| |||||||||||||||||||||||||||||||||||||
Neptunium (93Np) is usually considered an
artificial element, although trace quantities are found in nature, so a
standard atomic weight cannot be given. Like all trace or artificial elements, it has no
stable isotopes. The first
isotope to be synthesized and identified was 239Np in 1940, produced by bombarding
238
U
with
neutrons to produce 239
U
, which then underwent
beta decay to 239
Np
.
Trace quantities are found in nature from neutron capture reactions by uranium atoms, a fact not discovered until 1951. [2]
Twenty-five neptunium
radioisotopes have been characterized, with the most stable being 237
Np
with a
half-life of 2.14 million years, 236
Np
with a half-life of 154,000 years, and 235
Np
with a half-life of 396.1 days. All of the remaining
radioactive isotopes have half-lives that are less than 4.5 days, and the majority of these have half-lives that are less than 50 minutes. This element also has five
meta states, with the most stable being 236m
Np
(t1/2 22.5 hours).
The isotopes of neptunium range from 219
Np
to 244
Np
, though the intermediate isotope 221
Np
has not yet been observed. The primary
decay mode before the most stable isotope, 237
Np
, is
electron capture (with a good deal of
alpha emission), and the primary mode after is
beta emission. The primary
decay products before 237
Np
are
isotopes of uranium and
protactinium, and the primary products after are
isotopes of plutonium. Neptunium is the heaviest element for which the location of the
proton drip line is known; the lightest bound isotope is 220Np.
[3]
List of isotopes
| Nuclide [n 1] |
Z | N |
Isotopic mass (
Da)
[4] [n 2] [n 3] |
Half-life |
Decay mode [n 4] |
Daughter isotope [n 5] |
Spin and parity [n 6] [n 7] |
Isotopic abundance | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Excitation energy [n 7] | |||||||||||||||||||
| 219 Np [5] [n 8] |
93 | 126 | 219.03162(9) | 0.15+0.72 −0.07 ms |
α | 215Pa | (9/2−) | ||||||||||||
| 220 Np [3] |
93 | 127 | 220.03254(21)# | 25+14 −7 μs |
α | 216Pa | 1−# | ||||||||||||
| 222 Np [6] |
93 | 129 | 380+260 −110 ns |
α | 218Pa | 1-# | |||||||||||||
| 223 Np [7] |
93 | 130 | 223.03285(21)# | 2.15+100 −52 μs |
α | 219Pa | 9/2− | ||||||||||||
| 224 Np [8] |
93 | 131 | 224.03422(21)# | 38+26 −11 μs |
α (83%) | 220m1Pa | 1−# | ||||||||||||
| α (17%) | 220m2Pa | ||||||||||||||||||
| 225 Np |
93 | 132 | 225.03391(8) | 6(5) ms | α | 221Pa | 9/2−# | ||||||||||||
| 226 Np |
93 | 133 | 226.03515(10)# | 35(10) ms | α | 222Pa | |||||||||||||
| 227 Np |
93 | 134 | 227.03496(8) | 510(60) ms | α (99.95%) | 223Pa | 5/2−# | ||||||||||||
| β+ (.05%) | 227U | ||||||||||||||||||
| 228 Np |
93 | 135 | 228.03618(21)# | 61.4(14) s | β+ (59%) | 228U | |||||||||||||
| α (41%) | 224Pa | ||||||||||||||||||
| β+, SF (.012%) | (various) | ||||||||||||||||||
| 229 Np |
