| Cell biology | |
|---|---|
| centrosome | |
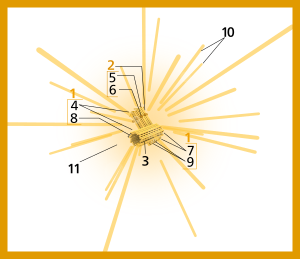 Components of a typical centrosome:
|

In cell biology a centriole is a cylindrical organelle composed mainly of a protein called tubulin. [1] Centrioles are found in most eukaryotic cells, but are not present in conifers ( Pinophyta), flowering plants ( angiosperms) and most fungi, and are only present in the male gametes of charophytes, bryophytes, seedless vascular plants, cycads, and Ginkgo. [2] [3] A bound pair of centrioles, surrounded by a highly ordered mass of dense material, called the pericentriolar material (PCM), [4] makes up a structure called a centrosome. [1]
Centrioles are typically made up of nine sets of short microtubule triplets, arranged in a cylinder. Deviations from this structure include crabs and Drosophila melanogaster embryos, with nine doublets, and Caenorhabditis elegans sperm cells and early embryos, with nine singlets. [5] [6] Additional proteins include centrin, cenexin and tektin. [7]
The main function of centrioles is to produce cilia during interphase and the aster and the spindle during cell division.
History
The centrosome was discovered jointly by Walther Flemming in 1875 [8] [9] and Edouard Van Beneden in 1876. [10] [9] Edouard Van Beneden made the first observation of centrosomes as composed of two orthogonal centrioles in 1883. [11] Theodor Boveri introduced the term "centrosome" in 1888 [12] [9] [13] [14] and the term "centriole" in 1895. [15] [9] The basal body was named by Theodor Wilhelm Engelmann in 1880. [16] [9] The pattern of centriole duplication was first worked out independently by Étienne de Harven and Joseph G. Gall c. 1950. [17] [18]
Role in cell division

Centrioles are involved in the organization of the mitotic spindle and in the completion of cytokinesis. [19] Centrioles were previously thought to be required for the formation of a mitotic spindle in animal cells. However, more recent experiments have demonstrated that cells whose centrioles have been removed via laser ablation can still progress through the G1 stage of interphase before centrioles can be synthesized later in a de novo fashion. [20] Additionally, mutant flies lacking centrioles develop normally, although the adult flies' cells lack flagella and cilia and as a result, they die shortly after birth. [21] The centrioles can self replicate during cell division.
Cellular organization
Centrioles are a very important part of centrosomes, which are involved in organizing microtubules in the cytoplasm. [22] [23] The position of the centriole determines the position of the nucleus and plays a crucial role in the spatial arrangement of the cell.

Fertility
Sperm centrioles are important for 2 functions: [24] (1) to form the sperm flagellum and sperm movement and (2) for the development of the embryo after fertilization. The sperm supplies the centriole that creates the centrosome and microtubule system of the zygote. [25]
Ciliogenesis
In flagellates and ciliates, the position of the flagellum or cilium is determined by the mother centriole, which becomes the basal body. An inability of cells to use centrioles to make functional flagella and cilia has been linked to a number of genetic and developmental diseases. In particular, the inability of centrioles to properly migrate prior to ciliary assembly has recently been linked to Meckel–Gruber syndrome. [26]
Animal development

Proper orientation of cilia via centriole positioning toward the posterior of embryonic node cells is critical for establishing left-right asymmetry, during mammalian development. [27]
Centriole duplication
Before DNA replication, cells contain two centrioles, an older mother centriole, and a younger daughter centriole. During cell division, a new centriole grows at the proximal end of both mother and daughter centrioles. After duplication, the two centriole pairs (the freshly assembled centriole is now a daughter centriole in each pair) will remain attached to each other orthogonally until mitosis. At that point the mother and daughter centrioles separate dependently on an enzyme called separase. [28]
The two centrioles in the centrosome are tied to one another. The mother centriole has radiating appendages at the distal end of its long axis and is attached to its daughter at the proximal end. Each daughter cell formed after cell division will inherit one of these pairs. Centrioles start duplicating when DNA replicates. [19]
Origin
The last common ancestor of all eukaryotes was a ciliated cell with centrioles.[ citation needed] Some lineages of eukaryotes, such as land plants, do not have centrioles except in their motile male gametes. Centrioles are completely absent from all cells of conifers and flowering plants, which do not have ciliate or flagellate gametes. [29] It is unclear if the last common ancestor had one [30] or two cilia. [31] Important genes such as centrins required for centriole growth, are only found in eukaryotes, and not in bacteria or archaea. [30]
Etymology and pronunciation
The word centriole ( /ˈsɛntrioʊl/) uses combining forms of centri- and -ole, yielding "little central part", which describes a centriole's typical location near the center of the cell.
