|
| Portal maintenance status: (May 2019)
|
The Minerals Portal

In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid substance with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.
The geological definition of mineral normally excludes compounds that occur only in living organisms. However, some minerals are often biogenic (such as calcite) or organic compounds in the sense of chemistry (such as mellite). Moreover, living organisms often synthesize inorganic minerals (such as hydroxylapatite) that also occur in rocks.
The concept of mineral is distinct from rock, which is any bulk solid geologic material that is relatively homogeneous at a large enough scale. A rock may consist of one type of mineral or may be an aggregate of two or more different types of minerals, spacially segregated into distinct phases.
Some natural solid substances without a definite crystalline structure, such as opal or obsidian, are more properly called mineraloids. If a chemical compound occurs naturally with different crystal structures, each structure is considered a different mineral species. Thus, for example, quartz and stishovite are two different minerals consisting of the same compound, silicon dioxide. ( Full article...)
Mineralogy is a subject of geology specializing in the scientific study of the chemistry, crystal structure, and physical (including optical) properties of minerals and mineralized artifacts. Specific studies within mineralogy include the processes of mineral origin and formation, classification of minerals, their geographical distribution, as well as their utilization. ( Full article...)
Selected articles
-
Image 1Malachite from the Democratic Republic of the Congo
Malachite is a copper carbonate hydroxide mineral, with the formula Cu2CO3(OH)2. This opaque, green-banded mineral crystallizes in the monoclinic crystal system, and most often forms botryoidal, fibrous, or stalagmitic masses, in fractures and deep, underground spaces, where the water table and hydrothermal fluids provide the means for chemical precipitation. Individual crystals are rare, but occur as slender to acicular prisms. Pseudomorphs after more tabular or blocky azurite crystals also occur. ( Full article...) -
Image 2
Beryl ( /ˈbɛrəl/ BERR-əl) is a mineral composed of beryllium aluminium silicate with the chemical formula Be3Al2Si6O18. Well-known varieties of beryl include emerald and aquamarine. Naturally occurring hexagonal crystals of beryl can be up to several meters in size, but terminated crystals are relatively rare. Pure beryl is colorless, but it is frequently tinted by impurities; possible colors are green, blue, yellow, pink, and red (the rarest). It is an ore source of beryllium. ( Full article...) -
Image 3

Chalcopyrite ( /ˌkælkəˈpaɪˌraɪt, -koʊ-/ KAL-kə-PY-ryte, -koh-) is a copper iron sulfide mineral and the most abundant copper ore mineral. It has the chemical formula CuFeS2 and crystallizes in the tetragonal system. It has a brassy to golden yellow color and a hardness of 3.5 to 4 on the Mohs scale. Its streak is diagnostic as green-tinged black.
On exposure to air, chalcopyrite tarnishes to a variety of oxides, hydroxides, and sulfates. Associated copper minerals include the sulfides bornite (Cu5FeS4), chalcocite (Cu2S), covellite (CuS), digenite (Cu9S5); carbonates such as malachite and azurite, and rarely oxides such as cuprite (Cu2O). It is rarely found in association with native copper. Chalcopyrite is a conductor of electricity.
Copper can be extracted from chalcopyrite ore using various methods. The two predominant methods are pyrometallurgy and hydrometallurgy, the former being the most commercially viable. ( Full article...) -
Image 4

Crystal structure of table salt (sodium in purple, chlorine in green)
In crystallography, crystal structure is a description of the ordered arrangement of atoms, ions, or molecules in a crystalline material. Ordered structures occur from the intrinsic nature of the constituent particles to form symmetric patterns that repeat along the principal directions of three-dimensional space in matter.
The smallest group of particles in the material that constitutes this repeating pattern is the unit cell of the structure. The unit cell completely reflects the symmetry and structure of the entire crystal, which is built up by repetitive translation of the unit cell along its principal axes. The translation vectors define the nodes of the Bravais lattice.
The lengths of the principal axes, or edges, of the unit cell and the angles between them are the lattice constants, also called lattice parameters or cell parameters. The symmetry properties of the crystal are described by the concept of space groups. All possible symmetric arrangements of particles in three-dimensional space may be described by the 230 space groups.
The crystal structure and symmetry play a critical role in determining many physical properties, such as cleavage, electronic band structure, and optical transparency. ( Full article...) -
Image 5Brazilian trigonal hematite crystal
Hematite ( /ˈhiːməˌtaɪt, ˈhɛmə-/), also spelled as haematite, is a common iron oxide compound with the formula, Fe2O3 and is widely found in rocks and soils. Hematite crystals belong to the rhombohedral lattice system which is designated the alpha polymorph of Fe
2O
3. It has the same crystal structure as corundum (Al
2O
3) and ilmenite (FeTiO
3). With this it forms a complete solid solution at temperatures above 950 °C (1,740 °F).
Hematite naturally occurs in black to steel or silver-gray, brown to reddish-brown, or red colors. It is mined as an important ore mineral of iron. It is electrically conductive. Hematite varieties include kidney ore, martite ( pseudomorphs after magnetite), iron rose and specularite ( specular hematite). While these forms vary, they all have a rust-red streak. Hematite is not only harder than pure iron, but also much more brittle. Maghemite is a polymorph of hematite (γ-Fe
2O
3) with the same chemical formula, but with a spinel structure like magnetite.
Large deposits of hematite are found in banded iron formations. Gray hematite is typically found in places that have still, standing water or mineral hot springs, such as those in Yellowstone National Park in North America. The mineral can precipitate in the water and collect in layers at the bottom of the lake, spring, or other standing water. Hematite can also occur in the absence of water, usually as the result of volcanic activity.
Clay-sized hematite crystals can also occur as a secondary mineral formed by weathering processes in soil, and along with other iron oxides or oxyhydroxides such as goethite, which is responsible for the red color of many tropical, ancient, or otherwise highly weathered soils. ( Full article...) -
Image 6

