| Corticosteroid | |
|---|---|
| Drug class | |

Cortisol (
hydrocortisone), a corticosteroid with both
glucocorticoid and
mineralocorticoid activity and effects. | |
| Class identifiers | |
| Synonyms | Corticoid |
| Use | Various |
| ATC code | H02 |
| Biological target | Glucocorticoid receptor, Mineralocorticoid receptor |
| Chemical class | Steroids |
| Legal status | |
| In Wikidata | |
Corticosteroids are a class of steroid hormones that are produced in the adrenal cortex of vertebrates, as well as the synthetic analogues of these hormones. Two main classes of corticosteroids, glucocorticoids and mineralocorticoids, are involved in a wide range of physiological processes, including stress response, immune response, and regulation of inflammation, carbohydrate metabolism, protein catabolism, blood electrolyte levels, and behavior. [1]
Some common naturally occurring steroid hormones are
cortisol (C
21H
30O
5),
corticosterone (C
21H
30O
4),
cortisone (C
21H
28O
5) and
aldosterone (C
21H
28O
5) (cortisone and
aldosterone are
isomers). The main corticosteroids produced by the adrenal cortex are cortisol and aldosterone.
[1]
Classes
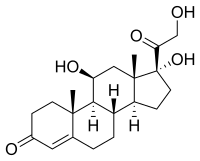


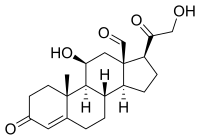
- Glucocorticoids such as cortisol affect carbohydrate, fat, and protein metabolism, and have anti-inflammatory, immunosuppressive, anti-proliferative, and vasoconstrictive effects. [2] Anti-inflammatory effects are mediated by blocking the action of inflammatory mediators ( transrepression) and inducing anti-inflammatory mediators ( transactivation). [2] Immunosuppressive effects are mediated by suppressing delayed hypersensitivity reactions by direct action on T-lymphocytes. [2] Anti-proliferative effects are mediated by inhibition of DNA synthesis and epidermal cell turnover. [2] Vasoconstrictive effects are mediated by inhibiting the action of inflammatory mediators such as histamine. [2]
- Mineralocorticoids such as aldosterone are primarily involved in the regulation of electrolyte and water balance by modulating ion transport in the epithelial cells of the renal tubules of the kidney. [2]
Medical uses
Synthetic pharmaceutical drugs with corticosteroid-like effects are used in a variety of conditions, ranging from hematological neoplasms [3] to brain tumors or skin diseases. Dexamethasone and its derivatives are almost pure glucocorticoids, while prednisone and its derivatives have some mineralocorticoid action in addition to the glucocorticoid effect. Fludrocortisone (Florinef) is a synthetic mineralocorticoid. Hydrocortisone (cortisol) is typically used for replacement therapy, e.g. for adrenal insufficiency and congenital adrenal hyperplasia.
Medical conditions treated with systemic corticosteroids: [2] [4]
-
Allergy and
respirology medicine
- Asthma (severe exacerbations)
- Chronic obstructive pulmonary disease (COPD)
- Allergic rhinitis
- Atopic dermatitis
- Hives
- Angioedema
- Anaphylaxis
- Food allergies
- Drug allergies
- Nasal polyps
- Hypersensitivity pneumonitis
- Sarcoidosis
- Eosinophilic pneumonia
- Some other types of pneumonia (in addition to the traditional antibiotic treatment protocols)
- Interstitial lung disease
- Dermatology
- Endocrinology (usually at physiologic doses)
- Gastroenterology
- Hematology
- Rheumatology/ Immunology
- Ophthalmology
- Other conditions
Topical formulations are also available for the skin, eyes ( uveitis), lungs ( asthma), nose ( rhinitis), and bowels. Corticosteroids are also used supportively to prevent nausea, often in combination with 5-HT3 antagonists (e.g., ondansetron).
