In organic chemistry and organometallic chemistry, carbon–hydrogen bond activation (C−H activation) is a type of organic reaction in which a carbon–hydrogen bond is cleaved and replaced with a C−X bond (X ≠ H is typically a main group element, like carbon, oxygen, or nitrogen). Some authors further restrict the term C–H activation to reactions in which a C–H bond, one that is typically considered to be "unreactive", interacts with a transition metal center M, resulting in its cleavage and the generation of an organometallic species with an M–C bond. The intermediate of this step (sometimes known as the C−H activation step) could then undergo subsequent reactions with other reagents, either in situ or in a separate step, to produce the functionalized product. [1]
The alternative term C−H functionalization is used to describe any reaction that converts a relatively inert C−H bond into a C−X bond, irrespective of the reaction mechanism (or with an agnostic attitude towards it). In particular, this definition does not require the cleaved C–H bond to initially interact with the transition metal in the reaction mechanism. This broader definition encompasses all reactions that would fall under the restricted definition of C–H activation given above. However, it also includes iron-catalyzed alkane C–H hydroxylation reactions that proceed through the oxygen rebound mechanism (e.g. cytochrome P450 enzymes and their synthetic analogues), in which a metal–carbon bond is not believed to be involved. Likewise, the ligand-based reactivity of many metal carbene species with hydrocarbons in which the carbene carbon inserts into a C–H bond, again without interaction of the hydrocarbon C–H bond with the metal, also falls under this category. Often, when authors make the distinction between C–H functionalization and C−H activation, they will restrict the latter to the narrow sense.
Classification
Mechanisms for C-H activations by metal centers can be classified into three general categories:
- (i) Oxidative addition, in which a low-valent metal center inserts into a carbon-hydrogen bond, which cleaves the bond and oxidizes the metal:
- LnM + RH → LnMR(H)
- (ii) Electrophilic activation in which an electrophilic metal attacks the hydrocarbon, displacing a proton:
- LnM+ + RH → LnMR + H+
- (iii) Sigma-bond metathesis, which proceeds through a "four-centered" transition state in which bonds break and form in a single step:
- LnMX + RH → LnMR + XH
Historic overview
The first C–H activation reaction is often attributed to Otto Dimroth, who in 1902, reported that benzene reacted with mercury(II) acetate (See: organomercury). Many electrophilic metal centers undergo this Friedel-Crafts-like reaction. Joseph Chatt observed the addition of C-H bonds of naphthalene by Ru(0) complexes. [2]
Chelation-assisted C-H activations are prevalent. Shunsuke Murahashi reported a cobalt-catalyzed chelation-assisted C-H functionalization of 2-phenylisoindolin-1-one from (E)-N,1-diphenylmethanimine. [3]

In 1969, A.E. Shilov reported that potassium tetrachloroplatinate induced isotope scrambling between methane and heavy water. The pathway was proposed to involve binding of methane to Pt(II). In 1972, the Shilov group was able to produce methanol and methyl chloride in a similar reaction involving a stoichiometric amount of potassium tetrachloroplatinate, catalytic potassium hexachloroplatinate, methane and water. Due to the fact that Shilov worked and published in the Soviet Union during the Cold War era, his work was largely ignored by Western scientists. This so-called Shilov system is today one of the few true catalytic systems for alkane functionalizations. [1] [4]
In some cases, discoveries in C-H activation were being made in conjunction with those of cross coupling. In 1969, [5] Yuzo Fujiwara reported the synthesis of (E)-1,2-diphenylethene from benzene and styrene with Pd(OAc)2 and Cu(OAc)2, a procedure very similar to that of cross coupling. On the category of oxidative addition, M. L. H. Green in 1970 reported on the photochemical insertion of tungsten (as a Cp2WH2 complex) in a benzene C–H bond [6] and George M. Whitesides in 1979 was the first to carry out an intramolecular aliphatic C–H activation [7]

The next breakthrough was reported independently by two research groups in 1982. R. G. Bergman reported the first transition metal-mediated intermolecular C–H activation of unactivated and completely saturated hydrocarbons by oxidative addition. Using a photochemical approach, photolysis of Cp*Ir(PMe3)H2, where Cp* is a pentamethylcyclopentadienyl ligand, led to the coordinatively unsaturated species Cp*Ir(PMe3) which reacted via oxidative addition with cyclohexane and neopentane to form the corresponding hydridoalkyl complexes, Cp*Ir(PMe3)HR, where R = cyclohexyl and neopentyl, respectively. [8] W.A.G. Graham found that the same hydrocarbons react with Cp*Ir(CO)2 upon irradiation to afford the related alkylhydrido complexes Cp*Ir(CO)HR, where R = cyclohexyl and neopentyl, respectively. [9] In the latter example, the reaction is presumed to proceed via the oxidative addition of alkane to a 16-electron iridium(I) intermediate, Cp*Ir(CO), formed by irradiation of Cp*Ir(CO)2.