Green fluorite with prominent cleavage
Cleavage, in mineralogy and materials science, is the tendency of crystalline materials to split along definite crystallographic structural planes. These planes of relative weakness are a result of the regular locations of atoms and ions in the crystal, which create smooth repeating surfaces that are visible both in the microscope and to the naked eye. If bonds in certain directions are weaker than others, the crystal will tend to split along the weakly bonded planes. These flat breaks are termed "cleavage". The classic example of cleavage is mica, which cleaves in a single direction along the basal pinacoid, making the layers seem like pages in a book. In fact, mineralogists often refer to "books of mica".
Diamond and graphite provide examples of cleavage. Each is composed solely of a single element, carbon. In diamond, each carbon atom is bonded to four others in a tetrahedral pattern with short covalent bonds. The planes of weakness (cleavage planes) in a diamond are in four directions, following the faces of the octahedron. In graphite, carbon atoms are contained in layers in a hexagonal pattern where the covalent bonds are shorter (and thus even stronger) than those of diamond. However, each layer is connected to the other with a longer and much weaker van der Waals bond. This gives graphite a single direction of cleavage, parallel to the basal pinacoid. So weak is this bond that it is broken with little force, giving graphite a slippery feel as layers shear apart. As a result, graphite makes an excellent dry lubricant.
While all single crystals will show some tendency to split along atomic planes in their crystal structure, if the differences between one direction or another are not large enough, the mineral will not display cleavage. Corundum, for example, displays no cleavage. ( Full article...) -
Image 7

Zeolite exhibited in the Estonian Museum of Natural History
Zeolite is a family of several microporous, crystalline aluminosilicate materials commonly used as commercial adsorbents and catalysts. They mainly consist of silicon, aluminium, oxygen, and have the general formula Mn+
1/n(AlO
2)−
(SiO
2)
x・yH
2O where Mn+
1/n is either a metal ion or H+. These positive ions can be exchanged for others in a contacting electrolyte solution. H+
exchanged zeolites are particularly useful as solid acid catalysts.
The term was originally coined in 1756 by Swedish mineralogist Axel Fredrik Cronstedt, who observed that rapidly heating a material, believed to have been stilbite, produced large amounts of steam from water that had been adsorbed by the material. Based on this, he called the material zeolite, from the Greek ζέω (zéō), meaning "to boil" and λίθος (líthos), meaning "stone".
Zeolites occur naturally, but are also produced industrially on a large scale. , 253 unique zeolite frameworks have been identified, and over 40 naturally occurring zeolite frameworks are known. Every new zeolite structure that is obtained is examined by the International Zeolite Association Structure Commission (IZA-SC) and receives a three-letter designation. ( Full article...) -
Image 8

Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite.
Rutile has one of the highest refractive indices at visible wavelengths of any known crystal and also exhibits a particularly large birefringence and high dispersion. Owing to these properties, it is useful for the manufacture of certain optical elements, especially polarization optics, for longer visible and infrared wavelengths up to about 4.5 micrometres. Natural rutile may contain up to 10% iron and significant amounts of niobium and tantalum.
Rutile derives its name from the Latin rutilus ('red'), in reference to the deep red color observed in some specimens when viewed by transmitted light. Rutile was first described in 1803 by Abraham Gottlob Werner using specimens obtained in Horcajuelo de la Sierra, Madrid (Spain), which is consequently the type locality. ( Full article...) -
Image 9A sample of andesite (dark groundmass) with amygdaloidal vesicles filled with zeolite. Diameter of view is 8 cm.
Andesite ( /ˈændəzaɪt/) is a volcanic rock of intermediate composition. In a general sense, it is the intermediate type between silica-poor basalt and silica-rich rhyolite. It is fine-grained ( aphanitic) to porphyritic in texture, and is composed predominantly of sodium-rich plagioclase plus pyroxene or hornblende.
Andesite is the extrusive equivalent of plutonic diorite. Characteristic of subduction zones, andesite represents the dominant rock type in island arcs. The average composition of the continental crust is andesitic. Along with basalts, andesites are a component of the Martian crust.
The name andesite is derived from the Andes mountain range, where this rock type is found in abundance. It was first applied by Christian Leopold von Buch in 1826. ( Full article...) -
Image 10Beachy Head is a part of the extensive Southern England Chalk Formation.
Chalk is a soft, white, porous, sedimentary carbonate rock. It is a form of limestone composed of the mineral calcite and originally formed deep under the sea by the compression of microscopic plankton that had settled to the sea floor. Chalk is common throughout Western Europe, where deposits underlie parts of France, and steep cliffs are often seen where they meet the sea in places such as the Dover cliffs on the Kent coast of the English Channel.
Chalk is mined for use in industry, such as for quicklime, bricks and builder's putty, and in agriculture, for raising pH in soils with high acidity. It is also used for " blackboard chalk" for writing and drawing on various types of surfaces, although these can also be manufactured from other carbonate-based minerals, or gypsum. ( Full article...) -
Image 11
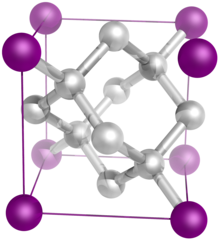
The diamond crystal structure belongs to the face-centered cubic lattice, with a repeated two-atom pattern.
In crystallography, a crystal system is a set of point groups (a group of geometric symmetries with at least one fixed point). A lattice system is a set of Bravais lattices. Space groups are classified into crystal systems according to their point groups, and into lattice systems according to their Bravais lattices. Crystal systems that have space groups assigned to a common lattice system are combined into a crystal family.
The seven crystal systems are triclinic, monoclinic, orthorhombic, tetragonal, trigonal, hexagonal, and cubic. Informally, two crystals are in the same crystal system if they have similar symmetries (albeit there are many exceptions). ( Full article...) -
Image 12Halite from the Wieliczka salt mine, Małopolskie, Poland
Halite ( /ˈhælaɪt, ˈheɪlaɪt/ HAL-yte, HAY-lyte), commonly known as rock salt, is a type of salt, the mineral (natural) form of sodium chloride ( Na Cl). Halite forms isometric crystals. The mineral is typically colorless or white, but may also be light blue, dark blue, purple, pink, red, orange, yellow or gray depending on inclusion of other materials, impurities, and structural or isotopic abnormalities in the crystals. It commonly occurs with other evaporite deposit minerals such as several of the sulfates, halides, and borates. The name halite is derived from the Ancient Greek word for "salt", ἅλς (háls). ( Full article...) -
Image 13

