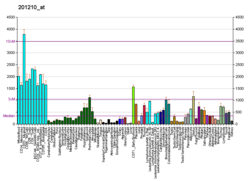
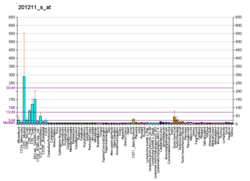
ATP-dependent RNA helicase DDX3X is an enzyme that in humans is encoded by the DDX3X gene. [5] [6] [7]
Function
DEAD box proteins, characterized by the conserved motif Asp-Glu-Ala-Asp (DEAD), are putative RNA helicases. They are implicated in a number of cellular processes involving alteration of RNA secondary structure such as translation initiation, nuclear and mitochondrial splicing, and ribosome and spliceosome assembly. Based on their distribution patterns, some members of this family are believed to be involved in embryogenesis, spermatogenesis, and cellular growth and division. This gene encodes a DEAD box protein, which interacts specifically with hepatitis C virus core protein resulting a change in intracellular location. This gene has a homolog located in the nonrecombining region of the Y chromosome. The protein sequence is 91% identical between this gene and the Y-linked homolog. [7]
Sub-cellular trafficking
DDX3X performs its functions in the cell nucleus and cytoplasm, exiting the nucleus via the exportin-1/CRM1 nuclear export pathway. It was initially reported that the DDX3X helicase domain was necessary for this interaction, while the canonical features of the trafficking pathway, namely the presence of a nuclear export signal (NES) on DDX3X and Ran-GTP binding to exportin-1, were dispensable. [8] DDX3X binding to, and trafficking by, exportin-1 has since been shown not to require the DDX3X helicase domain and be explicitly NES- and Ran-GTP-dependent. [9]
Role in cancer
DDX3X is involved in many different types of cancer. For example, it is abnormally expressed in breast epithelial cancer cells in which its expression is activated by HIF1A during hypoxia. [10] Increased expression of DDX3X by HIF1A in hypoxia is initiated by the direct binding of HIF1A to the HIF1A response element, [10] as verified with chromatin immunoprecipitation and luciferase reporter assay. Since the expression of DDX3X is affected by the activity of HIF1A, the co-localization of these proteins has also been demonstrated in MDA-MB-231 xenograft tumor samples. [10]
In HeLa cells DDX3X is reported to control cell cycle progression through Cyclin E1. [11] More specifically, DDX3X was shown to directly bind to the 5´ UTR of Cyclin E1 and thereby facilitating the translation of the protein. Increased protein levels of Cyclin E1 was demonstrated to mediate the transition of S phase entry. [11]
Melanoma survival, migration and proliferation is affected by DDX3X activity. [12] Melanoma cells with low DDX3X expression exhibit a high migratory capacity, low proliferation rate and reduced vemurafenib sensitivity. While high DDX3X expressing cells are drug sensitive, more proliferative and less migratory. These phenotypes can be explained by the translational effects on the melanoma transcription factor MITF. [12] The 5' UTR of the MITF mRNA contains a complex RNA regulon ( IRES) that is bound and activated by DDX3X. Activation of the IRES leads to translation of the MITF mRNA. Mice injected with melanoma cells with a deleted IRES display more aggressive tumor progression including increased lung metastasis. [12] Interestingly, the DDX3X in melanoma is affected by vemurafenib via an undiscovered mechanism. It is unknown how DDX3X is downregulated by the presence of vemurafenib. However, reduced levels of DDX3X during drug treatment explains the development of drug resistant cells frequently detected with low MITF expression. [12] [13] [14]
Clinical significance
Mutations of the DDX3X gene are associated with medulloblastoma. [15] [16] [17] In melanoma the low expression of the gene is linked to a poor distant metastasis free survival. [12] In addition, the mRNA level of DDX3X is lower in matched post-relapse melanoma biopsies for patients receiving vemurafenib and in progressing tumors.
Mutations of the DDX3X gene also cause DDX3X syndrome, which affects predominantly females and presents with developmental delay or disability, autism, ADHD, and low muscle tone.
See also
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000215301 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000000787 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Lahn BT, Page DC (October 1997). "Functional coherence of the human Y chromosome". Science. 278 (5338): 675–80. Bibcode: 1997Sci...278..675L. doi: 10.1126/science.278.5338.675. PMID 9381176.
