This article has multiple issues. Please help
improve it or discuss these issues on the
talk page. (
Learn how and when to remove these template messages)
|
| Exocrine pancreatic insufficiency | |
|---|---|
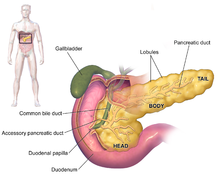 | |
| Anatomy of the pancreas | |
| Specialty | Endocrinology, gastroenterology |
| Causes | Diabetes (Type 2), Type 1 Diabetes, Pancreatitis, Celiac, Cystic Fibrosis, IBS-D, IBD, somatostatin analogues |
| Risk factors | older age, tobacco use, alcohol use |
| Diagnostic method | Fecal elastase test |
| Treatment | pancreatic enzyme replacement therapy (PERT) |
| Medication | pancrelipase |
Exocrine pancreatic insufficiency (EPI) is the inability to properly digest food due to a lack or reduction of digestive enzymes made by the pancreas. EPI can occur in humans and is prevalent in many conditions [1] such as cystic fibrosis, [2] Shwachman–Diamond syndrome, [3] different types of pancreatitis, [4] multiple types of diabetes mellitus ( Type 1 and Type 2 diabetes), [5] advanced renal disease, [6] older adults, [7] celiac disease, [8] IBS-D, [9] IBD, [10] HIV, [11] alcohol-related liver disease, [12] Sjogren syndrome, [13] tobacco use, [14] and use of somatostatin analogues. [15]
EPI is caused by a progressive loss of the pancreatic cells that make digestive enzymes. Loss of digestive enzymes leads to maldigestion and malabsorption of nutrients from normal digestive processes. EPI can cause symptoms even before reaching the stages of malnutrition: 'mild' or 'moderate' EPI is when fecal elastase levels are <200 ug/g, whereas 'severe' EPI is considered to be when fecal elastase levels is <100 ug/g. [16]
The exocrine pancreas is a portion of this organ that contains clusters of ducts (acini) producing bicarbonate anion, a mild alkali, as well as an array of digestive enzymes that together empty by way of the interlobular and main pancreatic ducts into the duodenum (upper small intestine). [17] The hormones cholecystokinin and secretin secreted by the stomach and duodenum in response to distension and the presence of food in turn stimulate the production of digestive enzymes by the exocrine pancreas. [18] The alkalization of the duodenum neutralizes the acidic chyme produced by the stomach that is passing into it; the digestive enzymes serve to catalyze the breakdown of complex foodstuffs into smaller molecules for absorption and integration into metabolic pathways. [18] The enzymes include proteases ( trypsinogen and chymotrypsinogen), hydrolytic enzymes that cleave lipids (the lipases phospholipase A2 and lysophospholipase, and cholesterol esterase), and amylase to digest starches. EPI results from progressive failure in the exocrine function of the pancreas to provide its digestive enzymes, often in response to a genetic condition or other disease state, resulting in the inability of the animal involved to properly digest food.[ citation needed]
Signs and symptoms
Loss of pancreatic enzymes leads to maldigestion and malabsorption. Other symptoms may include: [19]
- Abdominal discomfort or pain
- Bloating
- Diarrhea
- anemia ( Vitamin B12, iron, folate deficiency)
- bleeding disorders ( Vitamin K malabsorption)
- edema ( hypoalbuminemia)
- fatigue
- flatulence and abdominal distention (bacterial fermentation of unabsorbed food)
- hypocalcemia
- metabolic bone disease ( Vitamin D deficiency)
- neurologic manifestation
- steatorrhea
- weight loss
Causes
In humans, the most common causes of EPI are likely related to diabetes (10.5% global prevalence of diabetes, [20] with EPI rates of ranging from 30-50% in Type 1 and 20-30% of Type 2 [21]) and IBS-D (7.6-10.8% global prevalence of IBS-D, [22] with EPI rates around 5-6% [9]). Other causes of EPI include acute or chronic pancreatitis and cystic fibrosis, Crohn's disease, ulcerative colitis, celiac, advanced renal disease, older age, IBD, HIV, alcohol-related liver disease, Sjogren's syndrome, tobacco use, and use of somatostatin analogues.