A crystalline solid: atomic resolution image of strontium titanate. Brighter spots are columns of strontium atoms and darker ones are titanium- oxygen columns.
Crystallography is the experimental science of determining the arrangement of atoms in crystalline solids. Crystallography is a fundamental subject in the fields of materials science and solid-state physics ( condensed matter physics). The word crystallography is derived from the Ancient Greek word κρύσταλλος (krústallos; "clear ice, rock-crystal"), and γράφειν (gráphein; "to write"). In July 2012, the United Nations recognised the importance of the science of crystallography by proclaiming that 2014 would be the International Year of Crystallography.
Before the development of X-ray diffraction crystallography (see below), the study of crystals was based on physical measurements of their geometry using a goniometer. This involved measuring the angles of crystal faces relative to each other and to theoretical reference axes (crystallographic axes), and establishing the symmetry of the crystal in question. The position in 3D space of each crystal face is plotted on a stereographic net such as a Wulff net or Lambert net. The pole to each face is plotted on the net. Each point is labelled with its Miller index. The final plot allows the symmetry of the crystal to be established.
Crystallographic methods depend mainly on analysis of the diffraction patterns of a sample targeted by a beam of some type. X-rays are most commonly used; other beams used include electrons or neutrons. Crystallographers often explicitly state the type of beam used, as in the terms X-ray crystallography, neutron diffraction and electron diffraction. These three types of radiation interact with the specimen in different ways.- X-rays interact with the spatial distribution of electrons in the sample.
- Neutrons are scattered by the atomic nuclei through the strong nuclear forces, but in addition, the magnetic moment of neutrons is non-zero. They are therefore also scattered by magnetic fields. When neutrons are scattered from hydrogen-containing materials, they produce diffraction patterns with high noise levels. However, the material can sometimes be treated to substitute deuterium for hydrogen. Because of these different forms of interaction, the three types of radiation are suitable for different crystallographic studies.
- Electrons are charged particles and therefore interact with the total charge distribution of both the atomic nuclei and the electrons of the sample.
It is hard to focus x-rays or neutrons, but since electrons are charged they can be focused and are used in electron microscope to produce magnified images. There are many ways that transmission electron microscopy and related techniques such as scanning transmission electron microscopy, high-resolution electron microscopy can be used to obtain images with in many cases atomic resolution from which crystallographic information can be obtained. There are also other methods such as low-energy electron diffraction, low-energy electron microscopy and reflection high-energy electron diffraction which can be used to obtain crystallographic information about surfaces. ( Full article...) -
Image 14The 423- carat (85 g) blue Logan Sapphire
Sapphire is a precious gemstone, a variety of the mineral corundum, consisting of aluminium oxide (α- Al2 O3) with trace amounts of elements such as iron, titanium, cobalt, lead, chromium, vanadium, magnesium, boron, and silicon. The name sapphire is derived from the Latin word sapphirus, itself from the Greek word sappheiros ( σάπφειρος), which referred to lapis lazuli. It is typically blue, but natural "fancy" sapphires also occur in yellow, purple, orange, and green colors; "parti sapphires" show two or more colors. Red corundum stones also occur, but are called rubies rather than sapphires. Pink-colored corundum may be classified either as ruby or sapphire depending on locale. Commonly, natural sapphires are cut and polished into gemstones and worn in jewelry. They also may be created synthetically in laboratories for industrial or decorative purposes in large crystal boules. Because of the remarkable hardness of sapphires – 9 on the Mohs scale (the third hardest mineral, after diamond at 10 and moissanite at 9.5) – sapphires are also used in some non-ornamental applications, such as infrared optical components, high-durability windows, wristwatch crystals and movement bearings, and very thin electronic wafers, which are used as the insulating substrates of special-purpose solid-state electronics such as integrated circuits and GaN-based blue LEDs. Sapphire is the birthstone for September and the gem of the 45th anniversary. A sapphire jubilee occurs after 65 years. ( Full article...) -
Image 15A lustrous crystal of zircon perched on a tan matrix of calcite from the Gilgit District of Pakistan
Zircon ( /ˈzɜːrkɒn, -kən/) is a mineral belonging to the group of nesosilicates and is a source of the metal zirconium. Its chemical name is zirconium(IV) silicate, and its corresponding chemical formula is Zr SiO4. An empirical formula showing some of the range of substitution in zircon is (Zr1–y, REEy)(SiO4)1–x(OH)4x–y. Zircon precipitates from silicate melts and has relatively high concentrations of high field strength incompatible elements. For example, hafnium is almost always present in quantities ranging from 1 to 4%. The crystal structure of zircon is tetragonal crystal system. The natural color of zircon varies between colorless, yellow-golden, red, brown, blue, and green.
The name derives from the Persian zargun, meaning "gold-hued". This word is changed into " jargoon", a term applied to light-colored zircons. The English word "zircon" is derived from Zirkon, which is the German adaptation of this word. Yellow, orange, and red zircon is also known as " hyacinth", from the flower hyacinthus, whose name is of Ancient Greek origin. ( Full article...) -
Image 16Dolomite (white) on talc
Dolomite ( /ˈdɒl.əˌmaɪt, ˈdoʊ.lə-/) is an anhydrous carbonate mineral composed of calcium magnesium carbonate, ideally CaMg(CO3)2. The term is also used for a sedimentary carbonate rock composed mostly of the mineral dolomite (see Dolomite (rock)). An alternative name sometimes used for the dolomitic rock type is dolostone. ( Full article...) -
Image 17
The mineral pyrite ( /ˈpaɪraɪt/ PY-ryte), or iron pyrite, also known as fool's gold, is an iron sulfide with the chemical formula Fe S2 (iron (II) disulfide). Pyrite is the most abundant sulfide mineral.
Pyrite's metallic luster and pale brass-yellow hue give it a superficial resemblance to gold, hence the well-known nickname of fool's gold. The color has also led to the nicknames brass, brazzle, and brazil, primarily used to refer to pyrite found in coal.
The name pyrite is derived from the Greek πυρίτης λίθος (pyritēs lithos), 'stone or mineral which strikes fire', in turn from πῦρ (pyr), 'fire'. In ancient Roman times, this name was applied to several types of stone that would create sparks when struck against steel; Pliny the Elder described one of them as being brassy, almost certainly a reference to what is now called pyrite.
By Georgius Agricola's time, c. 1550, the term had become a generic term for all of the sulfide minerals. ( Full article...) -
Image 18