93 | 136 | 229.03626(9) | 4.0(2) min | α (51%) | 225Pa | 5/2+# | ||||||||||||
| β+ (49%) | 229U | ||||||||||||||||||
| 230 Np |
93 | 137 | 230.03783(6) | 4.6(3) min | β+ (97%) | 230U | |||||||||||||
| α (3%) | 226Pa | ||||||||||||||||||
| 231 Np |
93 | 138 | 231.03825(5) | 48.8(2) min | β+ (98%) | 231U | (5/2)(+#) | ||||||||||||
| α (2%) | 227Pa | ||||||||||||||||||
| 232 Np |
93 | 139 | 232.04011(11)# | 14.7(3) min | β+ (99.99%) | 232U | (4+) | ||||||||||||
| α (.003%) | 228Pa | ||||||||||||||||||
| 233 Np |
93 | 140 | 233.04074(5) | 36.2(1) min | β+ (99.99%) | 233U | (5/2+) | ||||||||||||
| α (.001%) | 229Pa | ||||||||||||||||||
| 234 Np |
93 | 141 | 234.042895(9) | 4.4(1) d | β+ | 234U | (0+) | ||||||||||||
| 234m Np |
~9 min [9] | IT | 234Np | 5+ | |||||||||||||||
| EC | 234U | ||||||||||||||||||
| 235 Np |
93 | 142 | 235.0440633(21) | 396.1(12) d | EC | 235U | 5/2+ | ||||||||||||
| α (.0026%) | 231Pa | ||||||||||||||||||
| 236 Np [n 9] |
93 | 143 | 236.04657(5) | 1.54(6)×105 y | EC (87.3%) | 236U | (6−) | ||||||||||||
| β− (12.5%) | 236Pu | ||||||||||||||||||
| α (.16%) | 232Pa | ||||||||||||||||||
| 236m Np |
60(50) keV | 22.5(4) h | EC (52%) | 236U | 1 | ||||||||||||||
| β− (48%) | 236Pu | ||||||||||||||||||
| 237 Np [n 10] |
93 | 144 | 237.0481734(20) | 2.144(7)×106 y | α | 233Pa | 5/2+ | Trace [n 11] | |||||||||||
| SF (2×10−10%) | (various) | ||||||||||||||||||
| CD (4×10−12%) | 207Tl 30Mg | ||||||||||||||||||
| 238 Np |
93 | 145 | 238.0509464(20) | 2.117(2) d | β− | 238Pu | 2+ | ||||||||||||
| 238m Np |
2300(200)# keV | 112(39) ns | |||||||||||||||||
| 239 Np |
93 | 146 | 239.0529390(22) | 2.356(3) d | β− | 239Pu | 5/2+ | Trace [n 11] | |||||||||||
| 240 Np |
93 | 147 | 240.056162(16) | 61.9(2) min | β− | 240Pu | (5+) | Trace [n 12] | |||||||||||
| 240m Np |
20(15) keV | 7.22(2) min | β− (99.89%) | 240Pu | 1(+) | ||||||||||||||
| IT (.11%) | 240Np | ||||||||||||||||||
| 241 Np |
93 | 148 | 241.05825(8) | 13.9(2) min | β− | 241Pu | (5/2+) | ||||||||||||
| 242 Np |
93 | 149 | 242.06164(21) | 2.2(2) min | β− | 242Pu | (1+) | ||||||||||||
| 242m Np |
0(50)# keV | 5.5(1) min | 6+# | ||||||||||||||||
| 243 Np |
93 | 150 | 243.06428(3)# | 1.85(15) min | β− | 243Pu | (5/2−) | ||||||||||||
| 244 Np |
93 | 151 | 244.06785(32)# | 2.29(16) min | β− | 244Pu | (7−) | ||||||||||||
| This table header & footer: | |||||||||||||||||||
- ^ mNp – Excited nuclear isomer.
- ^ ( ) – Uncertainty (1σ) is given in concise form in parentheses after the corresponding last digits.
- ^ # – Atomic mass marked #: value and uncertainty derived not from purely experimental data, but at least partly from trends from the Mass Surface (TMS).
-
^
Modes of decay:
CD: Cluster decay EC: Electron capture IT: Isomeric transition SF: Spontaneous fission - ^ Bold italics symbol as daughter – Daughter product is nearly stable.
- ^ ( ) spin value – Indicates spin with weak assignment arguments.