EPI can also occur in 10-20% of the general population. [23] [7] [24]
Diagnosis
The three main tests used in considering a diagnosis of EPI are: fecal elastase test, fecal fat test, and a direct pancreatic function test. [25] The latter is a less used test that assesses exocrine function in the pancreas by inserting a tube into the small intestine to collect pancreatic secretions.
The fecal elastase test is a less cumbersome test that has replaced the 72-hour fecal fat test; in the fecal elastase test, pancreatic enzyme replacement therapy (enzyme supplementation, the treatment for EPI) does not have to be stopped for or during fecal elastase testing. [26]
Treatment
EPI is treated with pancreatic enzyme replacement therapy (PERT) called pancrelipase, which is used to break down fats (via a lipase), proteins (via a protease), and carbohydrates (via amylase) into units that can be digested. [27] Pancrelipase is typically porcine derived in the prescription products although over-the-counter options also exist, including those made with plants and other non-porcine materials. In the US, there are 6 FDA-approved PERT products available on the market as of 2012.
Dosing can vary based on the need of the individual. [28] PERT is considered to be safe, effective, and tolerable for people with EPI regardless of the cause of EPI. [29]
In addition, various nutrient deficiencies that can be caused by EPI need to be evaluated, tested, and treated. The impact of nutrient deficiencies on the body's metabolic pathways, muscle tissue, bone density, organs, and overall health can cause a wide range of often misdiagnosed symptoms for those impacted by exocrine pancreatic insufficiency. [30]
Other animals
EPI also occurs in dogs and other animals. Chronic pancreatitis is the most common cause of EPI in cats. In dogs, the most common cause is pancreatic acinar atrophy, arising as a result of genetic conditions, a blocked pancreatic duct, or prior infection.
Causes and pathogenesis
Chronic pancreatitis is the most common cause of EPI in cats.[ citation needed] In dogs, where the condition has been deemed common,[ citation needed] the usual cause is by pancreatic acinar atrophy (Which is a condition in which a normal pancreas undergoes atrophy. [31]), arising as a result of genetic conditions, a blocked pancreatic duct, or prior infection. [32]
In dogs, EPI is most common in young German Shepherds, and in Finland Rough Collies, [33] and is inherited. [34] In German Shepherds, the method of inheritance is through an autosomal recessive gene. [35] In these two breeds, at least, the cause appears to be immune-mediated as a sequela to lymphocytic pancreatitis. [36] The German Shepherd makes up about two-thirds of cases seen with EPI. [37] Other breeds reported to be predisposed to EPI include terrier breeds, Cavalier King Charles Spaniels, Chow Chows, [34] and Picardy Shepherds.
Symptoms
In animals, signs of EPI are not present until 85 to 90 percent of the pancreas is unable to secrete its enzymes. [32] In dogs, symptoms include weight loss, poor hair coat, flatulence, increased appetite, coprophagia, and diarrhea. Feces are often yellow-gray in color with an oily texture. There are many concurrent diseases that mimic EPI, and severe pancreatitis is one that if allowed to continue unabated can lead to EPI.[ citation needed]
Diagnosis and treatment
The most reliable test for EPI in dogs and cats is serum trypsin-like immunoreactivity (TLI); [38] a low value indicates EPI. Fecal elastase levels may also be used for diagnosis in dogs. [39]
In dogs, the best treatment is to supplement the animals' food with dried pancreatic extracts. There are commercial preparations available, but chopped bovine pancreas from the butcher can also be used. (Pork pancreas should not be used because of the rare transmission of pseudorabies). [40] Symptoms usually improve within a few days, but lifelong treatment is required to manage the condition. A rare side-effect of use of dried pancreatic extracts is oral ulceration and bleeding. [41]
Because of malabsorption, serum levels of cyanocobalamin (vitamin B12) and tocopherol (vitamin E) may be low. These may be supplemented, although since cyanocobalamin contains the toxic chemical cyanide, dogs that have serious cobalamin issues should instead be treated with hydroxocobalamin or methylcobalamin.[ citation needed]
Cyanocobalamin deficiency is very common in cats with EPI because about 99 percent of intrinsic factor (which is required for cyanocobalamin absorption from the intestine) is secreted by the pancreas. In dogs, this figure is about 90 percent, and only about 50 percent of dogs have this deficiency. [40]
Cats may suffer from Vitamin K deficiencies. If there is bacterial overgrowth in the intestine, antibiotics should be used, especially if treatment is not working.
