of compounds consisting of repeating units, for example silico-oxide (-Si-O-Si-O-), silico-aluminate (-Si-O-Al-O-), ferro-silico-aluminate (-Fe-O-Si-O-Al-O-) or alumino-phosphate (-Al-O-P-O-), created through a process of geopolymerization. [7] This method of describing mineral synthesis (geosynthesis) was first presented by Davidovits at an IUPAC symposium in 1976. [8]
Even within the context of inorganic materials, there exist various definitions of the word geopolymer, which can include (or not) a relatively wide variety of low-temperature synthesized solid materials. [9] The most typical geopolymer is generally described as resulting from the reaction between metakaolin (calcined kaolinitic clay) and a solution of sodium or potassium silicate ( waterglass). The chemical reaction of geopolymerization tends to result in a highly connected, disordered network of tetrahedral oxide units - which are silicate and aluminate tetrahedra in the metakaolin-waterglass example mentioned above - and with the net negative charges that are associated with aluminate tetrahedra being balanced by the sodium or potassium ions.
In the simplest form, an example chemical formula for a geopolymer can be written as Na2O·Al2O3·nSiO2·wH2O, where n is usually between 2 and 4, and w is around 11-15. Geopolymers can be formulated with a wide variety of substitutions in both the framework (Si,Al) and non-framework (Na) sites; most commonly K or Ca takes on the non-framework (Na) sites, or Fe or P can in principle replace some of the Al or Si.
Geopolymerization usually occurs at ambient or slightly elevated temperature; the solid aluminosilicate raw materials (e.g. metakaolin) dissolve into the alkaline solution, and then cross-link and polymerize into a growing gel phase, which then continues to set, harden and gain strength.
Geopolymer synthesis
Covalent bonding
The fundamental unit within a geopolymer structure is a tetrahedral complex consisting of Si or Al coordinated through covalent bonds to four oxygens. The geopolymer framework results from the cross-linking between these tetrahedra, which leads to a 3-dimensional aluminosilicate network, where the negative charge associated with tetrahedral aluminium is balanced by a small cationic species, most commonly an alkali metal cation. These alkali metal cations are often ion-exchangeable, as they are associated with, but only loosely bonded to, the main covalent network, similarly to the non-framework cations present in zeolites.
Geopolymerization starts with oligomers

Geopolymerization is the process of combining many small molecules known as oligomers into a covalently bonded network. This reaction process takes place via formation of oligomers (dimer, trimer, tetramer, pentamer) which are believed to contribute to the formation of the actual structure of the three-dimensional macromolecular framework, either through direct incorporation or through rearrangement via monomeric species. These oligomers are named by some geopolymer chemists as sialates following the scheme developed by Davidovits, [1] although this terminology is not universally accepted within the research community due in part to confusion with the earlier (1952) use of the same word to refer to the salts of the important biomolecule sialic acid. [10]
The image shows 5 examples of small oligomeric potassium aluminosilicate species (labelled in the diagram according to the poly(sialate) / poly(sialate-siloxo) nomenclature), which are key intermediates in potassium-based alumino-silicate geopolymerization. The aqueous chemistry of aluminosilicate oligomers is complex, [11] and also plays an important role in the discussion of zeolite synthesis, a process which has many details in common with geopolymerization.
Example of geopolymerization of a metakaolin precursor, in an alkaline medium [12]
The reaction process broadly involves four main stages:
- Alkaline hydrolysis of the layered structure of the calcined kaolinite;
- Formation of monomeric and oligomeric species;
- In the presence of waterglass (soluble potassium or sodium silicate), cyclic Al-Si structures can form (e.g. #5 in the figure), whereby the hydroxide is liberated by condensation reactions and can react again;
- Geopolymerization ( polycondensation) into polymeric 3D-networks.
The reaction processes involving other aluminosilicate precursors, e.g. low-calcium fly ash, crushed or synthetic glasses, or natural pozzolans, are broadly similar to the steps described above.