Typical undesired effects of glucocorticoids present quite uniformly as drug-induced Cushing's syndrome. Typical mineralocorticoid side-effects are hypertension (abnormally high blood pressure), steroid induced diabetes mellitus, psychosis, poor sleep, hypokalemia (low potassium levels in the blood), hypernatremia (high sodium levels in the blood) without causing peripheral edema, metabolic alkalosis and connective tissue weakness. [5] Wound healing or ulcer formation may be inhibited by the immunosuppressive effects.
A variety of steroid medications, from anti-allergy nasal sprays ( Nasonex, Flonase) to topical skin creams, to eye drops ( Tobradex), to prednisone have been implicated in the development of central serous retinopathy (CSR). [6] [7]
Corticosteroids have been widely used in treating people with traumatic brain injury. [8] A systematic review identified 20 randomised controlled trials and included 12,303 participants, then compared patients who received corticosteroids with patients who received no treatment. The authors recommended people with traumatic head injury should not be routinely treated with corticosteroids. [9]
Pharmacology
Corticosteroids act as agonists of the glucocorticoid receptor and/or the mineralocorticoid receptor.
In addition to their corticosteroid activity, some corticosteroids may have some progestogenic activity and may produce sex-related side effects. [10] [11] [12] [13]
Pharmacogenetics
Asthma
Patients' response to inhaled corticosteroids has some basis in genetic variations. Two genes of interest are CHRH1 ( corticotropin-releasing hormone receptor 1) and TBX21 ( transcription factor T-bet). Both genes display some degree of polymorphic variation in humans, which may explain how some patients respond better to inhaled corticosteroid therapy than others. [14] [15] However, not all asthma patients respond to corticosteroids and large sub groups of asthma patients are corticosteroid resistant. [16]
A study funded by the Patient-Centered Outcomes Research Institute of children and teens with mild persistent asthma found that using the control inhaler as needed worked the same as daily use in improving asthma control, number of asthma flares, how well the lungs work, and quality of life. Children and teens using the inhaler as needed used about one-fourth the amount of corticosteroid medicine as children and teens using it daily. [17] [18]
Adverse effects
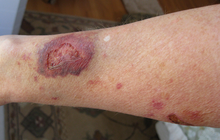
Use of corticosteroids has numerous side-effects, some of which may be severe:
- Severe amoebic colitis: Fulminant amoebic colitis is associated with high case fatality and can occur in patients infected with the parasite Entamoeba histolytica after exposure to corticosteroid medications. [19]
- Neuropsychiatric: steroid psychosis, [20] and anxiety, [21] depression. Therapeutic doses may cause a feeling of artificial well-being ("steroid euphoria"). [22] The neuropsychiatric effects are partly mediated by sensitization of the body to the actions of adrenaline. Therapeutically, the bulk of corticosteroid dose is given in the morning to mimic the body's diurnal rhythm; if given at night, the feeling of being energized will interfere with sleep. An extensive review is provided by Flores and Gumina. [23]
- Cardiovascular: Corticosteroids can cause sodium retention through a direct action on the kidney, in a manner analogous to the mineralocorticoid aldosterone. This can result in fluid retention and hypertension.