In dogs failing to gain weight or continuing to show symptoms, modifying the diet to make it low-fiber and highly digestible may help. Despite previous belief that low-fat diets are beneficial in dogs with EPI, more recent studies have shown that a high-fat diet may increase absorption of nutrients and better manage the disease. [42] However, it has been shown that different dogs respond to different dietary modifications, so the best diet must be determined on a case-by-case basis. [43]
One possible sequela, volvulus (mesenteric torsion), is a rare consequence of EPI in dogs. [32]
References
- ^ Capurso, Gabriele; Traini, Mariaemilia; Piciucchi, Matteo; Signoretti, Marianna; Arcidiacono, Paolo Giorgio (2019-12-31). "Exocrine pancreatic insufficiency: prevalence, diagnosis, and management". Clinical and Experimental Gastroenterology. 12: 129–139. doi: 10.2147/CEG.S168266. PMC 6432881. PMID 30962702.
- ^ Ritivoiu, Mirela-Elena; Drăgoi, Cristina Manuela; Matei, Dumitru; Stan, Iustina Violeta; Nicolae, Alina Crenguţa; Craiu, Mihai; Dumitrescu, Ion-Bogdan; Ciolpan, Alina Angelica (2023-01-03). "Current and Future Therapeutic Approaches of Exocrine Pancreatic Insufficiency in Children with Cystic Fibrosis in the Era of Personalized Medicine". Pharmaceutics. 15 (1): 162. doi: 10.3390/pharmaceutics15010162. ISSN 1999-4923. PMC 9862205. PMID 36678791.
- ^ Nelson, Adam; Myers, Kasiani (1993), Adam, Margaret P.; Everman, David B.; Mirzaa, Ghayda M.; Pagon, Roberta A. (eds.), "Shwachman-Diamond Syndrome", GeneReviews®, Seattle (WA): University of Washington, Seattle, PMID 20301722, retrieved 2023-02-02
- ^ Ashraf, Hamza; Colombo, John Paul; Marcucci, Vincent; Rhoton, Jonathan; Olowoyo, Oluwatofunmi (2021-11-20). "A Clinical Overview of Acute and Chronic Pancreatitis: The Medical and Surgical Management". Cureus. 13 (11): e19764. doi: 10.7759/cureus.19764. ISSN 2168-8184. PMC 8684888. PMID 34938639.
- ^ Piciucchi, Matteo; Capurso, Gabriele; Archibugi, Livia; Delle Fave, Martina Maria; Capasso, Marina; Delle Fave, Gianfranco (2015). "Exocrine Pancreatic Insufficiency in Diabetic Patients: Prevalence, Mechanisms, and Treatment". International Journal of Endocrinology. 2015: 595649. doi: 10.1155/2015/595649. ISSN 1687-8337. PMC 4393909. PMID 25892991.
- ^ Griesche-Philippi, J.; Otto, J.; Schwörer, H.; Maisonneuve, P.; Lankisch, P.G. (2010-12-01). "Exocrine pancreatic function in patients with end-stage renal disease". Clinical Nephrology. 74 (12): 457–464. doi: 10.5414/CNP74457. ISSN 0301-0430. PMID 21084049.
- ^ a b Rothenbacher, Dietrich; Löw, Michael; Hardt, Philip D.; Klör, Hans-Ulrich; Ziegler, Hartwig; Brenner, Hermann (2005-06-01). "Prevalence and determinants of exocrine pancreatic insufficiency among older adults: Results of a population-based study". Scandinavian Journal of Gastroenterology. 40 (6): 697–704. doi: 10.1080/00365520510023116. ISSN 0036-5521. PMID 16036530. S2CID 25243765.
- ^ Balaban, Daniel Vasile; Enache, Iulia; Ciochina, Marina; Popp, Alina; Jinga, Mariana (2022-06-28). "Pancreatic involvement in celiac disease". World Journal of Gastroenterology. 28 (24): 2680–2688. doi: 10.3748/wjg.v28.i24.2680. ISSN 1007-9327. PMC 9260863. PMID 35979168.