Geopolymer 3D-frameworks and water
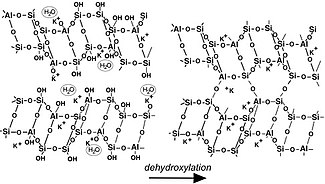
Geopolymerization forms aluminosilicate frameworks that are similar to those of some rock-forming minerals, but lacking in long-range crystalline order, and generally containing water in both chemically bound sites (hydroxyl groups) and in molecular form as pore water. This water can be removed at temperatures above 100 – 200 °C. Cation hydration and the locations and mobility of water molecules in pores are important for lower-temperature applications, such as in usage of geopolymers as cements. [13] [14] The figure shows a geopolymer containing both bound (Si-OH groups) and free water (left in the figure). Some water is associated with the framework similarly to zeolitic water, and some is in larger pores and can be readily released and removed. After dehydroxylation (and dehydration), generally above 250 °C, geopolymers can then crystallise above 800-1000 °C (depending on the nature of the alkali cation present). [15]
Commercial applications
There exist a wide variety of potential and existing applications. Some of the geopolymer applications are still in development whereas others are already industrialized and commercialized. See the incomplete list provided by the Geopolymer Institute. [16] They are listed in three major categories:
Geopolymer resins and binders
- Fire-resistant materials, thermal insulation, foams;
- Low-energy ceramic tiles, refractory items, thermal shock refractories;
- High-tech resin systems, paints, binders and grouts;
- Bio-technologies (materials for medicinal applications);
- Foundry industry (resins), tooling for the manufacture of organic fiber composites;
- Composites for infrastructures repair and strengthening, fire-resistant and heat-resistant high-tech carbon-fiber composites for aircraft interior and automobile;
- Radioactive and toxic waste containment.
Geopolymer cements and concretes
- Low-tech building materials (clay bricks);
- Low-CO2 cements and concretes.
Arts and archaeology
- Decorative stone artifacts, arts and decoration;
- Cultural heritage, archaeology and history of sciences.
Geopolymer cements
This article contains content that is written like
an advertisement. (November 2013) |
This article or section is in a state of significant expansion or restructuring. You are welcome to assist in its construction by editing it as well. If this article or section
has not been edited in several days, please remove this template. If you are the editor who added this template and you are actively editing, please be sure to replace this template with {{
in use}} during the active editing session. Click on the link for template parameters to use.
This article was
last edited by
Minh Duc le wiki (
talk |
contribs) 3 seconds ago. (
Update timer) |
From a terminological point of view, geopolymer cement [17] is a binding system that hardens at room temperature, like regular Portland cement.

Geopolymer cement is being developed and utilised as an alternative to conventional Portland cement for use in transportation, infrastructure, construction and offshore applications.[ citation needed]
Production of geopolymer cement requires an aluminosilicate precursor material such as metakaolin or fly ash, a user-friendly alkaline reagent [18][ promotional source?] (for example, sodium or potassium soluble silicates with a molar ratio (MR) SiO2:M2O ≥ 1.65, M being Na or K) and water (See the definition for "user-friendly" reagent below). Room temperature hardening is more readily achieved with the addition of a source of calcium cations, often blast furnace slag.[ citation needed]
Geopolymer cements can be formulated to cure more rapidly than Portland-based cements; some mixes gain most of their ultimate strength within 24 hours. However, they must also set slowly enough that they can be mixed at a batch plant, either for precasting or delivery in a concrete mixer. Geopolymer cement also has the ability to form a strong chemical bond with silicate rock-based aggregates.
There is often confusion between the meanings of the terms 'geopolymer cement' and 'geopolymer concrete'. A cement is a binder, whereas concrete is the composite material resulting from the mixing and hardening of cement with water (or an alkaline solution in the case of geopolymer cement), and stone aggregates. Materials of both types (geopolymer cements and geopolymer concretes) are commercially available in various markets internationally.
Alkali-activated materials vs. geopolymer cements
There exists some confusion in the terminology applied to geopolymers, alkali-activated cements and concretes, and related materials, whcih have been described by a variety of names including also "soil silicate concretes" and "soil cements". [5] Terminology related to alkali-activated materials or alkali-activated geopolymers is also in wide (but debated) use. These cements, sometimes abbreviated AAM, encompass the specific fields of alkali-activated slags, alkali-activated coal fly ashes, and various blended cementing systems (see RILEM Technical committee 247-DTA). [19]
User-friendly alkaline-reagents

Although geopolymerization does not rely on toxic organic solvents but only on water, it needs chemical ingredients that may be dangerous and therefore requires some safety procedures. Material Safety rules classify the alkaline products in two categories: corrosive products (named here: hostile) and irritant products (named here: friendly).[ citation needed] The two classes are recognizable through their respective logos.