- ^ a b # – Values marked # are not purely derived from experimental data, but at least partly from trends of neighboring nuclides (TNN).
- ^ Heaviest known nucleus, as of 2019 [update], that is beyond the proton drip line.
- ^ Fissile nuclide
- ^ Most common nuclide
- ^ a b Produced by neutron capture in uranium ore
- ^ Intermediate decay product of 244Pu
Actinides vs fission products
| Actinides [10] by decay chain |
Half-life range ( a) |
Fission products of 235U by yield [11] | ||||||
|---|---|---|---|---|---|---|---|---|
| 4n | 4n + 1 | 4n + 2 | 4n + 3 | 4.5–7% | 0.04–1.25% | <0.001% | ||
| 228Ra№ | 4–6 a | 155Euþ | ||||||
| 244Cmƒ | 241Puƒ | 250Cf | 227Ac№ | 10–29 a | 90Sr | 85Kr | 113mCdþ | |
| 232Uƒ | 238Puƒ | 243Cmƒ | 29–97 a | 137Cs | 151Smþ | 121mSn | ||
| 248Bk [12] | 249Cfƒ | 242mAmƒ | 141–351 a |
No fission products have a half-life | ||||
| 241Amƒ | 251Cfƒ [13] | 430–900 a | ||||||
| 226Ra№ | 247Bk | 1.3–1.6 ka | ||||||
| 240Pu | 229Th | 246Cmƒ | 243Amƒ | 4.7–7.4 ka | ||||
| 245Cmƒ | 250Cm | 8.3–8.5 ka | ||||||
| 239Puƒ | 24.1 ka | |||||||
| 230Th№ | 231Pa№ | 32–76 ka | ||||||
| 236Npƒ | 233Uƒ | 234U№ | 150–250 ka | 99Tc₡ | 126Sn | |||
| 248Cm | 242Pu | 327–375 ka | 79Se₡ | |||||
| 1.53 Ma | 93Zr | |||||||
| 237Npƒ | 2.1–6.5 Ma | 135Cs₡ | 107Pd | |||||
| 236U | 247Cmƒ | 15–24 Ma | 129I₡ | |||||
| 244Pu | 80 Ma |
... nor beyond 15.7 Ma [14] | ||||||
| 232Th№ | 238U№ | 235Uƒ№ | 0.7–14.1 Ga | |||||
| ||||||||
Notable isotopes
Neptunium-235
Neptunium-235 has 142 neutrons and a half-life of 396.1 days. This isotope decays by:
- Alpha emission: the decay energy is 5.2 MeV and the decay product is protactinium-231.
- Electron capture: the decay energy is 0.125 MeV and the decay product is uranium-235
This isotope of neptunium has a weight of 235.044 063 3 u.
Neptunium-236
Neptunium-236 has 143 neutrons and a half-life of 154,000 years. It can decay by the following methods:
- Electron capture: the decay energy is 0.93 MeV and the decay product is uranium-236. This usually decays (with a half-life of 23 million years) to thorium-232.
- Beta emission: the decay energy is 0.48 MeV and the decay product is plutonium-236. This usually decays (half-life 2.8 years) to uranium-232, which usually decays (half-life 69 years) to thorium-228, which decays in a few years to lead-208.
- Alpha emission: the decay energy is 5.007 MeV and the decay product is protactinium-232. This decays with a half-life of 1.3 days to uranium-232.
This particular isotope of neptunium has a mass of 236.04657 u. It is a fissile material; it has an estimated critical mass of 6.79 kg (15.0 lb), [15] though precise experimental data is not available. [16]
236
Np
is produced in small quantities via the (n,2n) and (γ,n) capture reactions of 237
Np
,
[17] however, it is nearly impossible to separate in any significant quantities from its parent 237
Np
.
[18] It is for this reason that despite its low critical mass and high neutron cross section, it has not been researched extensively as a nuclear fuel in weapons or reactors.
