| Coronavirus spike glycoprotein | |||||||||
|---|---|---|---|---|---|---|---|---|---|
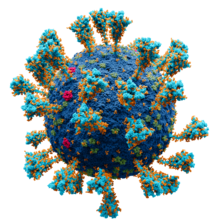 Model of the external structure of the SARS-CoV-2 virion.
[1]
● Blue: envelope ● Turquoise: spike glycoprotein (S) ● Red: envelope proteins (E) ● Green: membrane proteins (M) ● Orange: glycan | |||||||||
| Identifiers | |||||||||
| Symbol | CoV_S1 | ||||||||
| Pfam | PF01600 | ||||||||
| InterPro | IPR002551 | ||||||||
| |||||||||
Spike (S) glycoprotein (sometimes also called spike protein, [2] formerly known as E2 [3]) is the largest of the four major structural proteins found in coronaviruses. [4] The spike protein assembles into trimers that form large structures, called spikes or peplomers, [3] that project from the surface of the virion. [4] [5] The distinctive appearance of these spikes when visualized using negative stain transmission electron microscopy, "recalling the solar corona", [6] gives the virus family its main name. [2]
The function of the spike glycoprotein is to mediate viral entry into the host cell by first interacting with molecules on the exterior cell surface and then fusing the viral and cellular membranes. Spike glycoprotein is a class I fusion protein that contains two regions, known as S1 and S2, responsible for these two functions. The S1 region contains the receptor-binding domain that binds to receptors on the cell surface. Coronaviruses use a very diverse range of receptors; SARS-CoV (which causes SARS) and SARS-CoV-2 (which causes COVID-19) both interact with angiotensin-converting enzyme 2 (ACE2). The S2 region contains the fusion peptide and other fusion infrastructure necessary for membrane fusion with the host cell, a required step for infection and viral replication. Spike glycoprotein determines the virus' host range (which organisms it can infect) and cell tropism (which cells or tissues it can infect within an organism). [4] [5] [7] [8]
Spike glycoprotein is highly immunogenic. Antibodies against spike glycoprotein are found in patients recovered from SARS and COVID-19. Neutralizing antibodies target epitopes on the receptor-binding domain. [9] Most COVID-19 vaccine development efforts in response to the COVID-19 pandemic aim to activate the immune system against the spike protein. [10] [11] [12]
Structure

The spike protein is very large, often 1200 to 1400 amino acid residues long; [8] it is 1273 residues in SARS-CoV-2. [5] It is a single-pass transmembrane protein with a short C-terminal tail on the interior of the virus, a transmembrane helix, and a large N-terminal ectodomain exposed on the virus exterior. [5] [7]
Spike glycoprotein forms homotrimers in which three copies of the protein interact through their ectodomains. [5] [7] The trimer structures have been described as club- pear-, or petal-shaped. [3] Each spike protein contains two regions known as S1 and S2, and in the assembled trimer the S1 regions at the N-terminal end form the portion of the protein furthest from the viral surface while the S2 regions form a flexible "stalk" containing most of the protein-protein interactions that hold the trimer in place. [7]
S1
| Betacoronavirus spike glycoprotein S1, receptor binding | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | bCoV_S1_RBD | ||||||||
| Pfam | PF09408 | ||||||||
| InterPro | IPR018548 | ||||||||
| |||||||||
| Betacoronavirus-like spike glycoprotein S1, N-terminal | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | bCoV_S1_N | ||||||||
| Pfam | PF16451 | ||||||||
| InterPro | IPR032500 | ||||||||
| |||||||||
The S1 region of the spike glycoprotein is responsible for interacting with receptor molecules on the surface of the host cell in the first step of viral entry. [4] [7] S1 contains two domains, called the N-terminal domain (NTD) and C-terminal domain (CTD), [2] [7] sometimes also known as the A and B domains. [14] Depending on the coronavirus, either or both domains may be used as receptor-binding domains (RBD). Target receptors can be very diverse, including cell surface receptor proteins and sugars such as sialic acids as receptors or coreceptors. [2] [7] In general, the NTD binds sugar molecules while the CTD binds proteins, with the exception of mouse hepatitis virus which uses its NTD to interact with a protein receptor called CEACAM1. [7] The NTD has a galectin-like protein fold, but binds sugar molecules somewhat differently than galectins. [7] The observed binding of N-acetylneuraminic acid by the NTD [15] and loss of that binding through mutation of the corresponding sugar binding pocket in emergent variants of concern has suggested a potential role for tranisent sugar-binding in the zoonosis of SARS-CoV-2, consistent with prior evolutionary proposals. [16]
The CTD is responsible for the interactions of MERS-CoV with its receptor dipeptidyl peptidase-4, [7] and those of SARS-CoV [7] and SARS-CoV-2 [5] with their receptor angiotensin-converting enzyme 2 (ACE2). The CTD of these viruses can be further divided into two subdomains, known as the core and the extended loop or receptor-binding motif (RBM), where most of the residues that directly contact the target receptor are located. [5] [7] There are subtle differences, mainly in the RBM, between the SARS-CoV and SARS-CoV-2 spike proteins' interactions with ACE2. [5] Comparisons of spike proteins from multiple coronaviruses suggest that divergence in the RBM region can account for differences in target receptors, even when the core of the S1 CTD is structurally very similar. [7]
Within coronavirus lineages, as well as across the four major coronavirus subgroups, the S1 region is less well conserved than S2, as befits its role in interacting with virus-specific host cell receptors. [4] [5] [7] Within the S1 region, the NTD is more highly conserved than the CTD. [7]
S2
| Coronavirus spike glycoprotein S2 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | CoV_S2 | ||||||||
| Pfam | PF01601 | ||||||||
| InterPro | IPR002552 | ||||||||
| |||||||||
The S2 region of spike glycoprotein is responsible for membrane fusion between the viral envelope and the host cell, enabling entry of the virus' genome into the cell. [5] [7] [8] The S2 region contains the fusion peptide, a stretch of mostly hydrophobic amino acids whose function is to enter and destabilize the host cell membrane. [5] [8] S2 also contains two heptad repeat subdomains known as HR1 and HR2, sometimes called the "fusion core" region. [5] These subdomains undergo dramatic conformational changes during the fusion process to form a six-helix bundle, a characteristic feature of the class I fusion proteins. [5] [8] The S2 region is also considered to include the transmembrane helix and C-terminal tail located in the interior of the virion. [5]
Relative to S1, the S2 region is very well conserved among coronaviruses. [5] [7]
Post-translational modifications

