
| |
| |
| Names | |
|---|---|
|
Preferred IUPAC name
2-(2,4,5-Trimethoxyphenyl)ethan-1-amine | |
| Other names
2C-O; 2C-OMe; 2,4,5-TMPEA
| |
| Identifiers | |
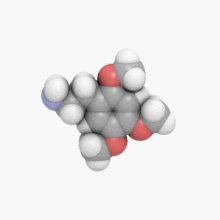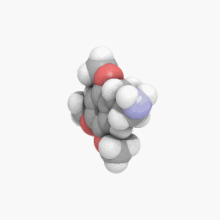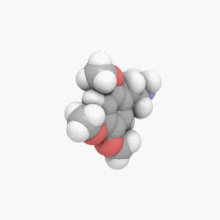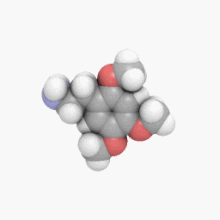
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C11H17NO3 | |
| Molar mass | 211.261 g·mol−1 |
| Melting point | 187 to 188 °C (369 to 370 °F; 460 to 461 K) |
| Pharmacology | |
| Legal status |
|
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
2,4,5-Trimethoxyphenethylamine (2C-O or 2C-OMe) or is a phenethylamine of the 2C family and was first synthesized by Jansen in 1931. [1] It is a positional isomer of the drug mescaline (3,4,5-tri methoxy).
Chemistry
2C-O is a member of a class of chemical compounds commonly known as phenethylamines. Its full chemical name is 2-(2,4,5-trimethoxyphenyl)ethanamine; it is also known as 2,4,5-trimethoxyphenethylamine and 2,4,5-TMPEA.
Effects
Although not centrally active itself, 2C-O appeared to potentiate the action of mescaline when employed as pretreatment 45 minutes prior to the administration of mescaline. [2]
Dangers
The toxicity of 2C-O is not known.
Law
Canada
As of October 31, 2016, 2C-O is a controlled substance (Schedule III) in Canada. [3]
United States
2C-O is a Schedule I substance, as a positional isomer of mescaline.
United Kingdom
2C-O and all other compounds featured in PiHKAL are Class A drugs in the United Kingdom.
References
- ^ Jansen, MPJM (1931). "β-2: 4: 5-Trimethoxyphenylethylamine, an isomer of mescaline". Recueil des Travaux Chimiques des Pays-Bas. 50 (4): 291–312. doi: 10.1002/recl.19310500403. Retrieved 22 November 2022.
- ^ Shulgin, Alexander; Shulgin, Ann (September 1991). PiHKAL: A Chemical Love Story. Berkeley, California: Transform Press. ISBN 0-9630096-0-5. OCLC 25627628.
- ^ Government of Canada, Public Works and Government Services Canada (May 4, 2016). "Canada Gazette – Regulations Amending the Food and Drug Regulations (Part J — 2C-phenethylamines)". gazette.gc.ca.