The selective activation and functionalization of alkane C–H bonds was reported using a tungsten complex outfitted with pentamethylcyclopentadienyl, nitrosyl, allyl and neopentyl ligands, Cp*W(NO)(η3-allyl)(CH2CMe3). [10]
In one example involving this system, the alkane pentane is selectively converted to the halocarbon 1-iodopentane. This transformation was achieved via the thermolysis of Cp*W(NO)(η3-allyl)(CH2CMe3) in pentane at room temperature, resulting in elimination of neopentane by a pseudo-first-order process, generating an undetectable electronically and sterically unsaturated 16-electron intermediate that is coordinated by an η2- butadiene ligand. Subsequent intermolecular activation of a pentane solvent molecule then yields an 18-electron complex possessing an n-pentyl ligand. In a separate step, reaction with iodine at −60 °C liberates 1-iodopentane from the complex.
Mechanistic understanding
An important aspect of improving chemical reactions is the understanding of the underlying reaction mechanism. To answer this question for C-H activation, time-resolved spectroscopic techniques can be used to follow the dynamics of the chemical reaction. This technique requires a trigger for initiating the process, which is in most cases illumination of the compound. Photoinitiated reactions of transition metal complexes with alkanes serve as a powerful model systems for understanding the cleavage of the strong C-H bond. [8] [9]

In such systems, the sample is illuminated with UV-light which excites an electron from the metal center to an unoccupied, antibonding ligand orbitals ( MLCT), leading to ligand dissociation. This creates a highly reactive, electron deficient 16-electron intermediate, with a vacant coordination site. This species then binds to an alkane molecule, forming a σ-complex coordinating to a C-H bond. In a third step, the metal atom inserts into the C-H bond, cleaving it and yielding the C-H bond activated product.
The intermediates and their kinetics can be observed using different time-resolved spectroscopic techniques (e.g. TR- IR, TR- XAS, TR- RIXS). Time-resolved infrared spectroscopy (TR-IR) is a rather convenient method to observe these intermediates. However, it is only limited to complexes which have IR-active ligands and is prone to correct assignments on the femtosecond timescale due to underlying vibrational cooling. To answer the question of difference in reactivity for distinct complexes, the electronic structure of those needs to be investigated. This can be achieved by X-ray absorption spectroscopy (XAS) or resonant inelastic X-ray scattering (RIXS). These methods have been successfully used to follow the steps of C-H activation with orbital resolution and provide detailed insights into the responsible interactions for the C-H bond breaking. [11] [12]
Full characterization of the structure of methane bound to a metal center was reported by Girolami in 2023: isotopic perturbation of equilibrium (IPE) studies involving deuterated isotopologs showed that methane binds to the metal center through a single M···H-C bridge; changes in the 1JCH coupling constants indicate clearly that the structure of the methane ligand is significantly perturbed relative to the free molecule. [13]
Directed C-H activation
Directed-, chelation-assisted-, or "guided" C-H activation involves directing groups that influence regio- and stereochemistry. [14] This is the most useful style of C-H activation in organic synthesis. N,N-dimethylbenzylamine undergoes cyclometalation readily by many transition metals. [15] A semi-practical implementations involve weakly coordinating directing groups, as illustrated by the Murai reaction. [16]

The mechanism for the Pd-catalyzed C-H activation reactions of 2-phenylpyridine involves a metallacycle intermediate. The intermediate is oxidized to form a PdIV species, followed by reductive elimination to form the C-O bond and release the product. [17]

Borylation
Transforming C-H bonds into C-B bonds through borylation has been thoroughly investigated due to their utility in synthesis (i.e. for cross-coupling reactions). John F. Hartwig reported a highly regioselective arene and alkane borylation catalyzed by a rhodium complex. In the case of alkanes, exclusive terminal functionalization was observed. [18]

Later, ruthenium catalysts were discovered to have higher activity and functional group compatibility. [19]
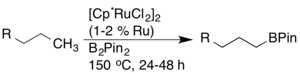
Other borylation catalysts have also been developed, including iridium-based catalysts, which successfully activate C-H bonds with high compatibility. [20] [21] [22]
For more information, consult borylation.