- Metabolic: Corticosteroids cause a movement of body fat to the face and torso, resulting in " moon face", "buffalo hump", and "pot belly" or "beer belly", and cause movement of body fat away from the limbs. This has been termed corticosteroid-induced lipodystrophy. Due to the diversion of amino-acids to glucose, they are considered anti-anabolic, and long term therapy can cause muscle wasting (muscle atrophy). [24] Besides muscle atrophy, steroid myopathy includes muscle pains (myalgias), muscle weakness (typically of the proximal muscles), serum creatine kinase normal, EMG myopathic, and some have type II (fast-twitch/glycolytic) fibre atrophy. [25]
- Endocrine: By increasing the production of glucose from amino-acid breakdown and opposing the action of insulin, corticosteroids can cause hyperglycemia, [26] insulin resistance and diabetes mellitus. [27]
- Skeletal: Steroid-induced osteoporosis may be a side-effect of long-term corticosteroid use. [28] [29] [30] Use of inhaled corticosteroids among children with asthma may result in decreased height. [31]
- Gastro-intestinal: While cases of colitis have been reported, corticosteroids are often prescribed when the colitis, although due to suppression of the immune response to pathogens, should be considered only after ruling out infection or microbe/fungal overgrowth in the gastrointestinal tract. While the evidence for corticosteroids causing peptic ulceration is relatively poor except for high doses taken for over a month, [32] the majority of doctors as of 2010 [update] still believe this is the case, and would consider protective prophylactic measures. [33]
- Eyes: chronic use may predispose to cataract and glaucoma. Clinical and experimental evidence indicates that corticosteroids can cause permanent eye damage by inducing central serous retinopathy (CSR, also known as central serous chorioretinopathy, CSC). [34] This should be borne in mind when treating patients with optic neuritis. There is experimental and clinical evidence that, at least in optic neuritis speed of treatment initiation is important. [35]
- Vulnerability to infection: By suppressing immune reactions (which is one of the main reasons for their use in allergies), steroids may cause infections to flare up, notably candidiasis. [36]
- Pregnancy: Corticosteroids have a low but significant teratogenic effect, causing a few birth defects per 1,000 pregnant women treated. Corticosteroids are therefore contraindicated in pregnancy. [37]
- Habituation: Topical steroid addiction (TSA) or red burning skin has been reported in long-term users of topical steroids (users who applied topical steroids to their skin over a period of weeks, months, or years). [38] [39] TSA is characterised by uncontrollable, spreading dermatitis and worsening skin inflammation which requires a stronger topical steroid to get the same result as the first prescription. When topical steroid medication is lost, the skin experiences redness, burning, itching, hot skin, swelling, and/or oozing for a length of time. This is also called 'red skin syndrome' or 'topical steroid withdrawal' (TSW). After the withdrawal period is over the atopic dermatitis can cease or is less severe than it was before. [40]
- In children the short term use of steroids by mouth increases the risk of vomiting, behavioral changes, and sleeping problems. [41]
Biosynthesis

The corticosteroids are synthesized from cholesterol within the adrenal cortex. [1] Most steroidogenic reactions are catalysed by enzymes of the cytochrome P450 family. They are located within the mitochondria and require adrenodoxin as a cofactor (except 21-hydroxylase and 17α-hydroxylase).
Aldosterone and corticosterone share the first part of their biosynthetic pathway. The last part is mediated either by the aldosterone synthase (for aldosterone) or by the 11β-hydroxylase (for corticosterone). These enzymes are nearly identical (they share 11β-hydroxylation and 18-hydroxylation functions), but aldosterone synthase is also able to perform an 18-oxidation. Moreover, aldosterone synthase is found within the zona glomerulosa at the outer edge of the adrenal cortex; 11β-hydroxylase is found in the zona fasciculata and zona glomerulosa.
Classification of corticosteroids
By chemical structure
In general, corticosteroids are grouped into four classes, based on chemical structure. Allergic reactions to one member of a class typically indicate an intolerance of all members of the class. This is known as the "Coopman classification". [42] [43]
The highlighted steroids are often used in the screening of allergies to topical steroids. [44]
Group A – Hydrocortisone type
Hydrocortisone, hydrocortisone acetate, cortisone acetate, tixocortol pivalate, prednisolone, methylprednisolone, and prednisone.
Amcinonide, budesonide, desonide, fluocinolone acetonide, fluocinonide, halcinonide, triamcinolone acetonide, and Deflazacort (O-isopropylidene derivative)
Group C – Betamethasone type
Beclometasone, betamethasone, dexamethasone, fluocortolone, halometasone, and mometasone.
Group D – Esters
Group D1 – Halogenated (less labile)
Alclometasone dipropionate, betamethasone dipropionate, betamethasone valerate, clobetasol propionate, clobetasone butyrate, fluprednidene acetate, and mometasone furoate.
Group D2 – Labile prodrug esters
Ciclesonide, cortisone acetate, hydrocortisone aceponate, hydrocortisone acetate, hydrocortisone buteprate, hydrocortisone butyrate, hydrocortisone valerate, prednicarbate, and tixocortol pivalate.
By route of administration
Topical steroids
For use topically on the skin, eye, and mucous membranes.