[16] Nevertheless, 236
Np
has been considered for use in
mass spectrometry and as a
radioactive tracer, because it decays predominantly by beta emission with a long half-life.
[19] Several alternative production routes for this isotope have been investigated, namely those that reduce isotopic separation from 237
Np
or the
isomer 236m
Np
. The most favorable reactions to accumulate 236
Np
were shown to be
proton and
deuteron irradiation of
uranium-238.
[19]
Neptunium-237
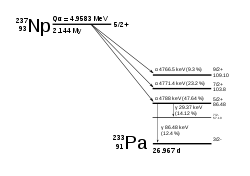
237
Np
decays via the
neptunium series, which terminates with
thallium-205, which is stable, unlike most other
actinides, which decay to stable
isotopes of lead.
In 2002, 237
Np
was shown to be capable of sustaining a chain reaction with
fast neutrons, as in a
nuclear weapon, with a critical mass of around 60 kg.
[20] However, it has a low probability of fission on bombardment with
thermal neutrons, which makes it unsuitable as a fuel for light water nuclear power plants (as opposed to
fast reactor or
accelerator-driven systems, for example).
Inventory in spent nuclear fuel
237
Np
is the only neptunium isotope produced in significant quantity in the
nuclear fuel cycle, both by successive
neutron capture by
uranium-235 (which fissions most but not all of the time) and
uranium-236, or (n,2n) reactions where a
fast neutron occasionally knocks a neutron loose from
uranium-238 or
isotopes of plutonium. Over the long term, 237
Np
also forms in
spent nuclear fuel as the decay product of
americium-241.
237
Np
is considered to be one of the most mobile
radionuclides at the site of the
Yucca Mountain nuclear waste repository (
Nevada) where
oxidizing conditions prevail in the
unsaturated zone of the
volcanic tuff above the
water table.
Raw material for 238
Pu production
When exposed to neutron bombardment 237
Np
can capture a neutron, undergo beta decay, and become
238
Pu
, this product being useful as a thermal energy source in a
radioisotope thermoelectric generator (RTG or RITEG) for the production of electricity and heat. The first type of thermoelectric generator SNAP (
Systems for Nuclear Auxiliary Power) was developed and used by
NASA in the 1960's and during the
Apollo missions to power the instruments left on the Moon surface by the astronauts. Thermoelectric generators were also embarked on board of deep
space probes such as for the
Pioneer 10 and 11 missions, the
Voyager program, the
Cassini–Huygens mission, and
New Horizons. They also deliver electrical and thermal power to the
Mars Science Laboratory (Curiosity rover) and
Mars 2020 mission (
Perseverance rover) both exploring the cold surface of
Mars. Curiosity and Perseverance rovers are both equipped with the last version of
multi-mission RTG, a more efficient and standardized system dubbed
MMRTG.
These applications are economically practical where photovoltaic power sources are weak or inconsistent due to probes being too far from the sun or rovers facing climate events that may obstruct sunlight for long periods (like Martian dust storms). Space probes and rovers also make use of the heat output of the generator to keep their instruments and internals warm. [21]
Shortage of 237
Np stockpiles
The long
half-life (T½ ~ 88 years) of 238
Pu
and the absence of
γ-radiation that could interfere with the operation of on-board electronic components, or irradiate people, makes it the radionuclide of choice for electric thermogenerators.
237
Np
is therefore a key radionuclide for the production of 238
Pu
, which is essential for deep space probes requiring a reliable and long-lasting source of energy without maintenance.
Stockpiles of
238
Pu
built up in the United States since the
Manhattan Project, thanks to the
Hanford nuclear complex (operating in
Washington State from 1943 to 1977) and the development of
atomic weapons, are now almost exhausted. The extraction and purification of sufficient new quantities of 237
Np
from
irradiated nuclear fuels is therefore necessary for the resumption of 238
Pu
production in order to replenish the stocks needed for space exploration by robotic probes.