- ^ a b Olmos, Juan I.; Piskorz, María M.; Litwin, Nestor; Schaab, Sara; Tevez, Adriana; Bravo-Velez, Gladys; Uehara, Tatiana; Hashimoto, Harumi; Rey, Enzo; Sorda, Juan A.; Olmos, Jorge A. (2022-12-01). "Exocrine Pancreatic Insufficiency is Undiagnosed in Some Patients with Diarrhea-Predominant Irritable Bowel Syndrome Using the Rome IV Criteria". Digestive Diseases and Sciences. 67 (12): 5666–5675. doi: 10.1007/s10620-022-07568-8. ISSN 1573-2568. PMID 35704255. S2CID 249676565.
- ^ Singh, Vikesh K.; Haupt, Mark E.; Geller, David E.; Hall, Jerry A.; Diez, Pedro M. Quintana (2017-10-21). "Less common etiologies of exocrine pancreatic insufficiency". World Journal of Gastroenterology. 23 (39): 7059–7076. doi: 10.3748/wjg.v23.i39.7059. PMC 5656454. PMID 29093615.
- ^ Yilmaz, Aylin; Hagberg, Lars (2018-03-04). "Exocrine pancreatic insufficiency is common in people living with HIV on effective antiretroviral therapy". Infectious Diseases. 50 (3): 193–199. doi: 10.1080/23744235.2017.1370126. ISSN 2374-4235. PMID 28838283. S2CID 4267100.
- ^ Leeds, John S.; Oppong, Kofi; Sanders, David S. (July 2011). "The role of fecal elastase-1 in detecting exocrine pancreatic disease". Nature Reviews Gastroenterology & Hepatology. 8 (7): 405–415. doi: 10.1038/nrgastro.2011.91. ISSN 1759-5045. PMID 21629239. S2CID 33640101.
- ^ Ebert, Ellen Catherine (January 2012). "Gastrointestinal and Hepatic Manifestations of Sjogren Syndrome". Journal of Clinical Gastroenterology. 46 (1): 25–30. doi: 10.1097/MCG.0b013e3182329d9c. ISSN 0192-0790. PMID 22157220.
- ^ Raphael, Kara L.; Chawla, Saurabh; Kim, Sungjin; Keith, Christopher G.; Propp, David R.; Chen, Zhengjia N.; Woods, Kevin E.; Keilin, Steven A.; Cai, Qiang; Willingham, Field F. (February 2017). "Pancreatic Insufficiency Secondary to Tobacco Exposure: A Controlled Cross-Sectional Evaluation". Pancreas. 46 (2): 237–243. doi: 10.1097/MPA.0000000000000721. ISSN 0885-3177. PMC 6355146. PMID 27846134.
- ^ Lamarca, Angela; McCallum, Lynne; Nuttall, Christina; Barriuso, Jorge; Backen, Alison; Frizziero, Melissa; Leon, Rebecca; Mansoor, Was; McNamara, Mairéad G; Hubner, Richard A.; Valle, Juan W (2018-07-03). "Somatostatin analogue-induced pancreatic exocrine insufficiency in patients with neuroendocrine tumors: results of a prospective observational study". Expert Review of Gastroenterology & Hepatology. 12 (7): 723–731. doi: 10.1080/17474124.2018.1489232. ISSN 1747-4124. PMID 29923433. S2CID 49317847.
- ^ Shandro, Benjamin M; Ritehnia, Jennifer; Chen, Joshua; Nagarajah, Rani; Poullis, Andrew (November 2020). "The investigation and management of pancreatic exocrine insufficiency: A retrospective cohort study". Clinical Medicine. 20 (6): 535–540. doi: 10.7861/clinmed.2020-0506. ISSN 1470-2118. PMC 7687321. PMID 33199316.
- ^ Pandol, Stephen J. (2010-01-01). Anatomy. Morgan & Claypool Life Sciences.
- ^ a b Pandol, Stephen J. (2010-01-01). Regulation of Whole-Organ Pancreatic Secretion. Morgan & Claypool Life Sciences.
