| |||
| Names | |||
|---|---|---|---|
|
IUPAC name
Cyclopentadienide
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChemSpider | |||
PubChem
CID
|
|||
| |||
| |||
| Properties | |||
| [C 5H 5− or Cp− | |||
| Molar mass | 65.09 g/mol | ||
| Conjugate acid | Cyclopentadiene | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
In
chemistry, the cyclopentadienyl anion or cyclopentadienide is an
aromatic species with a formula of [C
5H
5−
and abbreviated as Cp−.
[1] It is formed by the deprotonation of
cyclopentadiene. The cyclopentadienyl anion is a
ligand which
binds to a
metal in
organometallic chemistry.
[2]
Resonance and aromaticity
The cyclopentadienyl anion is a planar, cyclic, regular- pentagonal ion; it has 6 π-electrons (4n + 2, where n = 1), which fulfills Hückel's rule of aromaticity. Each double bond and lone pair provides 2 π-electrons, which are delocalized into the ring. [3] The cyclopentadienyl anion is a conjugated system because there are alternating π and 𝜎 bonds. [4]
Cyclopentadiene has a pKa of about 16. It is acidic relative to many carbon acids. The enhanced acidity is attributed to stabilization of the conjugate base, cyclopentadienyl anion.
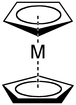
Ligand
Cyclopentadienyl anions form a variety of cyclopentadienyl complexes. [5]
See also
-
Cyclopentadienyl radical, [C
5H
5• -
Cyclopentadienyl cation, [C
5H
5+ -
Cyclooctatetraenide anion, [C
8H
82−
References
- ^ "Cyclopentadienide". PubChem Compound Database. National Center for Biotechnology Information. Retrieved 14 April 2016.
- ^ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, p. 66, ISBN 978-0-471-72091-1
- ^ Paul, Satadal; Goswami, Tamal; Misra, Anirban (2015-10-01). "Noncomparative scaling of aromaticity through electron itinerancy". AIP Advances. 5 (10): 107211. doi: 10.1063/1.4933191.
- ^ "Delocalised electrons- Definition and Examples of Delocalized electrons with FAQs". BYJUS. Retrieved 2023-04-01.
- ^ C. Elschenbroich (2006). Organometallics. VCH. ISBN 978-3-527-29390-2.