Spike glycoprotein is heavily glycosylated through N-linked glycosylation. [4] Studies of the SARS-CoV-2 spike protein have also reported O-linked glycosylation in the S1 region. [19] The C-terminal tail, located in the interior of the virion, is enriched in cysteine residues and is palmitoylated. [5] [20]
Spike proteins are activated through proteolytic cleavage. They are cleaved by host cell proteases at the S1-S2 boundary and later at what is known as the S2' site at the N-terminus of the fusion peptide. [4] [5] [7] [8]
Conformational change
Like other class I fusion proteins, the spike protein undergoes a very large conformational change during the fusion process. [4] [5] [7] [8] Both the pre-fusion and post-fusion states of several coronaviruses, especially SARS-CoV-2, have been studied by cryo-electron microscopy. [5] [21] [22] [23] Functionally important protein dynamics have also been observed within the pre-fusion state, in which the relative orientations of some of the S1 regions relative to S2 in a trimer can vary. In the closed state, all three S1 regions are packed closely and the region that makes contact with host cell receptors is sterically inaccessible, while the open states have one or two S1 RBDs more accessible for receptor binding, in an open or "up" conformation. [5]

Expression and localization
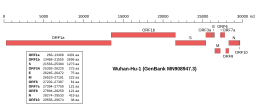
Genomic organisation of isolate Wuhan-Hu-1, the earliest sequenced sample of SARS-CoV-2, indicating the location of the S gene | |
| NCBI genome ID | 86693 |
|---|---|
| Genome size | 29,903 bases |
| Year of completion | 2020 |
| Genome browser ( UCSC) | |
The gene encoding the spike protein is located toward the 3' end of the virus's positive-sense RNA genome, along with the genes for the other three structural proteins and various virus-specific accessory proteins. [4] [5] Protein trafficking of spike proteins appears to depend on the coronavirus subgroup: when expressed in isolation without other viral proteins, spike proteins from betacoronaviruses are able to reach the cell surface, while those from alphacoronaviruses and gammacoronaviruses are retained intracellularly. In the presence of the M protein, spike protein trafficking is altered and instead is retained at the ERGIC, the site at which viral assembly occurs. [20] In SARS-CoV-2, both the M and the E protein modulate spike protein trafficking through different mechanisms. [24]

