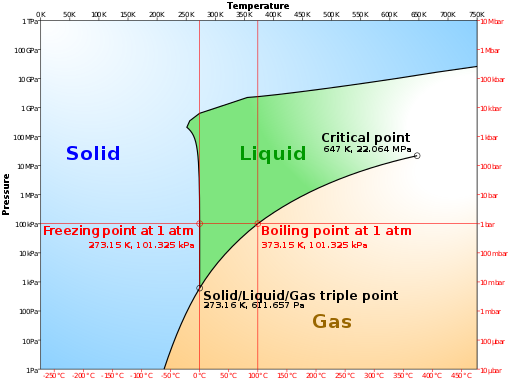
Size of this PNG preview of this SVG file:
512 × 379 pixels. Other resolutions:
320 × 237 pixels |
640 × 474 pixels |
1,024 × 758 pixels |
1,280 × 948 pixels |
2,560 × 1,895 pixels.
Original file (SVG file, nominally 512 × 379 pixels, file size: 34 KB)
File history
Click on a date/time to view the file as it appeared at that time.
| Date/Time | Thumbnail | Dimensions | User | Comment | |
|---|---|---|---|---|---|
| current | 20:41, 5 February 2023 |
 | 512 × 379 (34 KB) | N.Longo | File uploaded using svgtranslate tool (https://svgtranslate.toolforge.org/). Added translation for lij. |
| 11:38, 28 May 2022 |
 | 512 × 379 (22 KB) | Rainald62 | Reverted to version as of 22:28, 21 February 2021 (UTC) Vapour and Gas are not mutually exclusive. The dotted line is inappropriate (as is the sudden change green↔brown to the right of the critical point). Restoring also proper margins. | |
| 17:30, 12 February 2022 |
 | 512 × 724 (16 KB) | Germenfer | A substance in its gaseous state but below the critical temperature is called a "vapor", whereas a substance in its gaseous state but above the critical temperature is called a "gas". | |
| 22:28, 21 February 2021 |
 | 512 × 379 (22 KB) | TFerenczy | Maybe default last? | |
| 22:12, 21 February 2021 |
 | 512 × 379 (22 KB) | TFerenczy | +1 untranslated Czech, different declaration (fix?) | |
| 15:10, 18 February 2021 |
 | 512 × 379 (21 KB) | Egg | Added Czech translation | |
| 14:49, 18 February 2021 |
 | 512 × 379 (21 KB) | Egg | Wrong upload, sorry; Reverted to version as of 11:06, 12 February 2021 (UTC) | |
| 14:45, 18 February 2021 |
 | 512 × 432 (22 KB) | Egg | Added Czech translation | |
| 11:06, 12 February 2021 |
 | 512 × 379 (21 KB) | ApChrKey | Changed "vapour" to "gas". | |
| 18:32, 7 February 2021 |
 | 512 × 379 (21 KB) | Benjaminabel | Update french translation |
File usage
The following pages on the English Wikipedia use this file (pages on other projects are not listed):
Global file usage
The following other wikis use this file:
- Usage on cv.wikipedia.org
- Usage on de.wikipedia.org
- Usage on en.wikibooks.org
- Usage on en.wiktionary.org
- Usage on fa.wikipedia.org
- Usage on hi.wikipedia.org
- Usage on lij.wikipedia.org
- Usage on lv.wikipedia.org
- Usage on sl.wikipedia.org
- Usage on vi.wikipedia.org