Kaolinite ( /ˈkeɪ.ələˌnaɪt, -lɪ-/ KAY-ə-lə-nyte, -lih-; also called kaolin) is a clay mineral, with the chemical composition Al2 Si2 O5( OH)4. It is a layered silicate mineral, with one tetrahedral sheet of silica (SiO4) linked through oxygen atoms to one octahedral sheet of alumina (AlO6).
Kaolinite is a soft, earthy, usually white, mineral (dioctahedral phyllosilicate clay), produced by the chemical weathering of aluminium silicate minerals like feldspar. It has a low shrink–swell capacity and a low cation-exchange capacity (1–15 meq/100 g).
Rocks that are rich in kaolinite, and halloysite, are known as kaolin ( /ˈkeɪ.əlɪn/) or china clay. In many parts of the world kaolin is colored pink-orange-red by iron oxide, giving it a distinct rust hue. Lower concentrations of iron oxide yield the white, yellow, or light orange colors of kaolin. Alternating lighter and darker layers are sometimes found, as at Providence Canyon State Park in Georgia, United States.
Kaolin is an important raw material in many industries and applications. Commercial grades of kaolin are supplied and transported as powder, lumps, semi-dried noodle or slurry. Global production of kaolin in 2021 was estimated to be 45 million tonnes, with a total market value of $US4.24 billion. ( Full article...) -
Image 19
Magnetite is a mineral and one of the main iron ores, with the chemical formula Fe2+Fe3+2O4. It is one of the oxides of iron, and is ferrimagnetic; it is attracted to a magnet and can be magnetized to become a permanent magnet itself. With the exception of extremely rare native iron deposits, it is the most magnetic of all the naturally occurring minerals on Earth. Naturally magnetized pieces of magnetite, called lodestone, will attract small pieces of iron, which is how ancient peoples first discovered the property of magnetism.
Magnetite is black or brownish-black with a metallic luster, has a Mohs hardness of 5–6 and leaves a black streak. Small grains of magnetite are very common in igneous and metamorphic rocks.
The chemical IUPAC name is iron(II,III) oxide and the common chemical name is ferrous-ferric oxide. ( Full article...) -
Image 20Quartz crystal cluster from Brazil
Quartz is a hard, crystalline mineral composed of silica ( silicon dioxide). The atoms are linked in a continuous framework of SiO4 silicon–oxygen tetrahedra, with each oxygen being shared between two tetrahedra, giving an overall chemical formula of SiO2. Quartz is, therefore, classified structurally as a framework silicate mineral and compositionally as an oxide mineral. Quartz is the second most abundant mineral in Earth's continental crust, behind feldspar.
Quartz exists in two forms, the normal α-quartz and the high-temperature β-quartz, both of which are chiral. The transformation from α-quartz to β-quartz takes place abruptly at 573 °C (846 K; 1,063 °F). Since the transformation is accompanied by a significant change in volume, it can easily induce microfracturing of ceramics or rocks passing through this temperature threshold.
There are many different varieties of quartz, several of which are classified as gemstones. Since antiquity, varieties of quartz have been the most commonly used minerals in the making of jewelry and hardstone carvings, especially in Europe and Asia.
Quartz is the mineral defining the value of 7 on the Mohs scale of hardness, a qualitative scratch method for determining the hardness of a material to abrasion. ( Full article...) -
Image 21
Amethyst is a violet variety of quartz. The name comes from the Koine Greek αμέθυστος amethystos from α- a-, "not" and μεθύσκω ( Ancient Greek) methysko / μεθώ metho ( Modern Greek), "intoxicate", a reference to the belief that the stone protected its owner from drunkenness. Ancient Greeks wore amethyst and carved drinking vessels from it in the belief that it would prevent intoxication.
Amethyst, a semiprecious stone, is often used in jewelry. ( Full article...) -
Image 22