Natural gas
Naturally occurring methane is not utilized as a chemical feedstock, despite its abundance and low cost. Current technology makes prodigious use of methane by steam reforming to produce syngas, a mixture of carbon monoxide and hydrogen. This syngas is then used in Fischer-Tropsch reactions to make longer carbon chain products or methanol, one of the most important industrial chemical feedstocks. [23] [24] An intriguing method to convert these hydrocarbons involves C-H activation. Roy A. Periana, for example, reported that complexes containing late transition metals, such as Pt, Pd, Au, and Hg, react with methane (CH4) in H2SO4 to yield methyl bisulfate. [25] [26] The process has not however been implemented commercially.
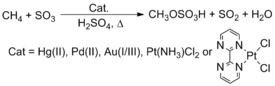
Asymmetric C-H activations

The total synthesis of lithospermic acid employs guided C-H functionalization late stage to a highly functionalized system. The directing group, a chiral nonracemic imine, is capable of performing an intramolecular alkylation, which allows for the rhodium-catalyzed conversion of imine to the dihydrobenzofuran. [28]

The total synthesis of calothrixin A and B features an intramolecular Pd-catalyzed cross coupling reaction via C-H activation, an example of a guided C-H activation. Cross coupling occurs between aryl C-I and C-H bonds to form a C-C bond. [29] The synthesis of a mescaline analogue employs the rhodium-catalyzed enantioselective annulation of an aryl imine via a C-H activation. [30]

See also
- Carbon-carbon bond activation
- Oxidative coupling of methane
- Cross dehydrogenative coupling [CDC reaction]
- Shilov system
- Meta-selective C-H functionalization
Older reviews
- Pre-2004
- Arndtsen, B. A.; Bergman, R. G.; Mobley, T. A.; Peterson, T. H. (1995). "Selective Intermolecular Carbon–Hydrogen Bond Activation by Synthetic Metal Complexes in Homogeneous Solution". Accounts of Chemical Research. 28 (3): 154–162. doi: 10.1021/ar00051a009.
- Crabtree, R. H. (2001). "Alkane C–H activation and functionalization with homogeneous transition metal catalysts: a century of progress – a new millennium in prospect". J. Chem. Soc., Dalton Trans. 17 (17): 2437–2450. doi: 10.1039/B103147N.
- 2004-7
- Crabtree, R. H. (2004). "Organometallic alkane CH activation". J. Organomet. Chem. 689 (24): 4083–4091. doi: 10.1016/j.jorganchem.2004.07.034. S2CID 95482372.
- Organometallic C–H Bond Activation: An Introduction Alan S. Goldman and Karen I. Goldberg ACS Symposium Series 885, Activation and Functionalization of C–H Bonds, 2004, 1–43
- Periana, R. A.; Bhalla, G.; Tenn, W. J.; III; Young, K. J. H.; Liu, X. Y.; Mironov, O.; Jones, C.; Ziatdinov, V. R. (2004). "Perspectives on some challenges and approaches for developing the next generation of selective, low temperature, oxidation catalysts for alkane hydroxylation based on the C–H activation reaction". Journal of Molecular Catalysis A: Chemical. 220 (1): 7–25. doi: 10.1016/j.molcata.2004.05.036.
- Lersch, M.Tilset (2005). "Mechanistic Aspects of C−H Activation by Pt Complexes". Chem. Rev. 105 (6): 2471–2526. doi: 10.1021/cr030710y. PMID 15941220., Vedernikov, A. N. (2007). "Recent Advances in the Platinum-mediated CH Bond Functionalization". Curr. Org. Chem. 11 (16): 1401–1416. doi: 10.2174/138527207782418708.
- 2008-2011
- Davies, H. M. L.; Manning, J. R. (2008). "Catalytic C–H functionalization by metalcarbenoid and nitrenoid insertion". Nature. 451 (7177): 417–424. Bibcode: 2008Natur.451..417D. doi: 10.1038/nature06485. PMC 3033428. PMID 18216847.
