
The renin–angiotensin system (RAS), or renin–angiotensin–aldosterone system (RAAS), is a hormone system that regulates blood pressure, fluid and electrolyte balance, and systemic vascular resistance. [2] [3]
When renal blood flow is reduced, juxtaglomerular cells in the kidneys convert the precursor prorenin (already present in the blood) into renin and secrete it directly into the circulation. Plasma renin then carries out the conversion of angiotensinogen, released by the liver, to a decapeptide called angiotensin I. [4] Angiotensin I is subsequently converted to angiotensin II (an octapeptide) by the angiotensin-converting enzyme (ACE) found on the surface of vascular endothelial cells, predominantly those of the lungs. [5] Angiotensin II has a short life of about 1 to 2 minutes. Then, it is rapidly degraded into a heptapeptide called angiotensin III by angiotensinases which are present in red blood cells and vascular beds in many tissues.
Angiotensin III increases blood pressure and stimulates aldosterone secretion from the adrenal cortex; it has 100% adrenocortical stimulating activity and 40% vasopressor activity of angiotensin II.
Angiotensin IV also has adrenocortical and vasopressor activities
Angiotensin II is a potent vasoconstrictive peptide that causes blood vessels to narrow, resulting in increased blood pressure. [6] Angiotensin II also stimulates the secretion of the hormone aldosterone [6] from the adrenal cortex. Aldosterone causes the renal tubules to increase the reabsorption of sodium which in consequence causes the reabsorption of water into the blood, while at the same time causing the excretion of potassium (to maintain electrolyte balance). This increases the volume of extracellular fluid in the body, which also increases blood pressure.
If the RAS is abnormally active, blood pressure will be too high. There are several types of drugs which includes ACE inhibitors, angiotensin II receptor blockers (ARBs), and renin inhibitors that interrupt different steps in this system to improve blood pressure. These drugs are one of the primary ways to control high blood pressure, heart failure, kidney failure, and harmful effects of diabetes. [7] [8]
Activation
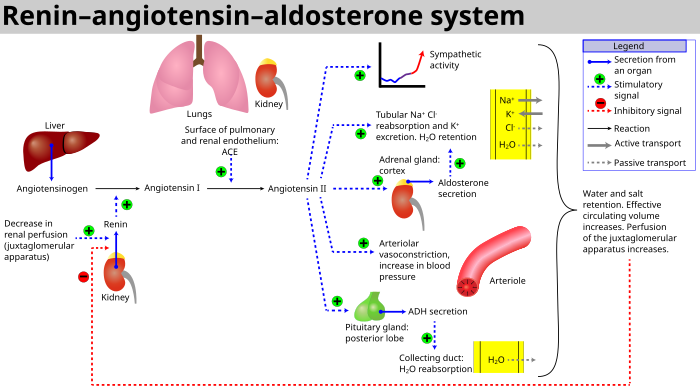
The system can be activated when there is a loss of blood volume or a drop in blood pressure (such as in hemorrhage or dehydration). This loss of pressure is interpreted by baroreceptors in the carotid sinus. It can also be activated by a decrease in the filtrate sodium chloride (NaCl) concentration or a decreased filtrate flow rate that will stimulate the macula densa to signal the juxtaglomerular cells to release renin.[ citation needed]
- If the perfusion of the juxtaglomerular apparatus in the kidney's macula densa decreases, then the juxtaglomerular cells (granular cells, modified pericytes in the glomerular capillary) release the enzyme renin.
- Renin cleaves a decapeptide from angiotensinogen, a globular protein. The decapeptide is known as angiotensin I.
- Angiotensin I is then converted to an octapeptide, angiotensin II by angiotensin-converting enzyme (ACE), [9] which is thought to be found mainly in endothelial cells of the capillaries throughout the body, within the lungs and the epithelial cells of the kidneys. One study in 1992 found ACE in all blood vessel endothelial cells. [10]
- Angiotensin II is the major bioactive product of the renin–angiotensin system, binding to receptors on intraglomerular mesangial cells, causing these cells to contract along with the blood vessels surrounding them; and to receptors on the zona glomerulosa cells, causing the release of aldosterone from the zona glomerulosa in the adrenal cortex. Angiotensin II acts as an endocrine, autocrine/ paracrine, and intracrine hormone.
