Size of this PNG preview of this SVG file:
374 × 342 pixels. Other resolutions:
263 × 240 pixels |
525 × 480 pixels |
840 × 768 pixels |
1,120 × 1,024 pixels |
2,240 × 2,048 pixels.
Original file (SVG file, nominally 374 × 342 pixels, file size: 47 KB)
File history
Click on a date/time to view the file as it appeared at that time.
| Date/Time | Thumbnail | Dimensions | User | Comment | |
|---|---|---|---|---|---|
| current | 16:43, 18 January 2009 |
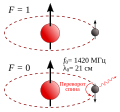
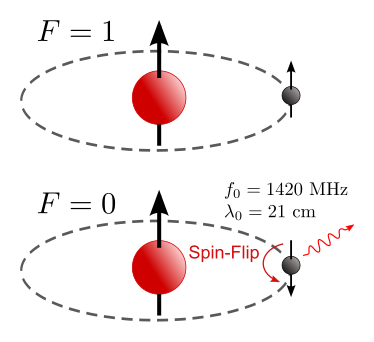
 | 374 × 342 (47 KB) | Tiltec~commonswiki | ({{Information |Description={{en|1=Ground state hyperfine levels of hydrogen (parallel and antiparallel) with the spin-flip transition, emitting radiation at 1420 MHz. The corresponding wavelength is 21 cm. (21-cm line, hydrogen line)}} {{de|1=Wasserstoff |
| 16:21, 18 January 2009 |
 | 374 × 342 (41 KB) | Tiltec~commonswiki | {{Information |Description={{en|1=Ground state hyperfine levels of hydrogen (parallel and antiparallel) with the spin-flip transition, emitting radiation at 1420 MHz. The corresponding wavelength is 21 cm. (21-cm line, hydrogen line)}} {{de|1=Wasserstoff |
File usage
The following pages on the English Wikipedia use this file (pages on other projects are not listed):
Global file usage
The following other wikis use this file:
- Usage on ar.wikipedia.org
- Usage on ca.wikipedia.org
- Usage on de.wikipedia.org
- Usage on de.wikibooks.org
- Usage on fr.wikipedia.org
- Usage on he.wikipedia.org
- Usage on ko.wikipedia.org
- Usage on lb.wikipedia.org
- Usage on pl.wikipedia.org
- Usage on sr.wikipedia.org
- Usage on tr.wikipedia.org
- Usage on uk.wikipedia.org
- Usage on www.wikidata.org
- Usage on zh.wikipedia.org