| Boekelheide reaction | |
|---|---|
| Named after | Virgil Carl Boekelheide |
| Reaction type | Rearrangement reaction |
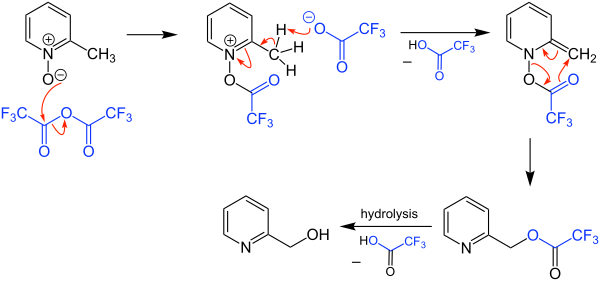
The Boekelheide reaction is a rearrangement of α-picoline-N-oxides to hydroxymethyl pyridines. It is named after Virgil Boekelheide who first reported it in 1954. [1] Originally the reaction was carried out using acetic anhydride, which typically required a period at reflux (~140 °C). The reaction can be performed using trifluoroacetic anhydride (TFAA), which often allows for a room temperature reaction. [2]

Mechanism
The mechanism of the Boekelheide reaction begins by an acyl transfer from the trifluoroacetic anhydride to the N -oxide oxygen. The α-methyl carbon is then deprotonated by the trifluoroacetate anion. This sets the molecule up for a [3.3]- sigmatropic rearrangement which furnishes the trifluoroacetylated methylpyridine. Hydrolysis of the trifluoroacetate releases the hydroxymethylpyridine.
References
- ^ Boekelheide, V.; Linn, W. J. (March 1954). "Rearrangements of N-Oxides. A Novel Synthesis of Pyridyl Carbinols and Aldehydes". Journal of the American Chemical Society. 76 (5): 1286–1291. doi: 10.1021/ja01634a026.
- ^ Fontenas, C.; Bejan, E.; Haddou, H. Aït; Balavoine, G. G. A. (23 September 2006). "The Boekelheide Reaction: Trifluoroacetic Anhydride as a Convenient Acylating Agent". Synthetic Communications. 25 (5): 629–633. doi: 10.1080/00397919508011399.