Neptunium-239
Neptunium-239 has 146 neutrons and a half-life of 2.356 days. It is produced via β− decay of the short-lived uranium-239, and undergoes another β− decay to plutonium-239. This is the primary route for making plutonium, as 239U can be made by neutron capture in uranium-238. [22]
Uranium-237 and neptunium-239 are regarded as the leading hazardous radioisotopes in the first hour-to-week period following nuclear fallout from a nuclear detonation, with 239Np dominating "the spectrum for several days." [23] [24]
References
- ^ Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties" (PDF). Chinese Physics C. 45 (3): 030001. doi: 10.1088/1674-1137/abddae.
- ^ Peppard, D. F.; Mason, G. W.; Gray, P. R.; Mech, J. F. (1952). "Occurrence of the (4n + 1) series in nature" (PDF). Journal of the American Chemical Society. 74 (23): 6081–6084. doi: 10.1021/ja01143a074.
- ^ a b Zhang, Z. Y.; Gan, Z. G.; Yang, H. B.; et al. (2019). "New isotope 220Np: Probing the robustness of the N = 126 shell closure in neptunium". Physical Review Letters. 122 (19): 192503. Bibcode: 2019PhRvL.122s2503Z. doi: 10.1103/PhysRevLett.122.192503. PMID 31144958. S2CID 169038981.
- ^ Wang, M.; Audi, G.; Kondev, F. G.; Huang, W. J.; Naimi, S.; Xu, X. (2017). "The AME2016 atomic mass evaluation (II). Tables, graphs, and references" (PDF). Chinese Physics C. 41 (3): 030003-1–030003-442. doi: 10.1088/1674-1137/41/3/030003.
- ^ Yang, H; Ma, L; Zhang, Z; Yang, C; Gan, Z; Zhang, M; et al. (2018). "Alpha decay properties of the semi-magic nucleus 219Np". Physics Letters B. 777: 212–216. Bibcode: 2018PhLB..777..212Y. doi: 10.1016/j.physletb.2017.12.017.
- ^ Ma, L.; Zhang, Z. Y.; Gan, Z. G.; et al. (2020). "Short-Lived α-emitting isotope 222Np and the Stability of the N=126 Magic Shell". Physical Review Letters. 125 (3): 032502. Bibcode: 2020PhRvL.125c2502M. doi: 10.1103/PhysRevLett.125.032502. PMID 32745401. S2CID 220965400.
- ^ Sun, M. D.; et al. (2017). "New short-lived isotope 223Np and the absence of the Z = 92 subshell closure near N = 126". Physics Letters B. 771: 303–308. Bibcode: 2017PhLB..771..303S. doi: 10.1016/j.physletb.2017.03.074.
- ^ Huang, T. H.; et al. (2018). "Identification of the new isotope 224Np" (pdf). Physical Review C. 98 (4): 044302. Bibcode: 2018PhRvC..98d4302H. doi: 10.1103/PhysRevC.98.044302. S2CID 125251822.
- ^ Asai, M.; Suekawa, Y.; Higashi, M.; et al. Discovery of 234 Np isomer and its decay properties (PDF) (Report) (in Japanese).
- ^ Plus radium (element 88). While actually a sub-actinide, it immediately precedes actinium (89) and follows a three-element gap of instability after polonium (84) where no nuclides have half-lives of at least four years (the longest-lived nuclide in the gap is radon-222 with a half life of less than four days). Radium's longest lived isotope, at 1,600 years, thus merits the element's inclusion here.
- ^ Specifically from thermal neutron fission of uranium-235, e.g. in a typical nuclear reactor.
-
^ Milsted, J.; Friedman, A. M.; Stevens, C. M. (1965). "The alpha half-life of berkelium-247; a new long-lived isomer of berkelium-248". Nuclear Physics. 71 (2): 299.
Bibcode:
1965NucPh..71..299M.
doi:
10.1016/0029-5582(65)90719-4.