- ^ Othman, Mohamed O.; Harb, Diala; Barkin, Jodie A. (February 2018). "Introduction and practical approach to exocrine pancreatic insufficiency for the practicing clinician". International Journal of Clinical Practice. 72 (2): e13066. doi: 10.1111/ijcp.13066. PMC 5873407. PMID 29405509.
- ^ Sun, Hong; Saeedi, Pouya; Karuranga, Suvi; Pinkepank, Moritz; Ogurtsova, Katherine; Duncan, Bruce B.; Stein, Caroline; Basit, Abdul; Chan, Juliana C. N.; Mbanya, Jean Claude; Pavkov, Meda E.; Ramachandaran, Ambady; Wild, Sarah H.; James, Steven; Herman, William H. (2022-01-01). "IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045". Diabetes Research and Clinical Practice. 183: 109119. doi: 10.1016/j.diabres.2021.109119. hdl: 20.500.11820/85919e8b-f73d-433d-a236-058a9561e24d. ISSN 0168-8227. PMID 34879977. S2CID 244927138.
- ^ Capurso, Gabriele; Traini, Mariaemilia; Piciucchi, Matteo; Signoretti, Marianna; Arcidiacono, Paolo Giorgio (2019-03-21). "Exocrine pancreatic insufficiency: prevalence, diagnosis, and management". Clinical and Experimental Gastroenterology. 12: 129–139. doi: 10.2147/CEG.S168266. PMC 6432881. PMID 30962702.
- ^ Oka, Priya; Parr, Heather; Barberio, Brigida; Black, Christopher J; Savarino, Edoardo V; Ford, Alexander C (2020-10-01). "Global prevalence of irritable bowel syndrome according to Rome III or IV criteria: a systematic review and meta-analysis". The Lancet Gastroenterology & Hepatology. 5 (10): 908–917. doi: 10.1016/S2468-1253(20)30217-X. ISSN 2468-1253. PMID 32702295. S2CID 220731155.
- ^ Campbell, Jennifer A.; Sanders, David S.; Francis, Katherine A.; Kurien, Matthew; Lee, Sai; Taha, Hatim; Ramadas, Arvind; Joy, Diamond; Hopper, Andrew D. (2016-09-01). "Should we Investigate Gastroenterology Patients for Pancreatic Exocrine Insufficiency? A Dual Centre UK Study". Journal of Gastrointestinal and Liver Diseases. 25 (3): 303–309. doi: 10.15403/jgld.2014.1121.253.uks. ISSN 1842-1121. PMID 27689193.
- ^ Herzig, Karl-Heinz; Purhonen, Anna-Kaisa; Räsänen, Kati M; Idziak, Joanna; Juvonen, Petri; Phillps, Ryszard; Walkowiak, Jaroslaw (December 2011). "Fecal pancreatic elastase-1 levels in older individuals without known gastrointestinal diseases or diabetes mellitus". BMC Geriatrics. 11 (1): 4. doi: 10.1186/1471-2318-11-4. ISSN 1471-2318. PMC 3039595. PMID 21266058.
- ^ Domínguez-Muñoz, JE (2011). "Pancreatic insufficiency: diagnosis and treatment". J Gastroenterol Hepatol. 26 (Supplement s2): 12–16. doi: 10.1111/j.1440-1746.2010.06600.x. PMID 21323992. S2CID 38770503.
- ^ Struyvenberg, Maarten R.; Martin, Camilia R.; Freedman, Steven D. (December 2017). "Practical guide to exocrine pancreatic insufficiency – Breaking the myths". BMC Medicine. 15 (1): 29, s12916–017–0783-y. doi: 10.1186/s12916-017-0783-y. ISSN 1741-7015. PMC 5301368. PMID 28183317.
- ^ "FDA rulemaking history of OTC EPI drug products". Fda.gov. Retrieved 2011-11-08.
- ^ Trang, Tony; Chan, Johanna; Graham, David Y (2014-09-07). "Pancreatic enzyme replacement therapy for pancreatic exocrine insufficiency in the 21st century". World Journal of Gastroenterology. 20 (33): 11467–11485. doi: 10.3748/wjg.v20.i33.11467. ISSN 1007-9327. PMC 4155341. PMID 25206255.
