| |
| Names | |
|---|---|
|
IUPAC name
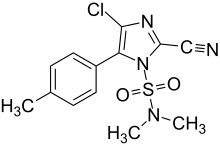
4-chloro-2-cyano-N,N-dimethyl-5-(4-methylphenyl)imidazole-1-sulfonamide
| |
| Other names
IKF 916
| |
| Identifiers | |
3D model (
JSmol)
|
|
| 9064229 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.111.751 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties [1] | |
| C13H13ClN4O2S | |
| Molar mass | 324.78 g·mol−1 |
| Density | 1.45 g/cm3 |
| Melting point | 152.7 °C |
| 0.107 mg/L (20 °C) | |
| log P | 3.2 |
| Hazards [2] | |
| GHS labelling: | |

| |
| Warning | |
| H410 | |
| P273, P391, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Cyazofamid is a fungicide that is highly-specific in controlling oomycete plant pathogens such as Phytophthora infestans, the organism which causes late blight in potato. Its mode of action is thought to involve binding to the Qi center of Coenzyme Q – cytochrome c reductase. [3] [4]
Cyazofamid is most often sold under the brand name Ranman. [5]
Synthesis
Processes to manufacture cyazofamid were disclosed in patents from Ishihara Sangyo Kaisha, Ltd. [6] An acetophenone derivative was first treated with aqueous glyoxal and hydroxylamine to form an oxime-substituted imidazole ring system. This intermediate was treated with thionyl chloride and disulfur dichloride to convert the oxime to a cyano group and chlorinate the imidazole in the position next to the phenyl ring. Finally, treatment with dimethylsulfamoyl chloride gave cyazofamid, with the desired regiochemistry. This placed the sulfamoyl group on the nitrogen adjacent to the phenyl ring rather than the chlorine atom. The structure of the fungicide has been confirmed by X-ray crystallography. [7]
References
- ^ Pesticide Properties Database. "Cyazofamid". University of Hertfordshire.
- ^ PubChem Database. "Cyazofamid".
- ^ Mitani, Shigeru; Araki, Satoshi; Takii, Yasuko; Ohshima, Takeshi; Matsuo, Norifusa; Miyoshi, Hideto (October 2001). "The Biochemical Mode of Action of the Novel Selective Fungicide Cyazofamid: Specific Inhibition of Mitochondrial Complex III in Phythium spinosum". Pesticide Biochemistry and Physiology. 71 (2): 107–115. doi: 10.1006/pest.2001.2569.
- ^ Mitani, Shigeru; Araki, Satoshi; Yamaguchi, Tomona; Takii, Yasuko; Ohshima, Takeshi; Matsuo, Norifusa (June 2001). "Antifungal Activity of the Novel Fungicide Cyazofamid against Phytophthora infestans and Other Plant Pathogenic Fungi in Vitro". Pesticide Biochemistry and Physiology. 70 (2): 92–99. doi: 10.1006/pest.2001.2541.
- ^ "Fungicides". ISKBC – ISK Biosciences CORPORATION. Retrieved 2021-05-25.
- ^ US patent 5869683, Jonishi H. et al., "Processes for producing 1-substituted-2-cyanoimidazole compounds", issued 1999-02-09, assigned to Ishihara Sangyo Kaisha Ltd.
- ^ Ning, Bin-Ke; Xu, Cheng; Li, Zong-Ying; Wang, Yue-Mei; Sun, Qiao-Nan (2013). "CCDC 958848: Experimental Crystal Structure Determination". Cambridge Structural Database. Cambridge Crystallographic Data Centre. doi: 10.5517/cc115rlk.
Further reading
- Ohshima, Takeshi; Komyoji, Terumasa; Mitani, Shigeru; Matsuo, Norifusa; Nakajima, Toshio (2004). "Development of a Novel Fungicide, Cyazofamid". Journal of Pesticide Science (in Japanese). 29 (2). Pesticide Science Society of Japan: 147–152. doi: 10.1584/jpestics.29.147. eISSN 1349-0923. ISSN 1348-589X. S2CID 84909424. AGRIS id JP2004007452. CiNii NAID 130004444938. ISSN-L 0385-1559.
- Anastassiadou, Maria; Arena, Maria; Auteri, Domenica; Brancato, Alba; Bura, Laszlo; Carrasco Cabrera, Luis; Chaideftou, Eugenia; Chiusolo, Arianna; Crivellente, Federica; De Lentdecker, Chloe; Egsmose, Mark; Fait, Gabriella; Greco, Luna; Ippolito, Alessio; Istace, Frederique; Jarrah, Samira; Kardassi, Dimitra; Leuschner, Renata; Lostia, Alfonso; Lythgo, Christopher; Magrans, Oriol; Mangas, Iris; Miron, Ileana; Molnar, Tunde; Padovani, Laura; Parra Morte, Juan Manuel; Pedersen, Ragnor; Reich, Hermine; Santos, Miguel; Sharp, Rachel; Sturma, Juergen; Szentes, Csaba; Terron, Andrea; Tiramani, Manuela; Vagenende, Benedicte; Villamar-Bouza, Laura (2020). "Updated peer review of the pesticide risk assessment of the active substance cyazofamid". EFSA Journal. 18 (9). European Food Safety Authority (Wiley): 1-25. doi: 10.2903/j.efsa.2020.6232. ISSN 1831-4732. PMC 7507320. PMID 32994826.
External links
- "Cyazofamid". PubChem. Retrieved 2021-05-17.
- "Pesticides - Fact Sheet for Cyazofamid - US EPA" (PDF). US EPA.