Atypical centrioles
Typical centrioles are made of 9 triplets of microtubules organized with radial symmetry. [32] Centrioles can vary the number of microtubules and can be made of 9 doublets of microtubules (as in Drosophila melanogaster) or 9 singlets of microtubules as in C. elegans. Atypical centrioles are centrioles that do not have microtubules, such as the Proximal Centriole-Like found in D. melanogaster sperm, [33] or that have microtubules with no radial symmetry, such as in the distal centriole of human spermatozoon. [34] Atypical centrioles may have evolved at least eight times independently during vertebrate evolution and may evolve in the sperm after internal fertilization evolves. [35]
It wasn't clear why centriole become atypical until recently. The atypical distal centriole forms a dynamic basal complex (DBC) that, together with other structures in the sperm neck, facilitates a cascade of internal sliding, coupling tail beating with head kinking. The atypical distal centriole's properties suggest that it evolved into a transmission system that couples the sperm tail motors to the whole sperm, thereby enhancing sperm function. [36]
References
- ^ a b Eddé, B; Rossier, J; Le Caer, JP; Desbruyères, E; Gros, F; Denoulet, P (1990). "Posttranslational glutamylation of alpha-tubulin". Science. 247 (4938): 83–5. Bibcode: 1990Sci...247...83E. doi: 10.1126/science.1967194. PMID 1967194.
- ^ Quarmby, LM; Parker, JD (2005). "Cilia and the cell cycle?". The Journal of Cell Biology. 169 (5): 707–10. doi: 10.1083/jcb.200503053. PMC 2171619. PMID 15928206.
- ^ Silflow, CD; Lefebvre, PA (2001). "Assembly and motility of eukaryotic cilia and flagella. Lessons from Chlamydomonas reinhardtii". Plant Physiology. 127 (4): 1500–1507. doi: 10.1104/pp.010807. PMC 1540183. PMID 11743094.
- ^ Lawo, Steffen; Hasegan, Monica; Gupta, Gagan D.; Pelletier, Laurence (November 2012). "Subdiffraction imaging of centrosomes reveals higher-order organizational features of pericentriolar material". Nature Cell Biology. 14 (11): 1148–1158. doi: 10.1038/ncb2591. ISSN 1476-4679. PMID 23086237. S2CID 11286303.
- ^ Delattre, M; Gönczy, P (2004). "The arithmetic of centrosome biogenesis" (PDF). Journal of Cell Science. 117 (Pt 9): 1619–30. doi: 10.1242/jcs.01128. PMID 15075224. S2CID 7046196. Archived (PDF) from the original on 18 August 2017.
- ^ Leidel, S.; Delattre, M.; Cerutti, L.; Baumer, K.; Gönczy, P (2005). "SAS-6 defines a protein family required for centrosome duplication in C. elegans and in human cells". Nature Cell Biology. 7 (2): 115–25. doi: 10.1038/ncb1220. PMID 15665853. S2CID 4634352.
- ^ Rieder, C. L.; Faruki, S.; Khodjakov, A. (October 2001). "The centrosome in vertebrates: more than a microtubule-organizing center". Trends in Cell Biology. 11 (10): 413–419. doi: 10.1016/S0962-8924(01)02085-2. ISSN 0962-8924. PMID 11567874.
- ^ Flemming, W. (1875). Studien uber die Entwicklungsgeschichte der Najaden. Sitzungsgeber. Akad. Wiss. Wien 71, 81–147
- ^ a b c d e Bloodgood RA. From central to rudimentary to primary: the history of an underappreciated organelle whose time has come. The primary cilium. Methods Cell Biol. 2009;94:3-52. doi: 10.1016/S0091-679X(08)94001-2. Epub 2009 Dec 23. PMID 20362083.