Topical corticosteroids are divided in potency classes I to IV in most countries (A to D in Japan). Seven categories are used in the United States to determine the level of potency of any given topical corticosteroid.
Inhaled steroids
For nasal mucosa, sinuses, bronchi, and lungs. [45]
This group includes:
- Flunisolide [46]
- Fluticasone furoate [46]
- Fluticasone propionate [46]
- Triamcinolone acetonide [46]
- Beclomethasone dipropionate [46]
- Budesonide [46]
- Mometasone furoate
- Ciclesonide
There also exist certain combination preparations such as Advair Diskus in the United States, containing fluticasone propionate and salmeterol (a long-acting bronchodilator), and Symbicort, containing budesonide and formoterol fumarate dihydrate (another long-acting bronchodilator). [46] They are both approved for use in children over 12 years old.
Oral forms
Such as prednisone, prednisolone, methylprednisolone, or dexamethasone. [47]
Systemic forms
Available in injectables for intravenous and parenteral routes. [47]
History
| Corticosteroid | Introduced |
|---|---|
| Cortisone | 1948 |
| Hydrocortisone | 1951 |
| Fludrocortisone acetate | 1954 [51] |
| Prednisolone | 1955 |
| Prednisone | 1955 [52] |
| Methylprednisolone | 1956 |
| Triamcinolone | 1956 |
| Dexamethasone | 1958 |
| Betamethasone | 1958 |
| Triamcinolone acetonide | 1958 |
| Fluorometholone | 1959 |
| Deflazacort | 1969 [53] |
Tadeusz Reichstein, Edward Calvin Kendall, and Philip Showalter Hench were awarded the Nobel Prize for Physiology and Medicine in 1950 for their work on hormones of the adrenal cortex, which culminated in the isolation of cortisone. [54]
Initially hailed as a miracle cure and liberally prescribed during the 1950s, steroid treatment brought about adverse events of such a magnitude that the next major category of anti-inflammatory drugs, the nonsteroidal anti-inflammatory drugs (NSAIDs), was so named in order to demarcate from the opprobrium. [55] Corticosteroids were voted Allergen of the Year in 2005 by the American Contact Dermatitis Society. [56]
Lewis Sarett of Merck & Co. was the first to synthesize cortisone, using a 36-step process that started with deoxycholic acid, which was extracted from ox bile. [57] The low efficiency of converting deoxycholic acid into cortisone led to a cost of US$200 per gram in 1947. Russell Marker, at Syntex, discovered a much cheaper and more convenient starting material, diosgenin from wild Mexican yams. His conversion of diosgenin into progesterone by a four-step process now known as Marker degradation was an important step in mass production of all steroidal hormones, including cortisone and chemicals used in hormonal contraception. [58]
In 1952, D.H. Peterson and H.C. Murray of Upjohn developed a process that used Rhizopus mold to oxidize progesterone into a compound that was readily converted to cortisone. [59] The ability to cheaply synthesize large quantities of cortisone from the diosgenin in yams resulted in a rapid drop in price to US$6 per gram[ when?], falling to $0.46 per gram by 1980. Percy Julian's research also aided progress in the field. [60] The exact nature of cortisone's anti-inflammatory action remained a mystery for years after, however, until the leukocyte adhesion cascade and the role of phospholipase A2 in the production of prostaglandins and leukotrienes was fully understood in the early 1980s.
Etymology
The cortico- part of the name refers to the adrenal cortex, which makes these steroid hormones. Thus a corticosteroid is a "cortex steroid".
See also
- List of corticosteroids
- List of corticosteroid cyclic ketals
- List of corticosteroid esters
- List of steroid abbreviations
References
- ^ a b c Nussey S, Whitehead S (2001). "The adrenal gland". Endocrinology: An Integrated Approach. Oxford: BIOS Scientific Publishers.
- ^ a b c d e f g Liu D, Ahmet A, Ward L, Krishnamoorthy P, Mandelcorn ED, Leigh R, et al. (August 2013). "A practical guide to the monitoring and management of the complications of systemic corticosteroid therapy". Allergy, Asthma, and Clinical Immunology. 9 (1): 30. doi: 10.1186/1710-1492-9-30. PMC 3765115. PMID 23947590.