- Boutadla, Y.; Davies, D. L.; Macgregor, S. A.; Poblador-Bahamonde, A. I. (2009). "Mechanisms of C–H bond activation: rich synergy between computation and experiment". Dalton Trans. 2009 (30): 5820–5831. doi: 10.1039/B904967C. PMID 19623381.
- Wencel-Delord, J.; Dröge, T.; Liu, F.; Glorius, F. (2011). "Towards Mild Metal-Catalyzed C–H Bond Activation". Chem. Soc. Rev. 40 (9): 4740–4761. doi: 10.1039/C1CS15083A. PMID 21666903.
- Shulpin, G. B. (2010). "Selectivity enhancement in functionalization of C–H bonds: A review". Org. Biomol. Chem. 8 (19): 4217–4228. doi: 10.1039/c004223d. PMID 20593075.
- Lyons, T. W.; Sanford, M. S. (2010). "Palladium-Catalyzed Ligand-Directed C–H Functionalization Reactions". Chem. Rev. 110 (2): 1147–1169. doi: 10.1021/cr900184e. PMC 2836499. PMID 20078038.*Balcells, D.; Clot, E.; Eisenstein, O. (2010). "C–H Bond Activation in Transition Metal Species from a Computational Perspective". Chem. Rev. 110 (2): 749–823. doi: 10.1021/cr900315k. PMID 20067255.
- 2012-2015
- Hashiguchi, B. G.; Bischof, S. M.; Konnick, M. M.; Periana, R. A. (2012). "Designing Catalysts for Functionalization of Unactivated C–H Bonds Based on the CH Activation Reaction". Acc. Chem. Res. 45 (6): 885–898. doi: 10.1021/ar200250r. PMID 22482496.
- Kuhl, N.; Hopkinson, M. N.; Wencel-Delord, J.; Glorius, F. (2012). "Beyond Directing Groups: Transition Metal-Catalyzed C H Activation of Simple Arenes". Angew. Chem. Int. Ed. 51 (41): 10236–10254. doi: 10.1002/anie.201203269. PMID 22996679.
- Wencel-Delord, J.; Glorius, F. (2013). "C–H bond activation enables the rapid construction and late-stage diversification of functional molecules". Nature Chemistry. 5 (5): 369–375. Bibcode: 2013NatCh...5..369W. doi: 10.1038/nchem.1607. PMID 23609086.
Additional sources
- Bergman FAQ in Nature on C-H activation (2007)
- Literature Presentation by Ramtohul in Stoltz group on applications of C-H activation
- Powerpoint on John Bercaw's work
- Center for Selective C-H Functionalization
References
- ^ a b Gandeepan, Parthasarathy; Müller, Thomas; Zell, Daniel; Cera, Gianpiero; Warratz, Svenja; Ackermann, Lutz (2019). "3d Transition Metals for C–H Activation". Chemical Reviews. 119 (4): 2192–2452. doi: 10.1021/acs.chemrev.8b00507. PMID 30480438. S2CID 53726772.
- ^ Chatt, J.; Davidson, J. M. (1965). "The tautomerism of arene and ditertiary phosphine complexes of ruthenium(0), and the preparation of new types of hydrido-complexes of ruthenium(II)". J. Chem. Soc. 1965: 843. doi: 10.1039/JR9650000843.
- ^ Murahashi, Shunsuke (1955-12-01). "Synthesis of Phthalimidines from Schiff Bases and Carbon Monoxide". Journal of the American Chemical Society. 77 (23): 6403–6404. doi: 10.1021/ja01628a120. ISSN 0002-7863.
- ^ Fekl, U.; Goldberg, K. I. (2003). Homogeneous Hydrocarbon C-H Bond Activation and Functionalization with Platinum. Advances in Inorganic Chemistry. Vol. 54. pp. 259–320. doi: 10.1016/S0898-8838(03)54005-3. ISBN 9780120236541.
- ^ Fujiwara, Yuzo; Noritani, Ichiro; Danno, Sadao; Asano, Ryuzo; Teranishi, Shiichiro (1969-12-01). "Aromatic substitution of olefins. VI. Arylation of olefins with palladium(II) acetate". Journal of the American Chemical Society. 91 (25): 7166–7169. doi: 10.1021/ja01053a047. ISSN 0002-7863. PMID 27462934.
- ^ Green, M. L.; Knowles, P. J. (1970). "Formation of a tungsten phenyl hydride derivatives from benzene". J. Chem. Soc. D. 24 (24): 1677. doi: 10.1039/C29700001677.