The table lists some alkaline chemicals and their corresponding safety label. [20] The corrosive products must be handled with gloves, glasses and masks. They are user-hostile and cannot be implemented in mass applications without the appropriate safety procedures. In the second category one finds Portland cement or hydrated lime, typical mass products. Geopolymeric alkaline reagents belonging to this class may also be termed as User-friendly, although the irritant nature of the alkaline component and the potential inhalation risk of powders still require the selection and use of appropriate personal protective equipment, as in any situation where chemicals or powders are handled.
The development of so-called alkali-activated-cements or alkali-activated geopolymers (the latter considered by some to be incorrect terminology), as well as several recipes found in the literature and on the Internet, especially those based on fly ashes, use alkali silicates with molar ratios SiO2:M2O below 1.20, or systems based on pure NaOH (8M or 12M). These conditions are not user-friendly for the ordinary labor force, and require careful consideration of personal protective equipment if employed in the field. Indeed, laws, regulations, and state directives push to enforce for more health protections and security protocols for workers’ safety.
Conversely, Geopolymer cement recipes employed in the field generally involve alkaline soluble silicates with starting molar ratios ranging from 1.45 to 1.95, particularly 1.60 to 1.85, i.e. user-friendly conditions. It may happen that for research, some laboratory recipes have molar ratios in the 1.20 to 1.45 range.
Geopolymer cement categories
The categories comprise:
- Slag-based geopolymer cement; [21]
- Rock-based geopolymer cement; [22]
- Fly ash-based geopolymer cement:
- Ferro-sialate-based geopolymer cement. [27]
The first geopolymer cement developed in the 1980s was of the type (K,Na,Ca)-poly(sialate) (or slag-based geopolymer cement) and resulted from the research developments carried out by Joseph Davidovits and J.L. Sawyer at Lone Star Industries, USA and yielded the invention of Pyrament® cement. The American patent application was filed in 1984 and the patent US 4,509,985 was granted on April 9, 1985 with the title 'Early high-strength mineral polymer'.
In the 1990s, building on the works conducted on geopolymeric cements and on the synthesis of zeolites from fly ashes on the other hand, Wastiels et al. [28], Silverstrim et al. [29] and van Jaarsveld and van Deventer [30] developed geopolymeric fly ash-based cements.
Presently two types based on siliceous (EN 197) or class F (ASTM C618) fly ashes:
- Type 1: alkali-activated fly ash geopolymer:
- In many cases requires heat curing at 60-80°C; not manufactured separately as a cement, but rather produced directly as a fly-ash based concrete. NaOH + fly ash: partially-reacted fly ash particles embedded in an alumino-silicate gel with Si:Al= 1 to 2, zeolitic type ( chabazite-Na and sodalite) structures.
- Type 2: slag/fly ash-based geopolymer cement:
- Room-temperature cement hardening. User-friendly silicate solution + blast furnace slag + fly ash: fly ash particles embedded in a geopolymeric matrix with Si:Al ~ 2.
Ferro-sialate-based geopolymer cement The properties are similar to those of rock-based geopolymer cement but involve geological elements with high iron oxide content. The geopolymeric make up is of the type (Ca,K)-(Fe-O)-(Si-O-Al-O). This user-friendly geopolymer cement is in the development and commercialization phase.[ citation needed]
CO2 emissions during manufacturing
|
| This section needs expansion. You can help by
adding to it. (April 2024) |
Geopolymer cements may be able to be designed to have a lower attributed emission of carbon dioxide CO2 than some other widely-used materials. [31]
The need for standards
In June 2012, the institution ASTM International organized a symposium on Geopolymer Binder Systems. The introduction to the symposium states:[ citation needed] When performance specifications for Portland cement were written, non-portland binders were uncommon...New binders such as geopolymers are being increasingly researched, marketed as specialty products, and explored for use in structural concrete. This symposium is intended to provide an opportunity for ASTM to consider whether the existing cement standards provide, on the one hand, an effective framework for further exploration of geopolymer binders and, on the other hand, reliable protection for users of these materials.