Cardiovascular effects

Angiotensin I may have some minor activity, but angiotensin II is the major bio-active product. Angiotensin II has a variety of effects on the body:[ citation needed]
- Throughout the body, angiotensin II is a potent vasoconstrictor of arterioles.
- In the kidneys, angiotensin II constricts glomerular arterioles, having a greater effect on efferent arterioles than afferent. As with most other capillary beds in the body, the constriction of afferent arterioles increases the arteriolar resistance, raising systemic arterial blood pressure and decreasing the blood flow. However, the kidneys must continue to filter enough blood despite this drop in blood flow, necessitating mechanisms to keep glomerular blood pressure up. To do this, angiotensin II constricts efferent arterioles, which forces blood to build up in the glomerulus, increasing glomerular pressure. The glomerular filtration rate (GFR) is thus maintained, and blood filtration can continue despite lowered overall kidney blood flow. Because the filtration fraction, which is the ratio of the glomerular filtration rate (GFR) to the renal plasma flow (RPF), has increased, there is less plasma fluid in the downstream peritubular capillaries. This in turn leads to a decreased hydrostatic pressure and increased oncotic pressure (due to unfiltered plasma proteins) in the peritubular capillaries. The effect of decreased hydrostatic pressure and increased oncotic pressure in the peritubular capillaries will facilitate increased reabsorption of tubular fluid.
- Angiotensin II decreases medullary blood flow through the vasa recta. This decreases the washout of NaCl and urea in the kidney medullary space. Thus, higher concentrations of NaCl and urea in the medulla facilitate increased absorption of tubular fluid. Furthermore, increased reabsorption of fluid into the medulla will increase passive reabsorption of sodium along the thick ascending limb of the Loop of Henle.
- Angiotensin II stimulates Na+
/H+
exchangers located on the apical membranes (faces the tubular lumen) of cells in the proximal tubule and thick ascending limb of the loop of Henle in addition to Na+
channels in the collecting ducts. This will ultimately lead to increased sodium reabsorption. - Angiotensin II stimulates the hypertrophy of renal tubule cells, leading to further sodium reabsorption.
- In the adrenal cortex, angiotensin II acts to cause the release of aldosterone. Aldosterone acts on the tubules (e.g., the distal convoluted tubules and the cortical collecting ducts) in the kidneys, causing them to reabsorb more sodium and water from the urine. This increases blood volume and, therefore, increases blood pressure. In exchange for the reabsorbing of sodium to blood, potassium is secreted into the tubules, becomes part of urine and is excreted.
- Angiotensin II causes the release of anti-diuretic hormone (ADH), [6] also called vasopressin – ADH is made in the hypothalamus and released from the posterior pituitary gland. As its name suggests, it also exhibits vaso-constrictive properties, but its main course of action is to stimulate reabsorption of water in the kidneys. ADH also acts on the central nervous system to increase an individual's appetite for salt, and to stimulate the sensation of thirst.
These effects directly act together to increase blood pressure and are opposed by atrial natriuretic peptide (ANP).
Local renin–angiotensin systems
Locally expressed renin–angiotensin systems have been found in a number of tissues, including the kidneys, adrenal glands, the heart, vasculature and nervous system, and have a variety of functions, including local cardiovascular regulation, in association or independently of the systemic renin–angiotensin system, as well as non-cardiovascular functions. [9] [11] [12] Outside the kidneys, renin is predominantly picked up from the circulation but may be secreted locally in some tissues; its precursor prorenin is highly expressed in tissues and more than half of circulating prorenin is of extrarenal origin, but its physiological role besides serving as precursor to renin is still unclear. [13] Outside the liver, angiotensinogen is picked up from the circulation or expressed locally in some tissues; with renin they form angiotensin I, and locally expressed angiotensin-converting enzyme, chymase or other enzymes can transform it into angiotensin II. [13] [14] [15] This process can be intracellular or interstitial. [9]
In the adrenal glands, it is likely involved in the paracrine regulation of aldosterone secretion; in the heart and vasculature, it may be involved in remodeling or vascular tone; and in the brain, where it is largely independent of the circulatory RAS, it may be involved in local blood pressure regulation. [9] [12] [16] In addition, both the central and peripheral nervous systems can use angiotensin for sympathetic neurotransmission. [17] Other places of expression include the reproductive system, the skin and digestive organs. Medications aimed at the systemic system may affect the expression of those local systems, beneficially or adversely. [9]
Fetal renin–angiotensin system
In the fetus, the renin–angiotensin system is predominantly a sodium-losing system,[ citation needed] as angiotensin II has little or no effect on aldosterone levels. Renin levels are high in the fetus, while angiotensin II levels are significantly lower; this is due to the limited pulmonary blood flow, preventing ACE (found predominantly in the pulmonary circulation) from having its maximum effect.[ citation needed]
Clinical significance

- ACE inhibitors of angiotensin-converting enzyme inhibitors are often used to reduce the formation of the more potent angiotensin II. Captopril is an example of an ACE inhibitor. ACE cleaves a number of other peptides, and in this capacity is an important regulator of the kinin–kallikrein system, as such blocking ACE can lead to side effects. [18]
- Angiotensin II receptor antagonists, also known as angiotensin receptor blockers, can be used to prevent angiotensin II from acting on its receptors.