- ^ Faggiano A, Mazzilli R, Natalicchio A, Adinolfi V, Argentiero A, Danesi R, et al. (December 2022). "Corticosteroids in oncology: Use, overuse, indications, contraindications. An Italian Association of Medical Oncology (AIOM)/ Italian Association of Medical Diabetologists (AMD)/ Italian Society of Endocrinology (SIE)/ Italian Society of Pharmacology (SIF) multidisciplinary consensus position paper". Critical Reviews in Oncology/Hematology. 180: 103826. doi: 10.1016/j.critrevonc.2022.103826. hdl: 10447/582211. PMID 36191821. S2CID 252663155.
- ^ Mohamadi A, Chan JJ, Claessen FM, Ring D, Chen NC (January 2017). "Corticosteroid Injections Give Small and Transient Pain Relief in Rotator Cuff Tendinosis: A Meta-analysis". Clinical Orthopaedics and Related Research. 475 (1): 232–243. doi: 10.1007/s11999-016-5002-1. PMC 5174041. PMID 27469590.
- ^ Werner R (2005). A massage therapist's guide to Pathology (3rd ed.). Pennsylvania: Lippincott Williams & Wilkins.
- ^ Carvalho-Recchia CA, Yannuzzi LA, Negrão S, Spaide RF, Freund KB, Rodriguez-Coleman H, et al. (October 2002). "Corticosteroids and central serous chorioretinopathy". Ophthalmology. 109 (10): 1834–1837. doi: 10.1016/S0161-6420(02)01117-X. PMID 12359603.
- ^ "The New York Times :: A Breathing Technique Offers Help for People With Asthma". buteykola.com. Archived from the original on 2012-07-24. Retrieved 2012-11-30.
- ^ Alderson P, Roberts I. "Plain Language Summary". Corticosteroids for acute traumatic brain injury. The Cochrane Collaboration. p. 2.
- ^ Alderson P, Roberts I (January 2005). Alderson P (ed.). "Corticosteroids for acute traumatic brain injury". The Cochrane Database of Systematic Reviews. 2005 (1): CD000196. doi: 10.1002/14651858.CD000196.pub2. PMC 7043302. PMID 15674869.
- ^ Lumry WR (October 1999). "A review of the preclinical and clinical data of newer intranasal steroids used in the treatment of allergic rhinitis". The Journal of Allergy and Clinical Immunology. 104 (4 Pt 1): S150–S158. doi: 10.1016/s0091-6749(99)70311-8. PMID 10518812.
- ^ Brook EM, Hu CH, Kingston KA, Matzkin EG (March 2017). "Corticosteroid Injections: A Review of Sex-Related Side Effects". Orthopedics. 40 (2): e211–e215. doi: 10.3928/01477447-20161116-07. PMID 27874912.
- ^ Luzzani F, Gallico L, Glässer A (1982). "In vitro and ex vivo binding to uterine progestin receptors of the rat as a tool to assay progestational activity of glucocorticoids". Methods and Findings in Experimental and Clinical Pharmacology. 4 (4): 237–242. PMID 7121132.
- ^ Cunningham GR, Goldzieher JW, de la Pena A, Oliver M (January 1978). "The mechanism of ovulation inhibition by triamcinolone acetonide". The Journal of Clinical Endocrinology and Metabolism. 46 (1): 8–14. doi: 10.1210/jcem-46-1-8. PMID 376542.
- ^ Tantisira KG, Lake S, Silverman ES, Palmer LJ, Lazarus R, Silverman EK, et al. (July 2004). "Corticosteroid pharmacogenetics: association of sequence variants in CRHR1 with improved lung function in asthmatics treated with inhaled corticosteroids". Human Molecular Genetics. 13 (13): 1353–1359. doi: 10.1093/hmg/ddh149. PMID 15128701.
- ^ Tantisira KG, Hwang ES, Raby BA, Silverman ES, Lake SL, Richter BG, et al. (December 2004). "TBX21: a functional variant predicts improvement in asthma with the use of inhaled corticosteroids". Proceedings of the National Academy of Sciences of the United States of America. 101 (52): 18099–18104. Bibcode: 2004PNAS..10118099T. doi: 10.1073/pnas.0408532102. PMC 539815. PMID 15604153.