The existing Portland cement standards are not adapted to geopolymer cements. They must be elaborated by an ad hoc committee. Yet, to do so, requires also the presence of standard geopolymer cements. Presently, every expert is presenting his own recipe based on local raw materials (wastes, by-products or extracted). There is a need for selecting the right geopolymer cement category. The 2012 State of the Geopolymer R&D, [32] suggested to select two categories, namely:
- type 2 slag/fly ash-based geopolymer cement: fly ashes are available in the major emerging countries;
- ferro-sialate-based geopolymer cement: this geological iron-rich raw material is present in all countries throughout the globe.
along with the appropriate user-friendly geopolymeric reagent.
Health effects
|
| This section needs expansion. You can help by
adding to it. (April 2024) |
Geopolymer applications to arts and archaeology
Because geopolymer artifacts can look like natural stone, several artists started to cast in silicone rubber molds replications of their sculptures. For example, in the 1980s, the French artist Georges Grimal worked on several geopolymer castable stone formulations. [33]
Egyptian pyramid stones
With respects to archaeological applications, in the mid-1980s, Joseph Davidovits presented his first analytical results carried out on genuine pyramid stones. He claimed that the ancient Egyptians knew how to generate a geopolymeric reaction in the making of a re-agglomerated limestone blocks. [34] The Ukrainian scientist G.V. Glukhovsky endorsed Davidovits' research in his keynote paper to the First Intern. Conf. on Alkaline Cements and Concretes, Kiev, Ukraine, 1994. [35] Later on, several materials scientists and physicists took over these archaeological studies and are publishing their results, essentially on pyramid stones. [36] [37] [38] [39]
Roman cements
From the digging of ancient Roman ruins, one knows that approximately 95% of the concretes and mortars constituting the Roman buildings consist of a very simple lime cement, which hardened slowly through the precipitating action of carbon dioxide CO2, from the atmosphere and formation of calcium silicate hydrate (C-S-H). This is a very weak to medium good material that was used essentially in the making of foundations and in buildings for the populace.
But for the building of their "ouvrages d’art", especially works related to water storage (cisterns, aqueducts), the Roman architects did not hesitate to use more sophisticated and expensive ingredients. These outstanding Roman cements are based on the calcic activation of ceramic aggregates (in Latin testa, analogue to our modern metakaolin MK-750) and alkali-rich volcanic tuffs (cretoni, zeolitic pozzolan), respectively with lime. MAS-NMR Spectroscopy investigations were carried out on these high-tech Roman cements dating to the 2nd century AD. They show their geopolymeric make-up. [40]
See also
References
- ^ a b An article published by the Commission of the European Communities in 1982, outlines the reasons why the generic term geopolymer was chosen for this new chemistry. See: J. Davidovits, The Need to Create a New Technical Language For the Transfer of Basic Scientific Information, in Transfer and Exploitation of Scientific and Technical Information, Proceedings of the symposium, Luxemburg, 10–12 June 1981, pp. 316-320. It is available as a pdf-file and may be downloaded from the European Parliament Bookshop. Go to < http://bookshop.europa.eu/en/transfer-and-exploitation-of-scientific-and-technical-information-pbCD3381271/> and click on 'download'.
- ^ Wagh, A.S. (2004). Chemically Bonded Phosphate Ceramics – A Novel Class of Geopolymers. Proceedings of the 106th annual meeting of the American Ceramic Society, Indianapolis. See also, Chapter 13, Phosphate-based Geopolymers, in J. Davidovits' book Geopolymer Chemistry and Applications.
- ^ Perera, D.S., Hanna, J.V., Davis, J., Blackford, M.G., Latella, B.A., Sasaki, Y. and Vance E.R. (2008). Relative strengths of phosphoric acid-reacted and alkali-reacted metakaolin materials. J. Mater. Sci., 43, 6562–6566.
- ^ Cao, D.; Su, D.; Lu, B. and Yang Y. (2005). Synthesis and structure characterization of geopolymeric material based on metakaolinite and phosphoric acid. Journal Chinese Ceramic Society, 33, 1385–89.