- ^ Van Beneden, E. (1876). Contribution a l’histoire de la vesiculaire germinative et du premier noyau embryonnaire. Bull. Acad. R. Belg (2me series) 42, 35–97.
- ^ Wunderlich, V. (2002). "JMM - Past and Present". Journal of Molecular Medicine. 80 (9): 545–548. doi: 10.1007/s00109-002-0374-y. PMID 12226736.
- ^ Boveri, T. (1888). Zellen-Studien II. Die Befruchtung und Teilung des Eies von Ascaris megalocephala. Jena. Z. Naturwiss. 22, 685–882.
- ^ Boveri, T. Ueber das Verhalten der Centrosomen bei der Befruchtung des Seeigel-Eies nebst allgemeinen Bemerkungen über Centrosomen und Verwandtes. Verh. d. Phys.-Med. Ges. zu Würzburg, N. F., Bd. XXIX, 1895. link.
- ^ Boveri, T. (1901). Zellen-Studien: Uber die Natur der Centrosomen. IV. Fischer, Jena. link.
- ^ Boveri, T. (1895). Ueber die Befruchtungs und Entwickelungsfahigkeit kernloser Seeigeleier und uber die Moglichkeit ihrer Bastardierung. Arch. Entwicklungsmech. Org. (Wilhelm Roux) 2, 394–443.
- ^ Engelmann, T. W. (1880). Zur Anatomie und Physiologie der Flimmerzellen. Pflugers Arch. 23, 505–535.
- ^ Wolfe, Stephen L. (1977). Biology: the foundations (First ed.). Wadsworth. ISBN 9780534004903.
- ^ Vorobjev, I. A.; Nadezhdina, E. S. (1987). The Centrosome and Its Role in the Organization of Microtubules. International Review of Cytology. Vol. 106. pp. 227–293. doi: 10.1016/S0074-7696(08)61714-3. ISBN 978-0-12-364506-7. PMID 3294718.. See also de Harven's own recollections of this work: de Harven, Etienne (1994). "Early observations of centrioles and mitotic spindle fibers by transmission electron microscopy". Biology of the Cell. 80 (2–3): 107–109. doi: 10.1111/j.1768-322X.1994.tb00916.x. PMID 8087058. S2CID 84594630.
- ^ a b Salisbury, JL; Suino, KM; Busby, R; Springett, M (2002). "Centrin-2 is required for centriole duplication in mammalian cells". Current Biology. 12 (15): 1287–92. doi: 10.1016/S0960-9822(02)01019-9. PMID 12176356. S2CID 1415623.
- ^ La Terra, S; English, CN; Hergert, P; McEwen, BF; Sluder, G; Khodjakov, A (2005). "The de novo centriole assembly pathway in HeLa cells: cell cycle progression and centriole assembly/maturation". The Journal of Cell Biology. 168 (5): 713–22. doi: 10.1083/jcb.200411126. PMC 2171814. PMID 15738265.
- ^ Basto, R; Lau, J; Vinogradova, T; Gardiol, A; Woods, CG; Khodjakov, A; Raff, JW (2006). "Flies without centrioles". Cell. 125 (7): 1375–86. doi: 10.1016/j.cell.2006.05.025. PMID 16814722. S2CID 2080684.
- ^ Feldman, JL; Geimer, S; Marshall, WF (2007). "The mother centriole plays an instructive role in defining cell geometry". PLOS Biology. 5 (6): e149. doi: 10.1371/journal.pbio.0050149. PMC 1872036. PMID 17518519.
- ^ Beisson, J; Wright, M (2003). "Basal body/centriole assembly and continuity". Current Opinion in Cell Biology. 15 (1): 96–104. doi: 10.1016/S0955-0674(02)00017-0. PMID 12517710.
- ^ Avidor-Reiss, T., Khire, A., Fishman, E. L., & Jo, K. H. (2015). Atypical centrioles during sexual reproduction. Frontiers in cell and developmental biology, 3, 21. Chicago
- ^ Hewitson, Laura & Schatten, Gerald P. (2003). "The biology of fertilization in humans". In Patrizio, Pasquale; et al. (eds.). A color atlas for human assisted reproduction: laboratory and clinical insights. Lippincott Williams & Wilkins. p. 3. ISBN 978-0-7817-3769-2. Retrieved 9 November 2013.