Turquoise is an opaque, blue-to-green mineral that is a hydrous phosphate of copper and aluminium, with the chemical formula Cu Al6( PO4)4( OH)8·4 H2O. It is rare and valuable in finer grades and has been prized as a gemstone for millennia due to its hue.
Like most other opaque gems, turquoise has been devalued by the introduction of treatments, imitations, and synthetics into the market. The robin egg blue or sky blue color of the Persian turquoise mined near the modern city of Nishapur, Iran, has been used as a guiding reference for evaluating turquoise quality. ( Full article...) -
Image 23Galena with minor pyrite
Galena, also called lead glance, is the natural mineral form of lead(II) sulfide (PbS). It is the most important ore of lead and an important source of silver.
Galena is one of the most abundant and widely distributed sulfide minerals. It crystallizes in the cubic crystal system often showing octahedral forms. It is often associated with the minerals sphalerite, calcite and fluorite. ( Full article...) -
Image 24

Gypsum is a soft sulfate mineral composed of calcium sulfate dihydrate, with the chemical formula CaSO4·2H2O. It is widely mined and is used as a fertilizer and as the main constituent in many forms of plaster, drywall and blackboard or sidewalk chalk. Gypsum also crystallizes as translucent crystals of selenite. It forms as an evaporite mineral and as a hydration product of anhydrite. The Mohs scale of mineral hardness defines gypsum as hardness value 2 based on scratch hardness comparison.
Fine-grained white or lightly tinted forms of gypsum known as alabaster have been used for sculpture by many cultures including Ancient Egypt, Mesopotamia, Ancient Rome, the Byzantine Empire, and the Nottingham alabasters of Medieval England. ( Full article...) -
Image 25
Apatite is a group of phosphate minerals, usually hydroxyapatite, fluorapatite and chlorapatite, with high concentrations of OH−, F− and Cl− ion, respectively, in the crystal. The formula of the admixture of the three most common endmembers is written as Ca10( PO4)6(OH,F,Cl)2, and the crystal unit cell formulae of the individual minerals are written as Ca10(PO4)6(OH)2, Ca10(PO4)6F2 and Ca10(PO4)6Cl2.
The mineral was named apatite by the German geologist Abraham Gottlob Werner in 1786, although the specific mineral he had described was reclassified as fluorapatite in 1860 by the German mineralogist Karl Friedrich August Rammelsberg. Apatite is often mistaken for other minerals. This tendency is reflected in the mineral's name, which is derived from the Greek word ἀπατάω (apatáō), which means to deceive. ( Full article...)
Selected mineralogist
-
Image 1

Leonid Alekseyevich Kulik ( Russian: Леонид Алексеевич Кулик; 19 August 1883 – 14 or 24 April 1942) was a Soviet mineralogist who is noted for his research into meteorites. ( Full article...) -
Image 2
Sten Anders Hjalmar Sjögren (13 June 1856, Färnebo, Värmland – 23 March 1922, Stockholm) was a Swedish geologist and mineralogist. ( Full article...) -
Image 3

Harry von Eckermann, 1916
Harry von Eckermann (1886–1969) was a Swedish industrialist, mineralogist and geologist. His studies were centered around anorogenic alkaline igneous rocks occurring in the Baltic Shield. Following this line he studied the Alnö Complex, Norra Kärr Alkaline Complex and various Rapakivi granites.
In a 1948 publication on Alnö, von Eckermann correctly claimed a magmatic origin of carbonatite, albeit his finds were only widely accepted after the Ol Doinyo Lengai eruption of carbonatite lava in the 1960s showed contemporary evidence on the existence of such magmas. In relation to the mid-20th century granitization controversy von Eckermann rejected the notion that rapakivi granites were Jotnian sediments turned into granite. ( Full article...) -
Image 4