- ^ Peters MC, Kerr S, Dunican EM, Woodruff PG, Fajt ML, Levy BD, et al. (January 2019). "Refractory airway type 2 inflammation in a large subgroup of asthmatic patients treated with inhaled corticosteroids". The Journal of Allergy and Clinical Immunology. 143 (1): 104–113.e14. doi: 10.1016/j.jaci.2017.12.1009. PMC 6128784. PMID 29524537.
- ^ Sumino K, Bacharier LB, Taylor J, Chadwick-Mansker K, Curtis V, Nash A, et al. (January 2020). "A Pragmatic Trial of Symptom-Based Inhaled Corticosteroid Use in African-American Children with Mild Asthma". The Journal of Allergy and Clinical Immunology. In Practice. 8 (1): 176–185.e2. doi: 10.1016/j.jaip.2019.06.030. PMID 31371165. S2CID 199380330.
- ^ "Managing Mild Asthma in Children Age Six and Older". Managing Mild Asthma in Children Age Six and Older | PCORI. 2021-08-13. Retrieved 2022-05-10.
- ^ Shirley DA, Moonah S (July 2016). "Fulminant Amebic Colitis after Corticosteroid Therapy: A Systematic Review". PLOS Neglected Tropical Diseases. 10 (7): e0004879. doi: 10.1371/journal.pntd.0004879. PMC 4965027. PMID 27467600.
- ^ Hall R. "Psychiatric Adverse Drug Reactions: Steroid Psychosis". Director of Research Monarch Health Corporation Marblehead, Massachusetts. Archived from the original on 2013-07-17. Retrieved 2013-06-23.
- ^ Korte SM (March 2001). "Corticosteroids in relation to fear, anxiety and psychopathology". Neuroscience and Biobehavioral Reviews. 25 (2): 117–142. doi: 10.1016/S0149-7634(01)00002-1. PMID 11323078. S2CID 8904351.
- ^ Swinburn CR, Wakefield JM, Newman SP, Jones PW (December 1988). "Evidence of prednisolone induced mood change ('steroid euphoria') in patients with chronic obstructive airways disease". British Journal of Clinical Pharmacology. 26 (6): 709–713. doi: 10.1111/j.1365-2125.1988.tb05309.x. PMC 1386585. PMID 3242575.
- ^ Benjamin H. Flores and Heather Kenna Gumina. The Neuropsychiatric Sequelae of Steroid Treatment. URL: http://www.dianafoundation.com/articles/df_04_article_01_steroids_pg01.html
- ^ Hasselgren PO, Alamdari N, Aversa Z, Gonnella P, Smith IJ, Tizio S (July 2010). "Corticosteroids and muscle wasting: role of transcription factors, nuclear cofactors, and hyperacetylation". Current Opinion in Clinical Nutrition and Metabolic Care. 13 (4): 423–428. doi: 10.1097/MCO.0b013e32833a5107. PMC 2911625. PMID 20473154.
- ^ Rodolico C, Bonanno C, Pugliese A, Nicocia G, Benvenga S, Toscano A (September 2020). "Endocrine myopathies: clinical and histopathological features of the major forms". Acta Myologica. 39 (3): 130–135. doi: 10.36185/2532-1900-017. PMC 7711326. PMID 33305169.
- ^ Donihi AC, Raval D, Saul M, Korytkowski MT, DeVita MA (2006). "Prevalence and predictors of corticosteroid-related hyperglycemia in hospitalized patients". Endocrine Practice. 12 (4): 358–362. doi: 10.4158/ep.12.4.358. PMID 16901792.
- ^ Blackburn D, Hux J, Mamdani M (September 2002). "Quantification of the Risk of Corticosteroid-induced Diabetes Mellitus Among the Elderly". Journal of General Internal Medicine. 17 (9): 717–720. doi: 10.1046/j.1525-1497.2002.10649.x. PMC 1495107. PMID 12220369.
