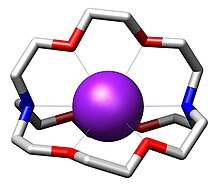
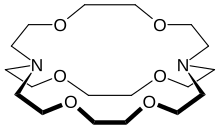
In chemistry, cryptands are a family of synthetic, bicyclic and polycyclic, multidentate ligands for a variety of cations. [2] The Nobel Prize for Chemistry in 1987 was given to Donald J. Cram, Jean-Marie Lehn, and Charles J. Pedersen for their efforts in discovering and determining uses of cryptands and crown ethers, thus launching the now flourishing field of supramolecular chemistry. [3] The term cryptand implies that this ligand binds substrates in a crypt, interring the guest as in a burial. These molecules are three-dimensional analogues of crown ethers but are more selective and strong as complexes for the guest ions. The resulting complexes are lipophilic. [4]
Structure
The most common and most important cryptand is N[CH2CH2OCH2CH2OCH2CH23N; the systematic IUPAC name for this compound is 1,10-diaza-4,7,13,16,21,24-hexaoxabicyclo[8.8.8]hexacosane. This compound is termed [2.2.2]cryptand, where the numbers indicate the number of ether oxygen atoms (and hence binding sites) in each of the three bridges between the amine nitrogen caps. Many cryptands are commercially available under the tradename Kryptofix. [5] All-amine cryptands exhibit particularly high affinity for alkali metal cations, which has allowed the isolation of salts of K−. [6]
Properties
Cation binding
The three-dimensional interior cavity of a cryptand provides
a binding site – or host – for "guest" ions. The complex between the
cationic guest and the cryptand is called a cryptate. Cryptands form complexes with many "hard cations" including NH+
4,
lanthanoids,
alkali metals, and
alkaline earth metals. In contrast to crown ethers, cryptands bind the guest ions using both
nitrogen and
oxygen donors. This three-dimensional encapsulation mode confers some size-selectivity, enabling discrimination among alkali metal cations (e.g. Na+ vs. K+). Some cryptands are luminescent.
[7]
Anion binding
Polyamine-based cryptands can be converted to polyammonium cages, which exhibit high affinities for anions. [8]
Laboratory uses
Cryptands enjoy some commercial applications (e.g. in homogenous-time-resolved-fluorescence, HTRF, technologies using Eu3+ as central ion). More importantly, they are reagents for the synthesis of inorganic and organometallic salts. Although more expensive and more difficult to prepare than crown ethers, cryptands bind alkali metals more strongly. [9] They are especially used to isolate salts of highly basic anions. [10] They convert solvated alkali metal cations into lipophilic cations, thereby conferring solubility in organic solvents to the resulting salts.
Referring to achievements that have been recognized in textbooks, cryptands enabled the synthesis of the
alkalides and
electrides. For example, addition of 2,2,2-cryptand to a solution of sodium in ammonia affords the salt [Na(2,2,2-crypt)]+e−, isolated a blue-black paramagnetic solid.
[11]
[12] Cryptands have also been used in the crystallization of
Zintl ions such as Sn4−
9.
[13]
Although rarely practical, cryptands can serve as phase transfer catalysts since their cationic complexes are lipophilic. [14]
See also
References
- ^ Alberto, R.; Ortner, K.; Wheatley, N.; Schibli, R.; Schubiger, A. P. (2001). "Synthesis and properties of boranocarbonate: a convenient in situ CO source for the aqueous preparation of [99mTc(OH2)3(CO)3+". J. Am. Chem. Soc. 121 (13): 3135–3136. doi: 10.1021/ja003932b. PMID 11457025.
- ^ Von Zelewsky, A. (1995). Stereochemistry of Coordination Compounds. Chichester: John Wiley. ISBN 0-471-95057-2.
- ^ Lehn, J. M. (1995). Supramolecular Chemistry: Concepts and Perspectives. Weinheim: VCH.
- ^ MacGillivray, Leonard R.; Atwood, Jerry L. (1999). "Structural Classification and General Principles for the Design of Spherical Molecular Hosts". Angewandte Chemie International Edition. 38 (8): 1018–1033. doi: 10.1002/(SICI)1521-3773(19990419)38:8<1018::AID-ANIE1018>3.0.CO;2-G. PMID 25138490.
- ^ 23978-09-8
- ^ Kim, J.; Ichimura, A. S.; Huang, R. H.; Redko, M.; Phillips, R. C.; Jackson, J. E.; Dye, J. L. (1999). "Crystalline Salts of Na− and K− (Alkalides) that Are Stable at Room Temperature". J. Am. Chem. Soc. 121 (45): 10666–10667. doi: 10.1021/ja992667v.
- ^ Valeur, B. (2000). "Design principles of fluorescent molecular sensors for cation recognition". Coordination Chemistry Reviews. 205: 3–40. doi: 10.1016/S0010-8545(00)00246-0.
- ^ Beer, Paul D.; Gale, Philip A. (2001). "Anion Recognition and Sensing: The State of the Art and Future Perspectives". Angewandte Chemie International Edition. 40 (3): 486–516. doi: 10.1002/1521-3773(20010202)40:3<486::AID-ANIE486>3.0.CO;2-P. PMID 11180358. S2CID 22334242.
- ^ Danil de Namor, Angela; Ghousseini, Lily; Lee, Walter (1985). "Stability constants and free energies of complexation of metal-ion cryptates in nitromethane. Derived parameters for the extraction of cations by cryptand 222 from water to pure nitromethane". Journal of the Chemical Society, Faraday Transactions 1: Physical Chemistry in Condensed Phases. 81 (10): 2495–2502. doi: 10.1039/F19858102495.
- ^ Dietrich, B. (1996). "Cryptands". In Gokel, G. W. (ed.). Comprehensive Supramolecular Chemistry. Vol. 1. Oxford: Elsevier. pp. 153–211. ISBN 0-08-040610-6.
- ^ Dye, J. L. (2003). "Electrons as Anions". Science. 301 (5633): 607–608. doi: 10.1126/science.1088103. PMID 12893933. S2CID 93768664.
- ^ Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. ISBN 0-12-352651-5
- ^ Edwards, Paul A.; Corbett, John D. (1977). "Stable Homopolyatomic Anions. Synthesis and Crystal Structures of Salts Containing the Pentaplumbide(2-) and Pentastannide(2-) Anions". Inorganic Chemistry. 16 (4): 903–907. doi: 10.1021/ic50170a036.
- ^ Landini, D.; Maia, A.; Montanari, F.; Tundo, P. (1979). "Lipophilic [2.2.2]cryptands as phase-transfer catalysts. Activation and Nucleophilicity of Anions in Aqueous-Organic Thirteen-Phase Systems and in Organic Solvents of Low Polarity". J. Am. Chem. Soc. 101 (10): 2526–2530. doi: 10.1021/ja00504a004.
General reading
- IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) " cryptand". doi: 10.1351/goldbook.C01426
- Lee, J.D. (1991). Concise Inorganic Chemistry (4th ed.). New York: Chapman & Hall. pp. 306–308 & 353. ISBN 0-412-40290-4.
- Krakowiak, K. E.; Bradshaw, J. S.; An, H.-Y.; Izatt, R. M. (1993). "Simple methods for the preparation of cryptands". Pure Appl. Chem. 65 (3): 511–514. doi: 10.1351/pac199365030511.