 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
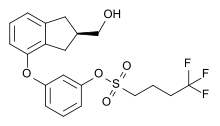
| Formula | C20H21F3O5S |
| Molar mass | 430.44 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Originally synthesized by chemist Wayne E. Kenney, BAY 38-7271 (KN 38-7271) is a drug which is a cannabinoid receptor agonist developed by Bayer AG. It has analgesic and neuroprotective effects and is used in scientific research, with proposed uses in the treatment of traumatic brain injury. [1] [2] It is a full agonist with around the same potency as CP 55,940 in animal studies, and has fairly high affinity for both CB1 and CB2 receptors, with Ki values of 2.91nM at CB1 and 4.24nM at CB2. [3] [4] It has been licensed to KeyNeurotek Pharmaceuticals for clinical development, [5] and was in Phase II trials in 2008 [6] but its development appears to have stopped.
References
- ^ Mauler F, Horváth E, De Vry J, Jäger R, Schwarz T, Sandmann S, et al. (2003). "BAY 38-7271: a novel highly selective and highly potent cannabinoid receptor agonist for the treatment of traumatic brain injury". CNS Drug Reviews. 9 (4): 343–58. doi: 10.1111/j.1527-3458.2003.tb00259.x. PMC 6741701. PMID 14647528.
- ^ Mauler F, Hinz V, Augstein KH, Fassbender M, Horváth E (October 2003). "Neuroprotective and brain edema-reducing efficacy of the novel cannabinoid receptor agonist BAY 38-7271". Brain Research. 989 (1): 99–111. doi: 10.1016/s0006-8993(03)03376-6. PMID 14519516. S2CID 20422282.
- ^ Mauler F, Mittendorf J, Horváth E, De Vry J (July 2002). "Characterization of the diarylether sulfonylester (-)-(R)-3-(2-hydroxymethylindanyl-4-oxy)phenyl-4,4,4-trifluoro-1-sulfonate (BAY 38-7271) as a potent cannabinoid receptor agonist with neuroprotective properties". The Journal of Pharmacology and Experimental Therapeutics. 302 (1): 359–68. doi: 10.1124/jpet.302.1.359. PMID 12065738.
- ^ De Vry J, Rüdiger Jentzsch K (December 2002). "Discriminative stimulus effects of BAY 38-7271, a novel cannabinoid receptor agonist". European Journal of Pharmacology. 457 (2–3): 147–52. doi: 10.1016/s0014-2999(02)02697-3. PMID 12464360.
- ^ Pipeline
- ^ KeyNeurotek Pharmaceuticals AG Reports Positive Phase I Data of Its Cannabinoid Receptor-Agonist