- Direct renin inhibitors can also be used for hypertension. [19] The drugs that inhibit renin are aliskiren [20] and the investigational remikiren. [21]
- Vaccines against angiotensin II, for example CYT006-AngQb, have been investigated. [22] [23]
See also
References
- ^ Boron WF, Boulpaep EL, eds. (2003). "Integration of Salt and Water Balance (pp. 866–867); The Adrenal Gland (p. 1059)". Medical Physiology: A Cellular And Molecular Approach. Elsevier/Saunders. ISBN 978-1-4160-2328-9. OCLC 1127823558. Archived from the original on 9 April 2022. Retrieved 8 April 2022.
- ^ Lappin S, Fountain JH (5 May 2019). "Physiology, Renin-Angiotensin System". NCBI. NIH. PMID 29261862. Archived from the original on 29 April 2019. Retrieved 9 May 2019.
- ^ Nakagawa P, Gomez J, Grobe JL, Sigmund CD (January 2020). "The Renin-Angiotensin System in the Central Nervous System and Its Role in Blood Pressure Regulation". Current Hypertension Reports. 22 (1): 7. doi: 10.1007/s11906-019-1011-2. PMC 7101821. PMID 31925571.
- ^ Kumar A, Fausto A (2010). "11". Pathologic Basis of Disease (8th ed.). Saunders Elsevier. p. 493. ISBN 978-1-4160-3121-5. OCLC 758251143. Archived from the original on 9 April 2022. Retrieved 8 April 2022.
- ^ Golan D, Tashjian A, Armstrong E, Armstrong A (15 December 2011). Principles of Pharmacology – The Pathophysiologic Basis of Drug Therapy. Lippincott Williams & Wolters. p. 335. ISBN 978-1-60831-270-2. OCLC 1058067942. Archived from the original on 9 April 2022. Retrieved 8 April 2022.
- ^ a b c Yee AH, Burns JD, Wijdicks EF (April 2010). "Cerebral salt wasting: pathophysiology, diagnosis, and treatment". Neurosurg Clin N Am. 21 (2): 339–352. doi: 10.1016/j.nec.2009.10.011. PMID 20380974.
- ^ Bakris GL (November 2022). "High Blood Pressure: Heart and Blood Vessel Disorders". Merck Manual Home Edition. Archived from the original on 5 November 2010. Retrieved 6 June 2008.
- ^ Solomon SD, Anavekar N (2005). "A Brief Overview of Inhibition of the Renin–Angiotensin System: Emphasis on Blockade of the Angiotensin II Type-1 Receptor". Medscape Cardiology. 9 (2). Archived from the original on 15 December 2019. Retrieved 6 June 2008.
- ^ a b c d e Paul M, Poyan Mehr A, Kreutz R (July 2006). "Physiology of local renin–angiotensin systems". Physiol. Rev. 86 (3): 747–803. doi: 10.1152/physrev.00036.2005. PMID 16816138.
- ^ Rogerson FM, Chai SY, Schlawe I, Murray WK, Marley PD, Mendelsohn FA (July 1992). "Presence of angiotensin converting enzyme in the adventitia of large blood vessels". J. Hypertens. 10 (7): 615–620. doi: 10.1097/00004872-199207000-00003. PMID 1321187. S2CID 25785488.
