| |
| Names | |
|---|---|
|
Preferred IUPAC name
(2Z)-2,3-Diaminobut-2-enedinitrile | |
| Other names
2,3-Diaminomaleonitrile; Hydrogen cyanide tetramer
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.013.361 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C4H4N4 | |
| Molar mass | 108.104 g·mol−1 |
| Melting point | 178–179 °C (352–354 °F; 451–452 K) [1] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
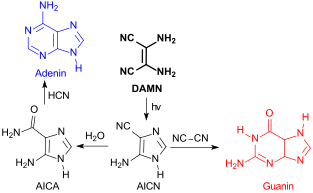
Diaminomaleonitrile (DAMN) is an organic compound composed of two amino groups and two nitrile groups bonded to a central alkene unit. The systematic name reflects its relationship to maleic acid. DAMN form by oligomerization of hydrogen cyanide. It is the starting point for the synthesis of several classes of heterocyclic compounds. It has been considered as a possible organic chemical present in prebiotic conditions. [2]
Isolation and synthesis
Diaminomaleonitrile was first isolated in 1873 as a black solid, when it was recognized as a polymer of hydrogen cyanide with the formula (HCN)x. [3] It was identified as the tetramer (HCN)4 by ebullioscopy in 1923. [4] The cis-configuration of the amino groups was shown in 1928 through reaction with glyoxal to give 2,3-diaminopyrazine, and the full structure was shown in 1955 to be diaminomaleonitrile, as opposed to the isomeric aminoiminosuccinonitrile (AISN). [5]
It can be prepared by cyanation of aminomalonitrile. [6] [7]
Possible role in abiogenesis
Diaminomaleonitrile has been proposed since the 1960s as a key substance for the prebiotic synthesis of nucleobases. Photochemical rearrangement of DAMN under UV light gives 4-aminoimidazole-5-carbonitrile (AICN), which can react further to form various nucleobases. [8] [9]
Early experiments have also suggested that certain amino acids, such as aspartic acid, alanine, and glycine, may have their probiotic origins in the acidic hydrolysis of diaminomaleonitrile. [10] [11]
Due to the ubiquity of hydrogen cyanide and its oligomers in space, it has been proposed that the dark material found in comets may consist of diaminomaleonitrile and higher oligomers, and that such polymers of HCN may have covered the surface of the early Earth. [12] [13]
References
- ^ "Diaminomaleonitrile". Sigma-Aldrich.
- ^ Al-Azmi, A.; Elassar, A.-Z. A.; Booth, B. L. (2003). "The Chemistry of Diaminomaleonitrile and its Utility in Heterocyclic Synthesis". Tetrahedron. 59 (16): 2749–2763. doi: 10.1016/S0040-4020(03)00153-4.
- ^ Lange, O. (1873). "Ueber eine neue Verbindung von der Zusammensetzung der Cyanwasserstoffsäure". Berichte der Deutschen Chemischen Gesellschaft. 6 (1): 99–101. doi: 10.1002/cber.18730060139. ISSN 0365-9496.
- ^ Bedel, C. (1923). "Sur un polymère de l'acide cyanhydrique". Comptes rendus hebdomadaires des séances de l'Académie des sciences: 168–171.
- ^ Webb, Richard L.; Frank, S.; Schneider, W. C. (1955). "The Structure of HCN Tetramer". Journal of the American Chemical Society. 77 (13): 3491–3493. doi: 10.1021/ja01618a021. ISSN 0002-7863.
- ^ Ferris, J. P.; Sanchez, R. A. (1968). "Diaminomaleonitrile (Hydrogen Cyanide Tetramer)". Organic Syntheses. 48: 60. doi: 10.15227/orgsyn.048.0060.
- ^ Ferris, J. P.; Sanchez, R. A.; Mancuso, R. W. (1968). "Aminomalonitrile p-Toluenesulfonate". Organic Syntheses. 48: 1. doi: 10.15227/orgsyn.048.0001.
- ^ Roy, Debjani; Najafian, Katayoun; von Ragué Schleyer, Paul (2007-10-30). "Chemical evolution: The mechanism of the formation of adenine under prebiotic conditions". Proceedings of the National Academy of Sciences. 104 (44): 17272–17277. doi: 10.1073/pnas.0708434104. ISSN 0027-8424. PMC 2077245. PMID 17951429.
- ^ Jeilani, Yassin Aweis; Williams, Phoenix N.; Walton, Sofia; Nguyen, Minh Tho (2016). "Unified reaction pathways for the prebiotic formation of RNA and DNA nucleobases". Physical Chemistry Chemical Physics. 18 (30): 20177–20188. doi: 10.1039/C6CP02686A. ISSN 1463-9076. PMID 27220279.
- ^ Moser, R.E.; Claggett, A.R.; Matthews, C.N. (1968). "Peptide formation from diaminomaleonitrile (hcn tetramer)". Tetrahedron Letters. 9 (13): 1599–1603. doi: 10.1016/S0040-4039(01)99011-2. PMID 5640452.
- ^ Matthews, Clifford N. (1995). "Hardware and software in biology: Simultaneous origin of proteins and nucleic acids via hydrogen cyanide polymers". Journal of Biological Physics. 20 (1–4): 275–281. doi: 10.1007/BF00700445. ISSN 0092-0606. S2CID 84612046.
- ^ Matthews, Clifford N. (1991). "Dark matter in the solar system: Hydrogen cyanide polymers". Origins of Life and Evolution of the Biosphere. 21 (5–6): 421–434. doi: 10.1007/BF01808312. ISSN 0169-6149. S2CID 2161383.
- ^ Matthews, Clifford N.; Minard, Robert D. (2006). "Hydrogen cyanide polymers, comets and the origin of life". Faraday Discussions. 133: 393–401, discussion 427-52. doi: 10.1039/b516791d. ISSN 1359-6640. PMID 17191459.