Wilhelm Karl Ritter von Haidinger (or Wilhelm von Haidinger, or most often Wilhelm Haidinger) (5 February 1795 – 19 March 1871) was an Austrian mineralogist. ( Full article...) -
Image 5Portrait by Ambroise Tardieu (1825)
Pierre Louis Antoine Cordier (31 March 1777 – 30 March 1861) was a French geologist and mineralogist, and a founder of the French Geological Society. He was professor of geology at the Muséum national d'histoire naturelle in Paris from 1819 to 1861, and was responsible for the development of the geological gallery in the museum. ( Full article...) -
Image 6
Georgius Agricola ( /əˈɡrɪkələ/; born Georg Bauer; 24 March 1494 – 21 November 1555) was a German Humanist scholar, mineralogist and metallurgist. Born in the small town of Glauchau, in the Electorate of Saxony of the Holy Roman Empire, he was broadly educated, but took a particular interest in the mining and refining of metals. He was the first to drop the Arabic definite article al-, exclusively writing chymia and chymista in describing activity that we today would characterize as chemical or alchemical, giving chemistry its modern name. For his groundbreaking work De Natura Fossilium published in 1546, he is generally referred to as the Father of Mineralogy and the founder of geology as a scientific discipline.
He is well known for his pioneering work De re metallica libri XII, that was published in 1556, one year after his death. This 12-volume work is a comprehensive and systematic study, classification and methodical guide on all available factual and practical aspects, that are of concern for mining, the mining sciences and metallurgy, investigated and researched in its natural environment by means of direct observation. Unrivalled in its complexity and accuracy, it served as the standard reference work for two centuries. Agricola stated in the preface, that he will exclude "all those things which I have not myself seen, or have not read or heard of". He continued, "That which I have neither seen, nor carefully considered after reading or hearing of, I have not written about." ( Full article...) -
Image 7

William Babington
William Babington FRS FGS (21 May 1756 – 29 April 1833) was an Anglo-Irish physician and mineralogist. ( Full article...) -
Image 8

A page from Foshag's field book.
William Frederick Foshag (March 17, 1894 – May 21, 1956) was an American geologist and mineralogist.
He published nearly 100 papers and described 13 new minerals, including foshagite. ( Full article...) -
Image 9Pencil drawing of Miers by William Rothenstein, 1917
Sir Henry Alexander Miers, FRS (25 May 1858 – 10 December 1942) was a British mineralogist and crystallographer.
Born in Rio de Janeiro, Brazil, he was educated at Eton College and Trinity College, Oxford. He was elected a Fellow of the Royal Society in 1896. ( Full article...) -
Image 10
Linus Carl Pauling FRS ( /ˈpɔːlɪŋ/ PAW-ling; February 28, 1901 – August 19, 1994) was an American chemist, biochemist, chemical engineer, peace activist, author, and educator. He published more than 1,200 papers and books, of which about 850 dealt with scientific topics. New Scientist called him one of the 20 greatest scientists of all time. For his scientific work, Pauling was awarded the Nobel Prize in Chemistry in 1954. For his peace activism, he was awarded the Nobel Peace Prize in 1962. He is one of five people to have won more than one Nobel Prize (the others being Marie Curie, John Bardeen, Frederick Sanger, and Karl Barry Sharpless). Of these, he is the only person to have been awarded two unshared Nobel Prizes, and one of two people to be awarded Nobel Prizes in different fields, the other being Marie Curie.
Pauling was one of the founders of the fields of quantum chemistry and molecular biology. His contributions to the theory of the chemical bond include the concept of orbital hybridisation and the first accurate scale of electronegativities of the elements. Pauling also worked on the structures of biological molecules, and showed the importance of the alpha helix and beta sheet in protein secondary structure. Pauling's approach combined methods and results from X-ray crystallography, molecular model building, and quantum chemistry. His discoveries inspired the work of Rosalind Franklin, James Watson, Francis Crick, and Maurice Wilkins on the structure of DNA, which in turn made it possible for geneticists to crack the DNA code of all organisms. ( Full article...) -
Image 11

Ernst Erhard Schmid
Ernst Erhard Friedrich Wilhelm Schmid (22 May 1815 in Hildburghausen – 16 February 1885 in Jena) was a German paleontologist. He was the son of law professor Karl Ernst Schmid (1774–1852).
He studied natural sciences at the universities of Jena and Vienna, receiving his doctorate in 1839. In 1843 he became an associate professor at Jena, where with Matthias Jakob Schleiden, he founded a physiological institute. At the institute he dealt with subjects that included mineralogy, geology, chemistry and physics. In 1856 he was appointed a professor of natural sciences at the University of Jena. ( Full article...) -
Image 12Robert Minard Garrels (August 24, 1916 – March 8, 1988) was an American geochemist. Garrels applied experimental physical chemistry data and techniques to geology and geochemistry problems. The book Solutions, Minerals, and Equilibria co-authored in 1965 by Garrels and Charles L. Christ revolutionized aqueous geochemistry.
Garrels earned a bachelor's degree in geology from the University of Michigan in 1937. He went on to earn an M.S. degree from Northwestern University in 1939, his thesis work was on iron ores of Newfoundland in 1938. His Ph.D. was awarded in 1941 based on lab studies of complex formation between lead and chloride ions in aqueous solution. ( Full article...) -
Image 13