- ^ Cui, Cheng; Chatterjee, Bishwanath; Francis, Deanne; Yu, Qing; SanAgustin, Jovenal T.; Francis, Richard; Tansey, Terry; Henry, Charisse; Wang, Baolin; Lemley, Bethan; Pazour, Gregory J.; Lo, Cecilia W. (2011). "Disruption of Mks1 localization to the mother centriole causes cilia defects and developmental malformations in Meckel-Gruber syndrome". Dis. Models Mech. 4 (1): 43–56. doi: 10.1242/dmm.006262. PMC 3008963. PMID 21045211.
- ^ Babu, Deepak; Roy, Sudipto (1 May 2013). "Left–right asymmetry: cilia stir up new surprises in the node". Open Biology. 3 (5): 130052. doi: 10.1098/rsob.130052. ISSN 2046-2441. PMC 3866868. PMID 23720541.
- ^ Tsou, MF; Stearns, T (2006). "Mechanism limiting centrosome duplication to once per cell cycle". Nature. 442 (7105): 947–51. Bibcode: 2006Natur.442..947T. doi: 10.1038/nature04985. PMID 16862117. S2CID 4413248.
- ^ Marshall, W.F. (2009). "Centriole Evolution". Current Opinion in Cell Biology. 21 (1): 14–19. doi: 10.1016/j.ceb.2009.01.008. PMC 2835302. PMID 19196504.
- ^ a b Bornens, M.; Azimzadeh, J. (2007). "Origin and Evolution of the Centrosome". Eukaryotic Membranes and Cytoskeleton. Advances in Experimental Medicine and Biology. Vol. 607. pp. 119–129. doi: 10.1007/978-0-387-74021-8_10. ISBN 978-0-387-74020-1. PMID 17977464.
- ^ Rogozin, I. B.; Basu, M. K.; Csürös, M.; Koonin, E. V. (2009). "Analysis of Rare Genomic Changes Does Not Support the Unikont-Bikont Phylogeny and Suggests Cyanobacterial Symbiosis as the Point of Primary Radiation of Eukaryotes". Genome Biology and Evolution. 1: 99–113. doi: 10.1093/gbe/evp011. PMC 2817406. PMID 20333181.
- ^ Avidor-Reiss, Tomer; Gopalakrishnan, Jayachandran (2013). "Building a centriole". Current Opinion in Cell Biology. 25 (1): 72–7. doi: 10.1016/j.ceb.2012.10.016. PMC 3578074. PMID 23199753.
- ^ Blachon, S; Cai, X; Roberts, K. A; Yang, K; Polyanovsky, A; Church, A; Avidor-Reiss, T (2009). "A Proximal Centriole-Like Structure is Present in Drosophila Spermatids and Can Serve as a Model to Study Centriole Duplication". Genetics. 182 (1): 133–44. doi: 10.1534/genetics.109.101709. PMC 2674812. PMID 19293139.
- ^ Fishman, Emily L; Jo, Kyoung; Nguyen, Quynh P. H; Kong, Dong; Royfman, Rachel; Cekic, Anthony R; Khanal, Sushil; Miller, Ann L; Simerly, Calvin; Schatten, Gerald; Loncarek, Jadranka; Mennella, Vito; Avidor-Reiss, Tomer (2018). "A novel atypical sperm centriole is functional during human fertilization". Nature Communications. 9 (1): 2210. Bibcode: 2018NatCo...9.2210F. doi: 10.1038/s41467-018-04678-8. PMC 5992222. PMID 29880810.
- ^ Turner, K., N. Solanki, H.O. Salouha, and T. Avidor-Reiss. 2022. Atypical Centriolar Composition Correlates with Internal Fertilization in Fish. Cells. 11:758, https://www.mdpi.com/2073-4409/11/5/758
- ^ Khanal, S., M.R. Leung, A. Royfman, E.L. Fishman, B. Saltzman, H. Bloomfield-Gadelha, T. Zeev-Ben-Mordehai, and T. Avidor-Reiss. 2021. A dynamic basal complex modulates mammalian sperm movement. Nat Commun. 12:3808.. https://doi.org/10.1038/s41467-021-24011-0