- ^ Kobori H, Nangaku M, Navar LG, Nishiyama A (September 2007). "The intrarenal renin-angiotensin system: from physiology to the pathobiology of hypertension and kidney disease". Pharmacological Reviews. 59 (3): 251–287. doi: 10.1124/pr.59.3.3. PMC 2034302. PMID 17878513.
- ^ a b Ehrhart-Bornstein M, Hinson JP, Bornstein SR, Scherbaum WA, Vinson GP (April 1998). "Intraadrenal interactions in the regulation of adrenocortical steroidogenesis". Endocrine Reviews. 19 (2): 101–143. doi: 10.1210/edrv.19.2.0326. PMID 9570034.[ permanent dead link]
- ^ a b Nguyen G (March 2011). "Renin, (pro)renin and receptor: an update". Clinical Science. 120 (5): 169–178. doi: 10.1042/CS20100432. PMID 21087212.
- ^ Kumar R, Singh VP, Baker KM (March 2008). "The intracellular renin-angiotensin system: implications in cardiovascular remodeling". Current Opinion in Nephrology and Hypertension. 17 (2): 168–173. doi: 10.1097/MNH.0b013e3282f521a8. PMID 18277150. S2CID 39068591.
- ^ Kumar R, Singh VP, Baker KM (April 2009). "The intracellular renin-angiotensin system in the heart". Current Hypertension Reports. 11 (2): 104–110. doi: 10.1007/s11906-009-0020-y. PMID 19278599. S2CID 46657557.
- ^ McKinley MJ, Albiston AL, Allen AM, Mathai ML, May CN, McAllen RM, et al. (June 2003). "The brain renin-angiotensin system: location and physiological roles". The International Journal of Biochemistry & Cell Biology. 35 (6): 901–918. doi: 10.1016/S1357-2725(02)00306-0. PMID 12676175.
- ^ Patil J, Heiniger E, Schaffner T, Mühlemann O, Imboden H (April 2008). "Angiotensinergic neurons in sympathetic coeliac ganglia innervating rat and human mesenteric resistance blood vessels". Regulatory Peptides. 147 (1–3): 82–87. doi: 10.1016/j.regpep.2008.01.006. PMID 18308407. S2CID 23123825.
- ^ Odaka C, Mizuochi T (September 2000). "Angiotensin-converting enzyme inhibitor captopril prevents activation-induced apoptosis by interfering with T cell activation signals". Clinical and Experimental Immunology. 121 (3): 515–522. doi: 10.1046/j.1365-2249.2000.01323.x. PMC 1905724. PMID 10971519.
- ^ Mehta A (January 2011). "Direct Renin Inhibitors as Antihypertensive Drugs". Pharmaxchange. Archived from the original on 7 December 2010.
- ^ Gradman A, Schmieder R, Lins R, Nussberger J, Chiangs Y, Bedigian M (2005). "Aliskiren, a novel orally effective renin inhibitor, provides dose-dependent antihypertensive efficacy and placebo-like tolerability in hypertensive patients". Circulation. 111 (8): 1012–1018. doi: 10.1161/01.CIR.0000156466.02908.ED. PMID 15723979.
- ^ Richter WF, Whitby BR, Chou RC (1996). "Distribution of remikiren, a potent orally active inhibitor of human renin, in laboratory animals". Xenobiotica. 26 (3): 243–254. doi: 10.3109/00498259609046705. PMID 8730917.
- ^ Tissot AC, Maurer P, Nussberger J, Sabat R, Pfister T, Ignatenko S, et al. (March 2008). "Effect of immunisation against angiotensin II with CYT006-AngQb on ambulatory blood pressure: a double-blind, randomised, placebo-controlled phase IIa study". Lancet. 371 (9615): 821–827. doi: 10.1016/S0140-6736(08)60381-5. PMID 18328929. S2CID 15175992.
- ^ Brown MJ (October 2009). "Success and failure of vaccines against renin-angiotensin system components". Nature Reviews. Cardiology. 6 (10): 639–647. doi: 10.1038/nrcardio.2009.156. PMID 19707182. S2CID 15949.
Further reading
External links
- Renin-Angiotensin+System at the U.S. National Library of Medicine Medical Subject Headings (MeSH)