- ^ Foley, Paul; Whitesides, George M. (1979). "Thermal generation of bis(triethylphosphine)-3,3-dimethylplatinacyclobutane from dineopentylbis(triethylphosphine)platinum(II)". J. Am. Chem. Soc. 101 (10): 2732–2733. doi: 10.1021/ja00504a041.
- ^ a b Janowicz, Andrew H.; Bergman, Robert G. (1982). "Carbon–hydrogen activation in saturated hydrocarbons: direct observation of M + R−H → M(R)(H)". J. Am. Chem. Soc. 104 (1): 352–354. doi: 10.1021/ja00365a091.
- ^ a b Hoyano, James K.; Graham, William A. G. (1982). "Oxidative addition of the carbon–hydrogen bonds of neopentane and cyclohexane to a photochemically generated iridium(I) complex". J. Am. Chem. Soc. 104 (13): 3723–3725. doi: 10.1021/ja00377a032.
- ^ Baillie, Rhett A.; Legzdins, Peter (2013). "Distinctive Activation and Functionalization of Hydrocarbon C–H Bonds Initiated by Cp*W(NO)(η3-allyl)(CH2CMe3) Complexes". Acc. Chem. Res. 47 (2): 330–340. doi: 10.1021/ar400108p. PMID 24047442.
- ^ Jay, Raphael M.; Banerjee, Ambar; Leitner, Torsten; Wang, Ru-Pan; Harich, Jessica; Stefanuik, Robert; Wikmark, Hampus; Coates, Michael R.; Beale, Emma V.; Kabanova, Victoria; Kahraman, Abdullah; Wach, Anna; Ozerov, Dmitry; Arrell, Christopher; Johnson, Philip J. M. (2023-06-02). "Tracking C–H activation with orbital resolution". Science. 380 (6648): 955–960. doi: 10.1126/science.adf8042. ISSN 0036-8075. PMID 37262165. S2CID 259002884.
- ^ Banerjee, Ambar; Jay, Raphael M.; Leitner, Torsten; Wang, Ru-Pan; Harich, Jessica; Stefanuik, Robert; Coates, Michael R.; Beale, Emma V.; Kabanova, Victoria (2023-07-26). Accessing Metal-Specific Orbital Interactions in C-H Activation using Resonant Inelastic X-ray Scattering (Report). Chemistry. doi: 10.26434/chemrxiv-2023-fm49h.
- ^ Sempsrott, Peter J.; Trinh, Brian B.; Flener Lovitt, Charity; Capra, Nicolas E.; Girolami, Gregory S. (2023-06-09). "An Osmium(II) Methane Complex: Elucidation of the Methane Coordination Mode". Science Advances. 9 (23). doi: 10.1126/sciadv.adg8130. PMC 10256148.
- ^ Brückl, T.; Baxter, R. D.; Ishihara, Y.; Baran, P. S. (2012). "Innate and Guided C-H Functionalization Logic". Accounts of Chemical Research. 45 (6): 826–839. doi: 10.1021/ar200194b. PMC 3288638. PMID 22017496.
- ^ Chetcuti, Michael J.; Ritleng, Vincent (2007). "Formation of a Ruthenium–Arene Complex, Cyclometallation with a Substituted Benzylamine, and Insertion of an Alkyne". J. Chem. Educ. 84 (6): 1014. Bibcode: 2007JChEd..84.1014C. doi: 10.1021/ed084p1014.
- ^ Murai, Shinji; Kakiuchi, Fumitoshi; Sekine, Shinya; Tanaka, Yasuo; Kamatani, Asayuki; Sonoda, Motohiro; Chatani, Naoto (1993). "Efficient catalytic addition of aromatic carbon–hydrogen bonds to olefins". Nature. 366 (6455): 529–531. Bibcode: 1993Natur.366..529M. doi: 10.1038/366529a0. S2CID 5627826.
- ^ Lyons, T. W.; Sanford, M. S. (2010). "Palladium-Catalyzed Ligand-Directed C–H Functionalization Reactions". Chem. Rev. 110 (2): 1147–1169. doi: 10.1021/cr900184e. PMC 2836499. PMID 20078038.
- ^ Chen, Huiyuan; Schlecht, Sabine; Semple, Thomas C.; Hartwig, John F. (2000). "Thermal, Catalytic, Regiospecific Functionalization of Alkanes". Science. 287 (5460): 1995–1997. Bibcode: 2000Sci...287.1995C. doi: 10.1126/science.287.5460.1995. PMID 10720320.