- ^ Chalitsios CV, Shaw DE, McKeever TM (January 2021). "Risk of osteoporosis and fragility fractures in asthma due to oral and inhaled corticosteroids: two population-based nested case-control studies". Thorax. 76 (1): 21–28. doi: 10.1136/thoraxjnl-2020-215664. PMID 33087546. S2CID 224822416.
- ^ Chalitsios CV, McKeever TM, Shaw DE (January 2021). "Incidence of osteoporosis and fragility fractures in asthma: a UK population-based matched cohort study". The European Respiratory Journal. 57 (1). doi: 10.1183/13993003.01251-2020. PMID 32764111. S2CID 221078530.
- ^ Chalitsios CV, Shaw DE, McKeever TM (May 2021). "Corticosteroids and bone health in people with asthma: A systematic review and meta-analysis". Respiratory Medicine. 181: 106374. doi: 10.1016/j.rmed.2021.106374. PMID 33799052. S2CID 232771681.
- ^ Zhang L, Prietsch SO, Ducharme FM (July 2014). "Inhaled corticosteroids in children with persistent asthma: effects on growth". The Cochrane Database of Systematic Reviews. 2014 (7): CD009471. doi: 10.1002/14651858.CD009471.pub2. PMC 8407362. PMID 25030198.
- ^ Pecora PG, Kaplan B (1996). "Corticosteroids and ulcers: is there an association?". The Annals of Pharmacotherapy. 30 (7–8): 870–872. doi: 10.1177/106002809603000729. PMID 8826575. S2CID 13594804.
- ^ Martínek J, Hlavova K, Zavada F, Seifert B, Rejchrt S, Urban O, Zavoral M (October 2010). ""A surviving myth"--corticosteroids are still considered ulcerogenic by a majority of physicians". Scandinavian Journal of Gastroenterology. 45 (10): 1156–1161. doi: 10.3109/00365521.2010.497935. PMID 20569095. S2CID 5140517.
- ^ Abouammoh MA (2015). "Advances in the treatment of central serous chorioretinopathy". Saudi Journal of Ophthalmology. 29 (4): 278–286. doi: 10.1016/j.sjopt.2015.01.007. PMC 4625218. PMID 26586979.
- ^ Petzold A, Braithwaite T, van Oosten BW, Balk L, Martinez-Lapiscina EH, Wheeler R, et al. (January 2020). "Case for a new corticosteroid treatment trial in optic neuritis: review of updated evidence". Journal of Neurology, Neurosurgery, and Psychiatry. 91 (1): 9–14. doi: 10.1136/jnnp-2019-321653. PMC 6952848. PMID 31740484.
- ^ Fukushima C, Matsuse H, Tomari S, Obase Y, Miyazaki Y, Shimoda T, Kohno S (June 2003). "Oral candidiasis associated with inhaled corticosteroid use: comparison of fluticasone and beclomethasone". Annals of Allergy, Asthma & Immunology. 90 (6): 646–651. doi: 10.1016/S1081-1206(10)61870-4. PMID 12839324.
- ^ Shepard TH, Brent RL, Friedman JM, Jones KL, Miller RK, Moore CA, Polifka JE (April 2002). "Update on new developments in the study of human teratogens". Teratology. 65 (4): 153–161. doi: 10.1002/tera.10032. PMID 11948561.
- ^ Nnoruka EN, Daramola OO, Ike SO (2007). "Misuse and abuse of topical steroids: implications". Expert Review of Dermatology. 2 (1): 31–40. doi: 10.1586/17469872.2.1.31. Retrieved 2014-12-18.
- ^ Rathi SK, D'Souza P (July 2012). "Rational and ethical use of topical corticosteroids based on safety and efficacy". Indian Journal of Dermatology. 57 (4): 251–259. doi: 10.4103/0019-5154.97655. PMC 3401837. PMID 22837556.
- ^ Fukaya M, Sato K, Sato M, Kimata H, Fujisawa S, Dozono H, et al. (2014). "Topical steroid addiction in atopic dermatitis". Drug, Healthcare and Patient Safety. 6: 131–138. doi: 10.2147/dhps.s69201. PMC 4207549. PMID 25378953.