- ^ Park SH, Lee SG, Kim Y, Song K (Oct 1998). "Assignment of a human putative RNA helicase gene, DDX3, to human X chromosome bands p11.3→p11.23". Cytogenetics and Cell Genetics. 81 (3–4): 178–9. doi: 10.1159/000015022. PMID 9730595. S2CID 46774908.
- ^ a b "Entrez Gene: DDX3X DEAD (Asp-Glu-Ala-Asp) box polypeptide 3, X-linked".
- ^ Yedavalli VS, Neuveut C, Chi YH, Kleiman L, Jeang KT (October 2004). "Requirement of DDX3 DEAD box RNA helicase for HIV-1 Rev-RRE export function". Cell. 119 (3): 381–92. doi: 10.1016/j.cell.2004.09.029. PMID 15507209.
- ^ Heaton SM, Atkinson SC, Sweeney MN, Yang SN, Jans DA, Borg NA (September 2019). "Exportin-1-Dependent Nuclear Export of DEAD-box Helicase DDX3X is Central to its Role in Antiviral Immunity". Cells. 8 (10): 1181. doi: 10.3390/cells8101181. PMC 6848931. PMID 31575075.
- ^ a b c Botlagunta M, Krishnamachary B, Vesuna F, Winnard PT, Bol GM, Patel AH, et al. (March 2011). "Expression of DDX3 is directly modulated by hypoxia inducible factor-1 alpha in breast epithelial cells". PLOS ONE. 6 (3): e17563. Bibcode: 2011PLoSO...617563B. doi: 10.1371/journal.pone.0017563. PMC 3063174. PMID 21448281.
- ^ a b Lai MC, Chang WC, Shieh SY, Tarn WY (November 2010). "DDX3 regulates cell growth through translational control of cyclin E1". Molecular and Cellular Biology. 30 (22): 5444–53. doi: 10.1128/MCB.00560-10. PMC 2976371. PMID 20837705.
- ^ a b c d e Phung B, Cieśla M, Sanna A, Guzzi N, Beneventi G, Cao Thi Ngoc P, et al. (June 2019). "The X-Linked DDX3X RNA Helicase Dictates Translation Reprogramming and Metastasis in Melanoma". Cell Reports. 27 (12): 3573–3586.e7. doi: 10.1016/j.celrep.2019.05.069. PMID 31216476.
- ^ Müller J, Krijgsman O, Tsoi J, Robert L, Hugo W, Song C, et al. (December 2014). "Low MITF/AXL ratio predicts early resistance to multiple targeted drugs in melanoma". Nature Communications. 5 (1): 5712. Bibcode: 2014NatCo...5.5712M. doi: 10.1038/ncomms6712. PMC 4428333. PMID 25502142.
- ^ Konieczkowski DJ, Johannessen CM, Abudayyeh O, Kim JW, Cooper ZA, Piris A, et al. (July 2014). "A melanoma cell state distinction influences sensitivity to MAPK pathway inhibitors". Cancer Discovery. 4 (7): 816–27. doi: 10.1158/2159-8290.CD-13-0424. PMC 4154497. PMID 24771846.
- ^ Robinson G, Parker M, Kranenburg TA, Lu C, Chen X, Ding L, et al. (August 2012). "Novel mutations target distinct subgroups of medulloblastoma". Nature. 488 (7409): 43–8. Bibcode: 2012Natur.488...43R. doi: 10.1038/nature11213. PMC 3412905. PMID 22722829.
- ^ Jones DT, Jäger N, Kool M, Zichner T, Hutter B, Sultan M, et al. (August 2012). "Dissecting the genomic complexity underlying medulloblastoma". Nature. 488 (7409): 100–5. Bibcode: 2012Natur.488..100J. doi: 10.1038/nature11284. PMC 3662966. PMID 22832583.
- ^ Pugh TJ, Weeraratne SD, Archer TC, Pomeranz Krummel DA, Auclair D, Bochicchio J, et al. (August 2012). "Medulloblastoma exome sequencing uncovers subtype-specific somatic mutations". Nature. 488 (7409): 106–10. Bibcode: 2012Natur.488..106P. doi: 10.1038/nature11329. PMC 3413789. PMID 22820256.
Further reading
- Li L, Li HS, Pauza CD, Bukrinsky M, Zhao RY (2006). "Roles of HIV-1 auxiliary proteins in viral pathogenesis and host-pathogen interactions". Cell Research. 15 (11–12): 923–34. doi: 10.1038/sj.cr.7290370. PMID 16354571.