- ^ Gan, Can; Chen, Yan-Hua; Liu, Ling; Gao, Jin-Hang; Tong, Huan; Tang, Cheng-Wei; Liu, Rui (2017-10-07). "Efficacy and safety of pancreatic enzyme replacement therapy on exocrine pancreatic insufficiency: a meta-analysis". Oncotarget. 8 (55): 94920–94931. doi: 10.18632/oncotarget.21659. ISSN 1949-2553. PMC 5706924. PMID 29212278.
- ^ Toxopeus, A. (2019). Primal Pancreas - Pancreas Damage Survival Guide. Delft Consulting LLC. pp. 180–200. ISBN 978-0-9600679-1-6.
- ^ "Exocrine Pancreatic Insufficiency - an overview | ScienceDirect Topics". www.sciencedirect.com. Retrieved 2023-04-07.
- ^ a b c Ettinger, Stephen J.; Feldman, Edward C. (1995). Textbook of Veterinary Internal Medicine (4th ed.). W.B. Saunders Company. ISBN 0-7216-6795-3.
- ^ Westermarck, E.; Wiberg, M. (2003). "Exocrine pancreatic insufficiency in dogs". Vet Clin N Am Small Animal Pract. 33 (5): 1165–79, viii–ix. doi: 10.1016/S0195-5616(03)00057-3. PMID 14552166.
- ^ a b Hall, Edward J. (2003). "Exocrine Pancreatic Insufficiency". Proceedings of the 28th World Congress of the World Small Animal Veterinary Association. Retrieved 2007-02-24.
- ^ Clark, L.; Wahl, J.; Steiner, J.; Zhou, W.; Ji W.; Famula, T.; Williams, D.; Murphy, K. (2005). "Linkage analysis and gene expression profile of pancreatic acinar atrophy in the German Shepherd Dog". Mamm Genome. 16 (12): 955–62. doi: 10.1007/s00335-005-0076-1. PMID 16341675. S2CID 33039280.
- ^ Wiberg, M.; Saari, S.; Westermarck, E. (1999). "Exocrine pancreatic atrophy in German Shepherd Dogs and Rough-coated Collies: an end result of lymphocytic pancreatitis". Vet Pathol. 36 (6): 530–41. doi: 10.1354/vp.36-6-530. PMID 10568434. S2CID 31368693.
- ^ Kim, J.; Jung, D.; Kang, B.; Kim, H.; Park, C.; Park, E.; Lim C.; Park, H. (2005). "Canine exocrine pancreatic insufficiency treated with porcine pancreatic extract". J Vet Sci. 6 (3): 263–6. doi: 10.4142/jvs.2005.6.3.263. PMID 16131834.
- ^ Steiner, J.; Rutz, G.; Williams, D. (2006). "Serum lipase activities and pancreatic lipase immunoreactivity concentrations in dogs with exocrine pancreatic insufficiency". Am J Vet Res. 67 (1): 84–7. doi: 10.2460/ajvr.67.1.84. PMID 16426216.
- ^ Rallis, Timoleon S.; Adamama-Moraitou, K. (2004). "Exocrine Pancreatic Insufficiency in Dogs and Cats: An Update". Proceedings of the 29th World Congress of the World Small Animal Veterinary Association. Retrieved 2007-02-24.
- ^ a b "Exocrine Pancreatic Insufficiency". The Merck Veterinary Manual. 2006. Retrieved 2007-02-24.
- ^ Snead E (2006). "Oral ulceration and bleeding associated with pancreatic enzyme supplementation in a German shepherd with pancreatic acinar atrophy". Can Vet J. 47 (6): 579–82. PMC 1461413. PMID 16808232.
- ^ Biourge V, Fontaine J (2004). "Exocrine pancreatic insufficiency and adverse reaction to food in dogs: a positive response to a high-fat, soy isolate hydrolysate-based diet". J Nutr. 134 (8 Suppl): 2166S–2168S. doi: 10.1093/jn/134.8.2166S. PMID 15284428.
- ^ Westermarck, E.; Wiberg, M. (2006). "Effects of diet on clinical signs of exocrine pancreatic insufficiency in dogs". J Am Vet Med Assoc. 228 (2): 225–9. doi: 10.2460/javma.228.2.225. PMID 16426193.