- ^ Aljebab F, Choonara I, Conroy S (April 2016). "Systematic review of the toxicity of short-course oral corticosteroids in children". Archives of Disease in Childhood. 101 (4): 365–370. doi: 10.1136/archdischild-2015-309522. PMC 4819633. PMID 26768830.
- ^ Rietschel RL (2007). Fisher's Contact Dermatitis, 6/e. Hamilton, Ont: BC Decker Inc. p. 256. ISBN 978-1-55009-378-0.
- ^ Coopman S, Degreef H, Dooms-Goossens A (July 1989). "Identification of cross-reaction patterns in allergic contact dermatitis from topical corticosteroids". The British Journal of Dermatology. 121 (1): 27–34. doi: 10.1111/j.1365-2133.1989.tb01396.x. PMID 2757954. S2CID 40425526.
- ^ Wolverton SE (2001). Comprehensive Dermatologic Drug Therapy. WB Saunders. p. 562.
- ^ "Asthma Steroids: Inhaled Steroids, Side Effects, Benefits, and More". Webmd.com. Retrieved 2012-11-30.
- ^ a b c d e f g Mayo Clinic Staff (September 2015). "Asthma Medications: Know your options". MayoClinic.org. Retrieved 2018-02-27.
- ^ a b "Systemic steroids (corticosteroids). DermNet NZ". . DermNet NZ. 2012-05-19. Retrieved 2012-11-30.
- ^ Khan MO, Park KK, Lee HJ (2005). "Antedrugs: an approach to safer drugs". Current Medicinal Chemistry. 12 (19): 2227–2239. doi: 10.2174/0929867054864840. PMID 16178782.
- ^ Calvert DN (August 1962). "Anti-inflammatory steroids". Wisconsin Medical Journal. 61: 403–404. PMID 13875857.
- ^ Alberto Conde-Taboada (2012). Dermatological Treatments. Bentham Science Publishers. pp. 35–36. ISBN 978-1-60805-234-9.
- ^ William Andrew Publishing (22 October 2013). Pharmaceutical Manufacturing Encyclopedia, 3rd Edition. Elsevier. pp. 1642–1643. ISBN 978-0-8155-1856-3.
- ^ Kim KW, Roh JK, Wee HJ, Kim C (14 November 2016). Cancer Drug Discovery: Science and History. Springer. pp. 169–. ISBN 978-94-024-0844-7.
- ^ Nayak, Surajit; Acharjya, Basanti (December 19, 2008). "Deflazacort versus other glucocorticoids: A comparison". Indian Journal of Dermatology. 53 (4): 167–170. doi: 10.4103/0019-5154.44786. PMC 2763756. PMID 19882026.
- ^ Kendall EC (April 1951). "The development of cortisone as a therapeutic agent" (PDF). Antibiotics & Chemotherapy (Northfield, Ill.). 1 (1): 7–15. PMID 24540604. Archived from the original (PDF) on 15 April 2017.
- ^ Buer JK (October 2014). "Origins and impact of the term 'NSAID'". Inflammopharmacology. 22 (5): 263–267. doi: 10.1007/s10787-014-0211-2. hdl: 10852/45403. PMID 25064056. S2CID 16777111.
- ^ "Contact Allergen of the Year: Corticosteroids: Introduction". Medscape.com. 2005-06-13. Retrieved 2012-11-30.
- ^ US 2462133, Sarett LH, "Process of Treating Pregnene Compounds", issued 1947
- ^ Marker RE, Wagner RB (September 1947). "Steroidal sapogenins". Journal of the American Chemical Society. 69 (9): 2167–2230. doi: 10.1021/ja01201a032. PMID 20262743.
- ^ Peterson DH, Murray HC (1952). "Microbiological Oxygenation of Steroids at Carbon 11". J. Am. Chem. Soc. 74 (7): 1871–2. doi: 10.1021/ja01127a531.
- ^ US 2752339, Julian L, Cole JW, Meyer EW, Karpel WJ, "Preparation of Cortisone", issued 1956