 | |
| Clinical data | |
|---|---|
| Trade names | Veozah, Veoza |
| Other names | ESN-364 |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a623051 |
| License data |
|
|
Pregnancy category |
|
|
Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 51% [5][ unreliable medical source?] |
| Metabolism | CYP1A2, ( CYP2C9, CYP2C19 to lesser extent) [2] |
| Metabolites | ES259564 [6] |
| Elimination half-life | 9.6h [2] |
| Excretion | Urine 76.9%, feces 14.7% [7][ unreliable medical source?] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
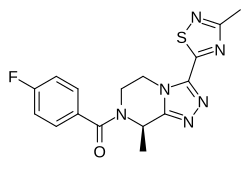
| Formula | C16H15FN6OS |
| Molar mass | 358.40 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Fezolinetant, sold under the brand name Veozah among others, is a medication used for the treatment of hot flashes (vasomotor symptoms) due to menopause. [2] [8] It is a small-molecule, orally active, selective neurokinin-3 (NK3) receptor antagonist which is under development by for the treatment of sex hormone-related disorders.[ medical citation needed] It is taken by mouth. [2] It is developed by Astellas Pharma which acquired it from Ogeda (formerly Euroscreen) in April 2017. [9] [10] [11]
The most common side effects include abdominal pain, diarrhea, insomnia, back pain, hot flush and elevated hepatic transaminases. [8]
Fezolinetant was approved for medical use in the United States in May 2023, [8] and in the European Union in December 2023. [3] [4] Fezolinetant is the first neurokinin 3 (NK3) receptor antagonist approved by the US Food and Drug Administration (FDA) to treat moderate to severe hot flashes from menopause. [8] The FDA considers it to be a first-in-class medication. [12]
Medical uses
Fezolinetant is indicated for the treatment of moderate to severe vasomotor symptoms due to menopause. [2] [3]
History
In May 2017, fezolinetant had completed phase I and phase IIa clinical trials for hot flashes in postmenopausal females. [11] Phase IIa trials in polycystic ovary syndrome patients are ongoing. [11]
In March 2023, results from SKYLIGHT 1, a Phase III clinical study of the treatment of moderate to severe hot flashes due to menopause, were published in The Lancet. [13] [14]
Fezolinetant shows high affinity for and potent inhibition of the NK3 receptor in vitro (Ki = 25 nM, IC50 = 20 nM). [10] Loss-of-function mutations in TACR and TACR3, the genes respectively encoding neurokinin B and its receptor, the NK3 receptor, have been found in patients with idiopathic hypogonadotropic hypogonadism. [10] In accordance, NK3 receptor antagonists like fezolinetant have been found to dose-dependently suppress luteinizing hormone (LH) secretion, though not that of follicle-stimulating hormone (FSH), and consequently to dose-dependently decrease estradiol and progesterone levels in females and testosterone levels in males. [15] As such, they are similar to GnRH modulators, and present as a potential clinical alternative to them for use in the same kinds of indications. [16] However, the inhibition of sex hormone production by NK3 receptor inactivation tends to be less complete and "non- castrating" relative to that of GnRH modulators, and so they may have a reduced incidence of menopausal-like side effects such as loss of bone mineral density. [15] [16]
Unlike GnRH modulators, but similarly to estrogens, NK3 receptor antagonists including fezolinetant and MLE-4901 (also known as AZD-4901, formerly AZD-2624) have been found to alleviate hot flashes in menopausal females. [17] [18] This would seem to be independent of their actions on the hypothalamic–pituitary–gonadal axis and hence on sex hormone production. [17] [18] NK3 receptor antagonists are anticipated as a useful clinical alternative to estrogens for management of hot flashes, but with potentially reduced risks and side effects. [17] [18]
The effectiveness of Veozah to treat moderate to severe hot flashes was demonstrated in each of the first 12-week, randomized, placebo-controlled, double-blind portions of two phase III clinical trials. [8] In both trials, after the first 12 weeks, the females on placebo were then re-randomized to Veozah for a 40-week extension study to evaluate safety. [8] Each trial ran a total of 52 weeks. [8] The average age of the trial participants was 54 years old. [8]
The FDA granted the application for fezolinetant priority review designation. [8] The approval of Veozah was granted to Astellas Pharma US, Inc. [8]
Society and culture
Legal status
In October 2023, the Committee for Medicinal Products for Human Use of the European Medicines Agency adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Veoza, intended for the treatment of hot flushes (vasomotor symptoms) associated with menopause. [3] The applicant for this medicinal product is Astellas Pharma Europe B.V. [3]
Fezolinetant was approved for medical use in the United States in May 2023, [8] and in the European Union in December 2023. [3] [4]
Brand names
Fezolinetant is the international nonproprietary name. [19]
Fezolinetant is sold under the brand names Veozah and Veoza. [2] [3]
References
- ^ a b "Veoza APMDS". Therapeutic Goods Administration (TGA). 6 March 2024. Retrieved 7 March 2024.