- ^ a b Gluchovskij V.D. (1959). "Gruntosilikaty" Gosstrojizdat Kiev, Patent USSR 245 627 (1967), Patent USSR 449894 (Patent appl. 1958, granted 1974).
- ^ Kim, D.; Lai, H.T.; Chilingar, G.V.; Yen T.F. (2006). Geopolymer formation and its unique properties, Environ. Geol, 51(1), 103–111.
- ^ See http://www.geopolymer.org/science/introduction
- ^ Pdf-file #20 Milestone paper IUPAC 76 at http://www.geopolymer.org/category/library/technical-papers
- ^ See, Discussion at the Geopolymer Camp 2012, video Geopolymer definition in Wikipedia at http://www.geopolymer.org/camp/gp-camp-2012.
- ^ Provis, J.L. and Van Deventer, J.S.J. (2009). Introduction to geopolymers, in: Geopolymers: Structure, Processing, Properties and Industrial Applications, J.L. Provis & Van Deventer (eds.), Woodhead, Cambridge UK, pp. 1–11
- ^ Swaddle, T.W., Salerno, J., and Tregloan, P.A. (1994). Aqueous aluminates, silicates, and aluminosilicates, Chemical Society Reviews, 23 (5), 319–325
- ^ See at http://www.geopolymer.org/science/about-geopolymerization
- ^ Barbosa, V.F.F; MacKenzie, K.J.D. and Thaumaturgo, C., (2000). Synthesis and characterization of materials based on inorganic polymers of alumina and silica: sodium polysialate polymers, International Journal of Inorganic Materials, 2, pp. 309–317.
- ^ Rowles, M.R. (2004). The Structural Nature of Aluminosilicate Inorganic Polymers: a Macro to Nanoscale Study, PhD Thesis, Curtin University of Technology, Perth, Australia.
- ^ Duxson, P.; Lukey, G.C., and van Deventer, J.S.J. (2007). The thermal evolution of metakaolin geopolymers: Part 2 – Phase stability and structural development, Journal of Non-Crystalline Solids, 353, pp. 2186-2200.
- ^ See at http://www.geopolymer.org/about/business-fellows
- ^ Davidovits, J., (1991). Geopolymers: Inorganic Polymeric New Materials, J. Thermal Analysis, 37, 1633–1656. See also Chapter 24 in Geopolymer Chemistry and Applications, Joseph Davidovits, Institut Géopolymère, Saint-Quentin, France, 2008, ISBN 9782951482050 (3rd ed., 2011).
- ^ See the examples at the Geopolymer Institute page http://www.geopolymer.org/applications/geopolymer-cement
- ^ http://www.rilem.org/gene/main.php?base=8750&gp_id=290
- ^ See in ref. 2
- ^ Davidovits, J. and Sawyer, J.L., (1985). Early high-strength mineral polymer, US Patent 4,509,985, 1985, filed February 22, 1984. The first commercial geopolymer cement was coined Pyrament 2000™ designed for repair and patching operations.
- ^ Gimeno, D.; Davidovits, J.; Marini, C.; Rocher, P.; Tocco, S.; Cara, S.; Diaz, N.; Segura, C. and Sistu, G. (2003). Development of silicate-based cement from glassy alkaline volcanic rocks: interpretation of preliminary data related to chemical- mineralogical composition of geologic raw materials. Paper in Spanish, Bol. Soc. Esp. Cerám. Vidrio, 42, 69–78. [Results from the European Research Project GEOCISTEM (1997), Cost Effective Geopolymeric Cements For Innocuous Stabilisation of Toxic Elements, Final Technical Report, April 30, 1997, Brussels, Project funded by the European Commission, Brite-Euram BE-7355-93, Jan. 1, 1994 to Feb. 28, 1997].
- ^ Palomo, A.; Grutzeck, M.W. and Blanco, M.T. (1999). Alkali-activated fly ashes: a cement for the future, Cement Concrete Res, 29, 1323–1329.
- ^ GEOASH (2004–2007), The GEOASH project was carried out with a financial grant from the Research Fund for Coal and Steel of the European Community, contract number RFC-CR-04005.
