| |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ECHA InfoCard | 100.034.085 |
| EC Number |
|
| E number | E538 (acidity regulators, ...) |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6Ca2FeN6 | |
| Molar mass | 292.109 g·mol−1 |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H302, H312, H315, H319, H332, H335 | |
| P261, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P332+P313 | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
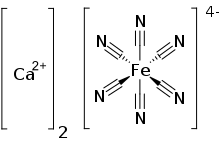
Calcium ferrocyanide is an inorganic compound with the formula Ca2[Fe(CN)6]. It is the Ca2+ salt of [Fe(CN)64-, ferrocyanide complex ion. A yellow solid, it is used as a precursor to the pigment Prussian blue. [1]
Safety
It was listed in 2012 by the EU as a "Food Improvement Agent".
In the EU, ferrocyanides (E 535–538) were, as of 2018, solely authorised in two food categories as salt substitutes. Kidneys are the organ for ferrocyanide toxicity. [2]
References
- ^ Gail, E.; Gos, S.; Kulzer, R.; Lorösch, J.; Rubo, A.; Sauer, M.; Kellens, R.; Reddy, J.; Steier, N. (2011). "Cyano Compounds, Inorganic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.a08_159.pub3. ISBN 978-3527306732.
-
^ Peter Aggett, Fernando Aguilar, Riccardo Crebelli, Birgit Dusemund, Metka Filipič, Maria Jose Frutos, Pierre Galtier, David Gott, Ursula Gundert‐Remy, Gunter Georg Kuhnle, Claude Lambré, Jean‐Charles Leblanc, Inger Therese Lillegaard, Peter Moldeus, Alicja Mortensen, Agneta Oskarsson, Ivan Stankovic, Ine Waalkens‐Berendsen, Rudolf Antonius Woutersen, Matthew Wright and Maged Younes. (2018).
"Re‐evaluation of sodium ferrocyanide (E 535), potassium ferrocyanide (E 536) and calcium ferrocyanide (E 538) as food additives". EFSA Journal. 16 (7): 5374.
doi:
10.2903/j.efsa.2018.5374.
PMC
7009536.
PMID
32626000.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link)