- ^ a b c d e f g "Veozah – fezolinetant tablet, film coated". DailyMed. 19 May 2023. Archived from the original on 25 May 2023. Retrieved 24 May 2023.
- ^ a b c d e f g "Veoza EPAR". European Medicines Agency (EMA). 7 December 2023. Archived from the original on 26 December 2023. Retrieved 27 December 2023. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ a b c "Veoza PI". Union Register of medicinal products. 12 December 2023. Archived from the original on 13 December 2023. Retrieved 26 December 2023.
- ^ "Fezolinetant". Archived from the original on 7 July 2023. Retrieved 7 July 2023.
- ^ "Pharmacokinetic Study of Fezolinetant - an Open-Label, Single and Multiple Dose Study to Evaluate the Pharmacokinetics and Safety of Fezolinetant in Healthy Chinese Female Subjects". 15 April 2022. Archived from the original on 7 July 2023. Retrieved 7 July 2023.
- ^ "Fezolinetant". DrugBank.com. Archived from the original on 7 July 2023. Retrieved 7 July 2023.
- ^
a
b
c
d
e
f
g
h
i
j
k
"FDA Approves Novel Drug to Treat Moderate to Severe Hot Flashes Caused by Menopause". U.S.
Food and Drug Administration (FDA) (Press release). 12 May 2023.
Archived from the original on 13 May 2023. Retrieved 13 May 2023.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^ "Astellas to Acquire Ogeda SA" (Press release). Astellas Pharma. Archived from the original on 5 November 2021. Retrieved 5 November 2021 – via PR Newswire.
- ^ a b c Hoveyda HR, Fraser GL, Dutheuil G, El Bousmaqui M, Korac J, Lenoir F, et al. (July 2015). "Optimization of Novel Antagonists to the Neurokinin‑3 Receptor for the Treatment of Sex-Hormone Disorders (Part II)". ACS Medicinal Chemistry Letters. 6 (7): 736–740. doi: 10.1021/acsmedchemlett.5b00117. PMC 4499830. PMID 26191358.
- ^ a b c "Fezolinetant – Ogeda". AdisInsight. Archived from the original on 5 November 2021. Retrieved 5 November 2021.
- ^ New Drug Therapy Approvals 2023 (PDF). U.S. Food and Drug Administration (FDA) (Report). January 2024. Archived from the original on 10 January 2024. Retrieved 9 January 2024.
- ^ Lederman S, Ottery FD, Cano A, Santoro N, Shapiro M, Stute P, et al. (April 2023). "Fezolinetant for treatment of moderate-to-severe vasomotor symptoms associated with menopause (SKYLIGHT 1): a phase 3 randomised controlled study". The Lancet. 401 (10382): 1091–1102. doi: 10.1016/S0140-6736(23)00085-5. PMID 36924778. S2CID 257498379.
- ^ "A Study to Find Out if Fezolinetant Helps Reduce Moderate to Severe Hot Flashes in Women Going Through Menopause (Skylight 1)". ClinicalTrials.gov. U.S. National Institutes of Health. 13 July 2022. Archived from the original on 29 March 2023. Retrieved 29 March 2023.
- ^ a b Fraser GL, Ramael S, Hoveyda HR, Gheyle L, Combalbert J (2016). "The NK3 Receptor Antagonist ESN364 Suppresses Sex Hormones in Men and Women". J. Clin. Endocrinol. Metab. 101 (2): 417–26. doi: 10.1210/jc.2015-3621. PMID 26653113.
- ^ a b Fraser GL, Hoveyda HR, Clarke IJ, Ramaswamy S, Plant TM, Rose C, et al. (2015). "The NK3 Receptor Antagonist ESN364 Interrupts Pulsatile LH Secretion and Moderates Levels of Ovarian Hormones Throughout the Menstrual Cycle". Endocrinology. 156 (11): 4214–25. doi: 10.1210/en.2015-1409. PMID 26305889.
- ^ a b c "Gone in a Flash: New Drug Class Targets Menopause Symptom". Medscape. Archived from the original on 12 May 2017. Retrieved 29 April 2017.
- ^ a b c "Ogeda Announces Positive Data From Phase IIa Trial Of Fezolinetant In The Treatment Of Menopausal Hot Flashes". www.clinicalleader.com. Archived from the original on 6 January 2017. Retrieved 29 April 2017.
- ^ World Health Organization (2017). "International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 77". WHO Drug Information. 31 (1). hdl: 10665/330984.