- ^ Izquierdo, M.; Querol, X.; Davidovits, J.; Antenucci, D.; Nugteren, H. and Fernández-Pereira, C., (2009). Coal fly ash-based geopolymers: microstructure and metal leaching, Journal of Hazardous Materials, 166, 561–566.
- ^ See: Chapter 12 in J. Davidovits' book Geopolymer Chemistry and Applications.
- ^ Davidovits, J. et al., Geopolymer cement of the Calcium-Ferroaluminium silicate polymer type and production process, PCT patent publication WO 2012/056125.
- ^ Wastiels J., Wu X., Faignet S., and Patfoort G. (1994). Mineral polymer based on fly ash, Journal of Resource Management and Technology, 22 (3), 135-141
- ^ Silverstrim, T.; Rostami, H.; Larralde, J.C and Samadi-Maybodi, A. (1997). Fly ash cementitious material and method of making a product, US Patent 5,601,643.
- ^ Van Jaarsveld, J.G.S., van Deventer, J.S.J. and Lorenzen L. (1997). The potential use of geopolymeric materials to immobilize toxic metals: Part I. Theory and Applications, Minerals Engineering, 10 (7), 659–669.
- ^ Rangan, B.V., (2008). Low-Calcium Fly Ash-Based Geopolymer Concrete, Chapter 26, in Concrete Construction Engineering Handbook, Editor-in-Chief E.G. Nawy, Second Edition, CRC Press, New York.
- ^ See the video at http://www.geopolymer.org/camp/gp-camp-2012
- ^ See Potential utilizations in art and decoration, at http://www.geopolymer.org/applications/potential-utilizations-in-art-and-decoration ; a pdf article #19 Dramatized sculptures with geopolymers at http://www.geopolymer.org/category/library/technical-papers/
- ^ Davidovits, J. (1986). X-Rays Analysis and X-Rays Diffraction of Casing Stones from the Pyramids of Egypt, and the Limestone of the Associated Quarries; pp. 511–20 in Science in Egyptology Symposia, Edited by R. A. David, Manchester University Press, Manchester, U.K. (Pdf-file #A in the Geopolymer Institute Library, Archaeological Papers); see also: Davidovits J., (1987). Ancient and modern concretes: what is the real difference? Concrete International: Des. Constr, 9 [12], 23–29. See also: Davidovits, J. and Morris, M., (1988). The Pyramids: An Enigma Solved. Hippocrene Books, New York, 1988.
- ^ G.V Glukhovsky passed away before the conference. His keynote paper titled: Ancient, Modern and Future Concretes, is included in the Proceedings of the First International Conference on Alkaline Cements and Concretes, pp. 1–9, Kiev, Ukraine, 1994.
- ^ Demortier, G. (2004). PIXE, PIGE and NMR study of the masonry of the pyramid of Cheops at Giza, Nuclear Instruments and Methods, Physics Research B, 226, 98–109.
- ^ Barsoum, M.W.; Ganguly, A. and Hug, G. (2006). Microstructural Evidence of Reconstituted Limestone Blocks in the Great Pyramids of Egypt, J. Am. Ceram. Soc. 89[12], 3788–3796.
- ^ MacKenzie, Kenneth J.D.; Smith, Mark E.; Wong, Alan; Hanna, John V.; Barry, Bernard and Barsoum, Michel W. (2011). Were the casing stones of Senefru's Bent Pyramid in Dahshour cast or carved? Multinuclear NMR evidence, Materials Letters 65, 350–352.
- ^ Túnyi, I. and El-hemaly, I. A. (2012). Paleomagnetic investigation of the great egyptian pyramids, Europhysics News 43/6, 28-31.
- ^ As part of the European research project GEOCISTEM [33], Davidovits J. and Davidovits F. sampled archaeological mortars and concretes dating back to the 2nd century AD and later, in Rome and Ostia, Italy. They selected two series of artifacts: Opus Signinum in Rome, Opus Caementicum / Testacaeum: mortars and concretes (carbunculus), in Ostia. Partly published in Geopolymer ’99 Proceedings, 283–295 and in Davidovits' book, Geopolymer Chemistry and Applications, Section 17.4. See also the NMR spectra at: http://www.geopolymer.org/applications/archaeological-analogues-roman-cements
External links
- Geopolymer science. Science Direct. Elsevier. 2024