- ^ Murphy, J. M.; Lawrence, J. D.; Kawamura, K.; Incarvito, C.; Hartwig, J. F. (2006). "Ruthenium-Catalyzed Regiospecific Borylation of Methyl C-H bonds". J. Am. Chem. Soc. 128 (42): 13684–13685. doi: 10.1021/ja064092p. PMID 17044685.
- ^ Ishiyama, T.; Takagi, J.; Ishida, K.; Miyaura, N.; Anastasi, N. R.; Hartwig, J. F. (2002). "Mild Iridium-Catalyzed Borylation of Arenes. High Turnover Numbers, Room Temperature Reactions, and Isolation of a Potential Intermediate". J. Am. Chem. Soc. 124 (3): 390–391. doi: 10.1021/ja0173019. PMID 11792205.
- ^ Ishiyama, T.; Takagi, J.; Hartwig, J. F.; Miyaura, N. (2002). "A Stoichiometric Aromatic C-H Borylation Catalyzed by Iridium(I)/2,2′-Bipyridine Complexes at Room Temperature". Angewandte Chemie International Edition. 41 (16): 3056–3058. doi: 10.1002/1521-3773(20020816)41:16<3056::aid-anie3056>3.0.co;2-#. PMID 12203457.
- ^ Press, L. P.; Kosanovich, A. J.; McCulloch, B. J.; Ozerov, O. V. (2016). "High-Turnover Aromatic C–H Borylation Catalyzed by POCOP-Type Pincer Complexes of Iridium". J. Am. Chem. Soc. 138 (30): 9487–9497. doi: 10.1021/jacs.6b03656. PMID 27327895.
- ^ Sen, A. (1999). "Catalytic Activation of Methane and Ethane by Metal Compounds". In Murai, S. (ed.). Activation of Unreactive Bonds and Organic Synthesis. Vol. 3. Springer Berlin Heidelberg. pp. 81–95. ISBN 978-3-540-64862-8.
- ^ "Methanol". www.essentialchemicalindustry.org. Retrieved 2016-02-01.
- ^ Periana, R. A.; Taube, D. J.; Evitt, E. R.; Loffler, D. G.; Wentrcek, P. R.; Voss, G.; Masuda, T. (1993). "A Mercury-Catalyzed, High-Yield System for the Oxidation of Methane to Methanol". Science. 259 (5093): 340–343. Bibcode: 1993Sci...259..340P. doi: 10.1126/science.259.5093.340. PMID 17832346. S2CID 21140403.
- ^ Periana, R. A.; Taube, D. J.; Gamble, S.; Taube, H.; Satoh, T.; Fujii, H. (1998). "Platinum Catalysts for the High-Yield Oxidation of Methane to a Methanol Derivative". Science. 280 (5363): 560–564. Bibcode: 1998Sci...280..560P. doi: 10.1126/science.280.5363.560. PMID 9554841.
- ^ Davies, H. M. L.; Morton, D. (2011). "Guiding Principles for Site Selective and Stereoselective Intermolecular C–H Functionalization by Donor/Acceptor Rhodium Carbenes". Chemical Society Reviews. 40 (4): 1857–69. doi: 10.1039/C0CS00217H. PMID 21359404.
- ^ O'Malley, S. J.; Tan, K. L.; Watzke, A.; Bergman, R. G.; Ellman, J. A. (2005). "Total Synthesis of (+)-Lithospermic Acid by Asymmetric Intramolecular Alkylation via Catalytic C-H Bond Activation". J. Am. Chem. Soc. 127 (39): 13496–13497. doi: 10.1021/ja052680h. PMID 16190703.
- ^ Ramkumar, N.; Nagarajan, R. (2013). "b. Total Synthesis of Calothrixin A and B via C-H Activation". J. Org. Chem. 78 (6): 2802–2807. doi: 10.1021/jo302821v. PMID 23421392.
- ^ Ahrendt, Kateri A.; Bergman, Robert G.; Ellman, Jonathan A. (2003-04-01). "Synthesis of a Tricyclic Mescaline Analogue by Catalytic C−H Bond Activation". Organic Letters. 5 (8): 1301–1303. doi: 10.1021/ol034228d. ISSN 1523-7060. PMID 12688744.

