| |
| Names | |
|---|---|
|
IUPAC name
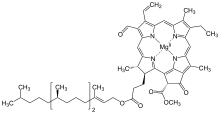
[methyl 14-ethyl-8-formyl-4,13,18-trimethyl-20-oxo-3-{3-oxo-3-[(3,7,11,15-tetramethylhexadec-2-en-1-yl)oxy]propyl}-9-vinylphorbine-21-carboxylatato(2−)-κ4N23,N24,N25,N26]magnesium
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
PubChem
CID
|
|
| |
| |
| Properties | |
| C55H70O6N4Mg [1] | |
| Molar mass | 907.4725 g/mol |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Chlorophyll f (Chl f) is a type form of chlorophyll that absorbs further in the red ( infrared light) than other chlorophylls. In 2010, it was reported by Min Chen to be present in stromatolites from Western Australia's Shark Bay. [2] [3]
The function of Chl f in photosynthetic reactions is uncertain and the ecological distribution of Chl f remains unknown. Chl f has been shown to support some of the roles in photosynthetic reactions, in both the energy transfer and in the charge separation processes. [4] [5] [6]
Chl f is produced from chlorophyllide f by chlorophyll synthase. Chlorophyllide f is made from chlorophyllide a by an enzyme known as PsbA4 or ChlF. [7]
References
- ^ Willows, Robert D.; Li, Yaqiong; Scheer, Hugo; Chen, Min (15 March 2013). "Structure of chlorophyll f". Organic Letters. 15 (7): 1588–1590. doi: 10.1021/ol400327j. PMID 23496297.
- ^ Chen, M.; Schliep, M.; Willows, R.D.; Cai, Z.-L.; Neilan, B.A.; Scheer, H. (2010). "A red-shifted chlorophyll". Science. 329 (5997): 1318–1319. Bibcode: 2010Sci...329.1318C. doi: 10.1126/science.1191127. PMID 20724585. S2CID 206527174.
- ^ Jabr, Ferris (August 19, 2010). "A new form of chlorophyll?". Scientific American. Retrieved 2010-09-07.
- ^ Nürnberg, Dennis J.; Morton, Jennifer; Santabarbara, Stefano; Telfer, Alison; Joliot, Pierre; Antonaru, Laura A.; et al. (2018). "Photochemistry beyond the red limit in chlorophyll f–containing photosystems". Science. 360 (6394): 1210–1213. Bibcode: 2018Sci...360.1210N. doi: 10.1126/science.aar8313. hdl: 10044/1/63104. ISSN 0036-8075. OCLC 7735829001. PMID 29903971.
- ^ Zamzam, Noura; Kaucikas, Marius; Nürnberg, Dennis J.; Rutherford, A. William; van Thor, Jasper J. (2019). "Femtosecond infrared spectroscopy of chlorophyll f-containing photosystem I". Physical Chemistry Chemical Physics. 21 (3): 1224–1234. Bibcode: 2019PCCP...21.1224Z. doi: 10.1039/C8CP05627G. hdl: 10044/1/66728. ISSN 1463-9076. OCLC 7943211172. PMID 30566126. S2CID 56477664.
- ^ Dunning, Hayley (June 14, 2018). "New type of photosynthesis discovered". Phys.org. Retrieved 2019-03-25.
- ^ Tsuzuki, Yuki; Tsukatani, Yusuke; Yamakawa, Hisanori; Itoh, Shigeru; Fujita, Yuichi; Yamamoto, Haruki (29 March 2022). "Effects of Light and Oxygen on Chlorophyll d Biosynthesis in a Marine Cyanobacterium Acaryochloris marina". Plants. 11 (7): 915. doi: 10.3390/plants11070915. PMC 9003380. PMID 35406896.