- Owsianka AM, Patel AH (May 1999). "Hepatitis C virus core protein interacts with a human DEAD box protein DDX3". Virology. 257 (2): 330–40. doi: 10.1006/viro.1999.9659. PMID 10329544.
- Mamiya N, Worman HJ (May 1999). "Hepatitis C virus core protein binds to a DEAD box RNA helicase". The Journal of Biological Chemistry. 274 (22): 15751–6. doi: 10.1074/jbc.274.22.15751. PMID 10336476.
- Yagüe J, Alvarez I, Rognan D, Ramos M, Vázquez J, de Castro JA (June 2000). "An N-acetylated natural ligand of human histocompatibility leukocyte antigen (HLA)-B39. Classical major histocompatibility complex class I proteins bind peptides with a blocked NH(2) terminus in vivo". The Journal of Experimental Medicine. 191 (12): 2083–92. doi: 10.1084/jem.191.12.2083. PMC 2193201. PMID 10859333.
- Kim YS, Lee SG, Park SH, Song K (October 2001). "Gene structure of the human DDX3 and chromosome mapping of its related sequences". Molecules and Cells. 12 (2): 209–14. doi: 10.1016/S1016-8478(23)17085-3. PMID 11710523.
- Li J, Hawkins IC, Harvey CD, Jennings JL, Link AJ, Patton JG (November 2003). "Regulation of alternative splicing by SRrp86 and its interacting proteins". Molecular and Cellular Biology. 23 (21): 7437–47. doi: 10.1128/MCB.23.21.7437-7447.2003. PMC 207616. PMID 14559993.
- Shu H, Chen S, Bi Q, Mumby M, Brekken DL (March 2004). "Identification of phosphoproteins and their phosphorylation sites in the WEHI-231 B lymphoma cell line". Molecular & Cellular Proteomics. 3 (3): 279–86. doi: 10.1074/mcp.D300003-MCP200. PMID 14729942.
- Bouwmeester T, Bauch A, Ruffner H, Angrand PO, Bergamini G, Croughton K, et al. (February 2004). "A physical and functional map of the human TNF-alpha/NF-kappa B signal transduction pathway". Nature Cell Biology. 6 (2): 97–105. doi: 10.1038/ncb1086. PMID 14743216. S2CID 11683986.
- Yedavalli VS, Neuveut C, Chi YH, Kleiman L, Jeang KT (October 2004). "Requirement of DDX3 DEAD box RNA helicase for HIV-1 Rev-RRE export function". Cell. 119 (3): 381–92. doi: 10.1016/j.cell.2004.09.029. PMID 15507209.
- Dayton AI (October 2004). "Within you, without you: HIV-1 Rev and RNA export". Retrovirology. 1: 35. doi: 10.1186/1742-4690-1-35. PMC 526764. PMID 15516266.
- Krishnan V, Zeichner SL (December 2004). "Alterations in the expression of DEAD-box and other RNA binding proteins during HIV-1 replication". Retrovirology. 1: 42. doi: 10.1186/1742-4690-1-42. PMC 543576. PMID 15588285.
- Rush J, Moritz A, Lee KA, Guo A, Goss VL, Spek EJ, et al. (January 2005). "Immunoaffinity profiling of tyrosine phosphorylation in cancer cells". Nature Biotechnology. 23 (1): 94–101. doi: 10.1038/nbt1046. PMID 15592455. S2CID 7200157.
- Tao WA, Wollscheid B, O'Brien R, Eng JK, Li XJ, Bodenmiller B, et al. (August 2005). "Quantitative phosphoproteome analysis using a dendrimer conjugation chemistry and tandem mass spectrometry". Nature Methods. 2 (8): 591–8. doi: 10.1038/nmeth776. PMID 16094384. S2CID 20475874.
- Gevaert K, Staes A, Van Damme J, De Groot S, Hugelier K, Demol H, et al. (September 2005). "Global phosphoproteome analysis on human HepG2 hepatocytes using reversed-phase diagonal LC". Proteomics. 5 (14): 3589–99. doi: 10.1002/pmic.200401217. PMID 16097034. S2CID 895879.
- Chang PC, Chi CW, Chau GY, Li FY, Tsai YH, Wu JC, et al. (March 2006). "DDX3, a DEAD box RNA helicase, is deregulated in hepatitis virus-associated hepatocellular carcinoma and is involved in cell growth control". Oncogene. 25 (14): 1991–2003. doi: 10.1038/sj.onc.1209239. PMID 16301996.