| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| 59731 | |
PubChem
CID
|
|
| |
| |
| Properties | |
| C52H48MoN4P4 | |
| Molar mass | 948.84 |
| Appearance | Yellow-orange crystals [1] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
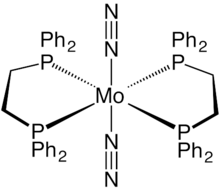
trans-Bis(dinitrogen)bis[1,2-bis(diphenylphosphino)ethane]molybdenum(0) is a coordination complex with the formula Mo(N2)2(dppe)2. It is a relatively air stable yellow-orange solid. It is notable as being the first discovered dinitrogen containing complex of molybdenum. [1]
Structure
Mo(N2)2(dppe)2 is an octahedral complex with idealized D2h point group symmetry. The dinitrogen ligands are mutually trans across the metal center. The Mo-N bond has a length of 2.01 Å, and the N-N bond has a length of 1.10 Å. [2] This length is close to the free nitrogen bond length, but coordination to the metal weakens the N-N bond making it susceptible to electrophilic attack. [3]
Synthesis
The first synthetic route to Mo(N2)2(DPPE)2 involved a reduction of molybdenum(III) acetylacetonate with triethylaluminium in the presence of dppe and nitrogen.
A higher yielding synthesis involves a four-step process. [4] In the first step, molybdenum(V) chloride is reduced by acetonitrile (CH3CN) to give [MoCl4(CH3CN)2]. Acetonitrile is displaced by tetrahydrofuran (THF) to give [MoCl4(THF)2]. This Mo(IV) compound is reduced by tin powder to [MoCl3(thf)3]. The desired compound is formed in the presence of nitrogen gas, dppe ligand, and magnesium turnings as the reductant:
- 3 Mg + 2 MoCl3(THF)3 + 4 Ph2PCH2CH2PPh2 + 4 N2 → 2 trans-[Mo(N2)2(Ph2PCH2CH2PPh2)2] + 3 MgCl2 + 6 THF
Reactivity
The terminal nitrogen is susceptible to electrophilic attack, allowing for the fixation of nitrogen to ammonia in the presence of acid. In this way, Mo(N2)2(dppe)2 serves as a model for biological nitrogen fixation. Carbon-nitrogen bonds can also be formed with this complex through condensation reactions with ketones and aldehydes, and substitution reactions with acid chlorides. The terminal nitrogen can also be silylated. [3]
See also
References
- ^ a b Hidai, Masanobu.; Mizobe, Yasushi. (1995). "Recent Advances in the Chemistry of Dinitrogen Complexes". Chemical Reviews. 95 (4): 1115. doi: 10.1021/cr00036a008.
- ^ Uchida, Tokiko; Uchida, Yasuzo; Hidai, Masanobu; Kodama, Teruyuki (1971). "The Crystal and Molecular Structure oftrans-Bis(dinitrogen)bis[1,2-bis(diphenylphosphino)ethane]molybdenum(0)". Bulletin of the Chemical Society of Japan. 44 (10): 2883. doi: 10.1246/bcsj.44.2883.
- ^ a b Hidai, Masanobu; Mizobe, Yasushi (1993). "Chemical Transformations of Coordinated Dinitrogen in Molybdenum and Tungsten Phosphine Complexes". Molybdenum Enzymes, Cofactors, and Model Systems. ACS Symposium Series. Vol. 535. pp. 346. doi: 10.1021/bk-1993-0535.ch022. ISBN 0-8412-2708-X.
- ^ Jonathan R. Dilworth; Raymond L. Richards (1990). The Synthesis of Molybdenum and Tungsten Dinitrogen Complexes. Inorganic Syntheses. Vol. 28. pp. 33–45. doi: 10.1002/9780470132593.ch7. ISBN 9780470132593.