Franz Ernst Brückmann (27 September 1697 – 21 March 1753) was a German mineralogist born at Marienthal near Helmstedt. Having qualified as a physician in 1721, he practised at Braunschweig and afterwards at Wolfenbüttel (from 1728). In 1747 he was appointed medical assessor in Braunschweig.
His leisure time was given up to natural history, and especially to mineralogy and botany. He appears to have been the first to introduce the term "oolithus" to rocks that resemble in structure the roe of a fish; whence the terms " oolite" and "oolitic". He died at Wolfenbüttel. ( Full article...) -
Image 14Sjur Aasmundsen Sexe (14 August 1808 – 17 February 1888) was a Norwegian mineralogist and educator. ( Full article...)
-
Image 15Serve-Dieu Abailard "Armand" Lévy (14 November 1795 – 29 July 1841) was a French mathematician and mineralogist. He is remembered in the Haüy-Lévy notation for describing mineral crystal structures. ( Full article...)
-
Image 16Douglas Saxon Coombs CNZM (23 November 1924 – 23 December 2016) was a New Zealand mineralogist and petrologist. ( Full article...)
-
Image 17

Ours-Pierre-Armand Petit-Dufrénoy (5 September 1792 – 20 March 1857) was a French geologist and mineralogist. ( Full article...) -
Image 18Ian Stuart Edward Carmichael, FRS (29 March 1930 –26 August 2011) was a British-born American igneous petrologist and volcanologist who established extensive quantitative methods for research in the thermodynamics of magmas. ( Full article...)
-
Image 19

Charles-Louis de la Vallée Poussin
Charles-Louis-Joseph-Xavier de la Vallée Poussin (6 April 1827, Namur – 15 March 1903, Brussels) was a Belgian geologist and mineralogist. His son was the mathematician Charles Jean de la Vallée Poussin. ( Full article...) -
Image 20Helen Dick Megaw (1 June 1907 – 26 February 2002) was an Irish crystallographer who was a pioneer in X-ray crystallography. She made measurements of the cell dimensions of ice and established the Perovskite crystal structure. ( Full article...)
-
Image 21
Marcel Alexandre Bertrand (2 July 1847 – 13 February 1907) was a French geologist born in Paris. He was the son of mathematician Joseph Louis François Bertrand (1822–1900), and son-in-law to physicist Éleuthère Mascart (1837-1908).
He studied at the École Polytechnique, and beginning in 1869 he attended the Ecole des Mines de Paris. From 1877 he carried out geological mapping studies of Provence, the Jura Mountains and the Alps. In 1886, he became an instructor at the École Nationale Supérieure des Mines, and in 1896 became a member of the Académie des sciences. ( Full article...) -
Image 22

August Ferdinand von Veltheim (18 September 1741 Harbke – 2 October 1801. Braunschweig) was a German mineralogist and geologist, and came from the aristocratic family of Veltheim. He was known as August Ferdinand Graf von Veltheim from 1798 after acquiring the title of ' Graf'.
He attended the Kloster Berge school (Pädagogium) near Magdeburg between 1756 and 1758. He was introduced to the world of mining by Friedrich Anton von Heynitz, a mining official and later Minister of Industry under Frederick the Great. In 1760 he enrolled for a course on mineralogy at the University of Helmstedt. In 1763 he became financial advisor to the Duke of Braunschweig, and in this capacity made numerous trips to mines and saltworks of the region in the company of his father Friedrich August von Veltheim (1709-1775). In 1766 he was promoted to Inspector of Mines in the mineral-rich mining district of the Harz Mountains. After the death of his first son and first wife in 1779, Veltheim resigned his post and for some years withdrew from public life.[ circular reference] ( Full article...) -
Image 23Alexander Karl von Kraatz-Koschlau (24 June 1867 – 18 May 1900) was a German geologist.
Karl von Kraatz-Koschlau was born in Reichenbach near Stettin. He studied philosophy and sciences in Freiburg and Munich, where he obtained his doctorate with a dissertation on tartaric acid and its salts (1892). Afterwards he was assigned to the mineralogical institute in Munich. One of his scientific excursions during this time period involved geological research of the Serra de Monchique in the Algarve. ( Full article...) -
Image 24