"The isotopic analyses disclosed a species of mass 248 in constant abundance in three samples analysed over a period of about 10 months. This was ascribed to an isomer of Bk248 with a half-life greater than 9 [years]. No growth of Cf248 was detected, and a lower limit for the β− half-life can be set at about 104 [years]. No alpha activity attributable to the new isomer has been detected; the alpha half-life is probably greater than 300 [years]." - ^ This is the heaviest nuclide with a half-life of at least four years before the " sea of instability".
- ^ Excluding those " classically stable" nuclides with half-lives significantly in excess of 232Th; e.g., while 113mCd has a half-life of only fourteen years, that of 113Cd is eight quadrillion years.
- ^ Final Report, Evaluation of nuclear criticality safety data and limits for actinides in transport (PDF) (Report). Republic of France, Institut de Radioprotection et de Sûreté Nucléaire, Département de Prévention et d'étude des Accidents. Archived from the original (PDF) on 2011-05-19.
- ^ a b Reed, B. C. (2017). "An examination of the potential fission-bomb weaponizability of nuclides other than 235U and 239Pu". American Journal of Physics. 85: 38–44. doi: 10.1119/1.4966630.
- ^ Analysis of the Reuse of Uranium Recovered from the Reprocessing of Commercial LWR Spent Fuel, United States Department of Energy, Oak Ridge National Laboratory.
- ^ **Jukka Lehto; Xiaolin Hou (2011). "15.15: Neptunium". Chemistry and Analysis of Radionuclides (1st ed.). John Wiley & Sons. 231. ISBN 978-3527633029.
- ^ a b Jerome, S.M.; Ivanov, P.; Larijani, C.; Parker, D.J.; Regan, P.H. (2014). "The production of Neptunium-236g". Journal of Environmental Radioactivity. 138: 315–322. doi: 10.1016/j.jenvrad.2014.02.029. PMID 24731718.
- ^ P. Weiss (26 October 2002). "Neptunium Nukes? Little-studied metal goes critical". Science News. 162 (17): 259. doi: 10.2307/4014034. JSTOR 4014034. Archived from the original on 15 December 2012. Retrieved 7 November 2013.
- ^ Witze, Alexandra (2014-11-27). "Nuclear power: Desperately seeking plutonium". Nature. 515 (7528): 484–486. Bibcode: 2014Natur.515..484W. doi: 10.1038/515484a. PMID 25428482.
- ^ "Periodic Table Of Elements: LANL - Neptunium". Los Alamos National Laboratory. Retrieved 2013-10-13.
- ^ [Film Badge Dosimetry in Atmospheric Nuclear Tests, By Committee on Film Badge Dosimetry in Atmospheric Nuclear Tests, Commission on Engineering and Technical Systems, Division on Engineering and Physical Sciences, National Research Council. pg24-35]
- ^ Bounding Analysis of Effects of Fractionation of Radionuclides in Fallout on Estimation of Doses to Atomic Veterans DTRA-TR-07-5. 2007
- Isotope masses from:
- Audi, Georges; Bersillon, Olivier; Blachot, Jean; Wapstra, Aaldert Hendrik (2003), "The NUBASE evaluation of nuclear and decay properties", Nuclear Physics A, 729: 3–128, Bibcode: 2003NuPhA.729....3A, doi: 10.1016/j.nuclphysa.2003.11.001
- Half-life, spin, and isomer data selected from the following sources.
- Audi, Georges; Bersillon, Olivier; Blachot, Jean; Wapstra, Aaldert Hendrik (2003), "The NUBASE evaluation of nuclear and decay properties", Nuclear Physics A, 729: 3–128, Bibcode: 2003NuPhA.729....3A, doi: 10.1016/j.nuclphysa.2003.11.001
- National Nuclear Data Center. "NuDat 2.x database". Brookhaven National Laboratory.
- Holden, Norman E. (2004). "11. Table of the Isotopes". In Lide, David R. (ed.). CRC Handbook of Chemistry and Physics (85th ed.). Boca Raton, Florida: CRC Press. ISBN 978-0-8493-0485-9.