Teachers of the Forestry Academy in Eberswalde (ca. 1868); Adolf Remelé, 3rd figure from the right (standing).
Adolf Karl Remelé (17 July 1839, Uerdingen – 16 November 1915, Eberswalde) was a German geologist and mineralogist.
He received his education at the University of Bonn, at the École des Mines in Paris and from the University of Berlin, receiving his doctorate in 1864 with the dissertation "De rubro uranico". In 1867 he qualified as a lecturer at Berlin, and during the following year, succeeded Lothar Meyer at the Forestry Academy in Eberswalde, where he taught classes in chemistry, geognosy and mineralogy. ( Full article...) -
Image 25
Alexandra Navrotsky (born 20 June 1943 in New York City) is a physical chemist in the field of nanogeoscience. She is an elected member of the United States National Academy of Sciences (NAS) and the American Philosophical Society (APS). She was a board member of the Earth Sciences and Resources division of the NAS from 1995 until 2000.
In 2005, she was awarded the Urey Medal, by the European Association of Geochemistry.
In 2006, she was awarded the Harry H. Hess Medal, by the American Geophysical Union.
She is currently the director of NEAT ORU (Nanomaterials in Environment, Agriculture, and Technology Organized Research Unit), a primary program in nanogeoscience. She is distinguished professor at University of California, Davis. ( Full article...)
Related portals
Get involved
For editor resources and to collaborate with other editors on improving Wikipedia's Minerals-related articles, see WikiProject Rocks and minerals.
General images
-
Image 1 Asbestiform tremolite, part of the amphibole group in the inosilicate subclass (from Mineral)
-
Image 2Black andradite, an end-member of the orthosilicate garnet group. (from Mineral)
-
Image 3 Hübnerite, the manganese-rich end-member of the wolframite series, with minor quartz in the background (from Mineral)
-
Image 5Muscovite, a mineral species in the mica group, within the phyllosilicate subclass (from Mineral)
-
Image 7Pink cubic halite (NaCl; halide class) crystals on a nahcolite matrix (NaHCO3; a carbonate, and mineral form of sodium bicarbonate, used as baking soda). (from Mineral)
-
Image 8Diamond is the hardest natural material, and has a Mohs hardness of 10. (from Mineral)
-
Image 10Gypsum desert rose (from Mineral)
-
Image 11An example of elbaite, a species of tourmaline, with distinctive colour banding. (from Mineral)
-
Image 12Mohs hardness kit, containing one specimen of each mineral on the ten-point hardness scale (from Mohs scale)
-
Image 16Perfect basal cleavage as seen in biotite (black), and good cleavage seen in the matrix (pink orthoclase). (from Mineral)
-
Image 17 Schist is a metamorphic rock characterized by an abundance of platy minerals. In this example, the rock has prominent sillimanite porphyroblasts as large as 3 cm (1.2 in). (from Mineral)
-
Image 19Sphalerite crystal partially encased in calcite from the Devonian Milwaukee Formation of Wisconsin (from Mineral)
-
Image 20Native gold. Rare specimen of stout crystals growing off of a central stalk, size 3.7 x 1.1 x 0.4 cm, from Venezuela. (from Mineral)
-
Image 21Mohs Scale versus Absolute Hardness (from Mineral)
-
Image 23Red cinnabar (HgS), a mercury ore, on dolomite. (from Mineral)
-
Image 24Epidote often has a distinctive pistachio-green colour. (from Mineral)
-
Image 25When minerals react, the products will sometimes assume the shape of the reagent; the product mineral is termed a pseudomorph of (or after) the reagent. Illustrated here is a pseudomorph of kaolinite after orthoclase. Here, the pseudomorph preserved the Carlsbad twinning common in orthoclase. (from Mineral)
Did you know ...?

- ... that leonite (pictured) has been found on Mars?
- ...that crystals of Paulingite, a rare zeolite mineral found in vesicles in the basaltic rocks from the Columbia River, form a perfect clear rhombic dodecahedron?
- ... that abernathyite is both fluorescent and radioactive and is named for the mine operator who discovered it?
Subcategories
Topics
| Overview | ||
|---|---|---|
| Common minerals | ||
Ore minerals, mineral mixtures and
ore deposits | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ores |
| ||||||||
| Deposit types | |||||||||
| Borates | |||||
|---|---|---|---|---|---|
| Carbonates | |||||
| Oxides |
| ||||
| Phosphates | |||||
| Silicates | |||||
| Sulfides | |||||
| Other |
| ||||
| Crystalline | |||||||
|---|---|---|---|---|---|---|---|
| Cryptocrystalline | |||||||
| Amorphous | |||||||
| Miscellaneous | |||||||
| Notable varieties |
| ||||||
| Oxide minerals |
| ||||
|---|---|---|---|---|---|
| Silicate minerals | |||||
| Other | |||||
Gemmological classifications by E. Ya. Kievlenko (1980), updated | |||||||||
| Jewelry stones |
| ||||||||
| Jewelry-Industrial stones |
| ||||||||
| Industrial stones |
| ||||||||
Mineral identification | |
|---|---|
| "Special cases" ("native elements and organic minerals") |
|
|---|---|
| "Sulfides and oxides" |
|
| "Evaporites and similars" |
|
| "Mineral structures with tetrahedral units" (sulfate anion, phosphate anion, silicon, etc.) |
|
Associated Wikimedia
The following Wikimedia Foundation sister projects provide more on this subject:
-
Commons
Free media repository -
Wikibooks
Free textbooks and manuals -
Wikidata
Free knowledge base -
Wikinews
Free-content news -
Wikiquote
Collection of quotations -
Wikisource
Free-content library -
Wikiversity
Free learning tools -
Wiktionary
Dictionary and thesaurus
References
- Pages using Template:Post-nominals with missing parameters
- Manually maintained portal pages from May 2019
- All manually maintained portal pages
- Portals with triaged subpages from May 2019
- All portals with triaged subpages
- Portals with named maintainer
- Automated article-slideshow portals with 31–40 articles in article list
- Automated article-slideshow portals with 201–500 articles in article list
- Portals needing placement of incoming links