The spike protein is not required for viral assembly or the formation of virus-like particles; [20] however, presence of spike may influence the size of the envelope. [26] Incorporation of the spike protein into virions during assembly and budding is dependent on protein-protein interactions with the M protein through the C-terminal tail. [20] [24] Examination of virions using cryo-electron microscopy suggests that there are approximately 25 [27] to 100 spike trimers per virion. [22] [26]
Function
The spike protein is responsible for viral entry into the host cell, a required early step in viral replication. It is essential for replication. [2] It performs this function in two steps, first binding to a receptor on the surface of the host cell through interactions with the S1 region, and then fusing the viral and cellular membranes through the action of the S2 region. [7] [8] [9] The location of fusion varies depending on the specific coronavirus, with some able to enter at the plasma membrane and others entering from endosomes after endocytosis. [8]
Attachment
The interaction of the receptor-binding domain in the S1 region with its target receptor on the cell surface initiates the process of viral entry. Different coronaviruses target different cell-surface receptors, sometimes using sugar molecules such as sialic acids, or forming protein-protein interactions with proteins exposed on the cell surface. [7] [9] Different coronaviruses vary widely in their target receptor. The presence of a target receptor that S1 can bind is a determinant of host range and cell tropism. [7] [9] [28] Human serum albumin binds to the S1 region, competing with ACE2 and therefore restricting viral entry into cells. [29]
Proteolytic cleavage
Proteolytic cleavage of the spike protein, sometimes known as "priming", is required for membrane fusion. Relative to other class I fusion proteins, this process is complex and requires two cleavages at different sites, one at the S1/S2 boundary and one at the S2' site to release the fusion peptide. [5] [7] [9] Coronaviruses vary in which part of the viral life cycle these cleavages occur, particularly the S1/S2 cleavage. Many coronaviruses are cleaved at S1/S2 before viral exit from the virus-producing cell, by furin and other proprotein convertases; [7] in SARS-CoV-2 a polybasic furin cleavage site is present at this position. [5] [9] Others may be cleaved by extracellular proteases such as elastase, by proteases located on the cell surface after receptor binding, or by proteases found in lysosomes after endocytosis. [7] The specific proteases responsible for this cleavage depends on the virus, cell type, and local environment. [8] In SARS-CoV, the serine protease TMPRSS2 is important for this process, with additional contributions from cysteine proteases cathepsin B and cathepsin L in endosomes. [8] [9] [37] Trypsin and trypsin-like proteases have also been reported to contribute. [8] In SARS-CoV-2, TMPRSS2 is the primary protease for S2' cleavage, and its presence is reported to be essential for viral infection, [5] [9] with cathepsin L protease being functional, but not essential. [37]
Membrane fusion

Like other class I fusion proteins, the spike protein in its pre-fusion conformation is in a metastable state. [7] A dramatic conformational change is triggered to induce the heptad repeats in the S2 region to refold into an extended six-helix bundle, causing the fusion peptide to interact with the cell membrane and bringing the viral and cell membranes into close proximity. [5] [7] Receptor binding and proteolytic cleavage (sometimes known as "priming") are required, but additional triggers for this conformational change vary depending on the coronavirus and local environment. [40] In vitro studies of SARS-CoV suggest a dependence on calcium concentration. [8] Unusually for coronaviruses, infectious bronchitis virus, which infects birds, can be triggered by low pH alone; for other coronaviruses, low pH is not itself a trigger but may be required for activity of proteases, which in turn are required for fusion. [8] [40] The location of membrane fusion—at the plasma membrane or in endosomes—may vary based on the availability of these triggers for conformational change. [40] Fusion of the viral and cell membranes permits the entry of the virus' positive-sense RNA genome into the host cell cytosol, after which expression of viral proteins begins. [2] [4] [9]
In addition to fusion of viral and host cell membranes, some coronavirus spike proteins can initiate membrane fusion between infected cells and neighboring cells, forming syncytia. [41] This behavior can be observed in infected cells in cell culture. [42] Syncytia have been observed in patient tissue samples from infections with SARS-CoV, MERS-CoV, and SARS-CoV-2, [42] though some reports highlight a difference in syncytia formation between the SARS-CoV and SARS-CoV-2 spikes attributed to sequence differences near the S1/S2 cleavage site. [43] [44] [45]
Immunogenicity
Because it is exposed on the surface of the virus, the spike protein is a major antigen to which neutralizing antibodies are developed. [2] [9] [46] [47] Its extensive glycosylation can serve as a glycan shield that hides epitopes from the immune system. [9] [18] Due to the outbreak of SARS and the COVID-19 pandemic, antibodies to SARS-CoV and SARS-CoV-2 spike proteins have been extensively studied. [46] Antibodies to the SARS-CoV and SARS-CoV-2 spike proteins have been identified that target epitopes on the receptor-binding domain [9] [46] [48] or interfere with the process of conformational change. [9] The majority of antibodies from infected individuals target the receptor-binding domain. [46] [49] [50] More recently antibodies targeting the S2 subunit of the spike protein have been reported with broad neutralization activities against variants. [51]
COVID-19 response
Vaccines
In response to the COVID-19 pandemic, a number of COVID-19 vaccines have been developed using a variety of technologies, including mRNA vaccines and viral vector vaccines. Most vaccine development has targeted the spike protein. [10] [11] [12] Building on techniques previously used in vaccine research aimed at respiratory syncytial virus and SARS-CoV, many SARS-CoV-2 vaccine development efforts have used constructs that include mutations to stabilize the spike protein's pre-fusion conformation, facilitating development of antibodies against epitopes exposed in this conformation. [52] [53]
According to a study published in January 2023, markedly elevated levels of full-length spike protein unbound by antibodies were found in people who developed postvaccine myocarditis (vs. controls that remained healthy). However, these results do not alter the risk-benefit ratio favoring vaccination against COVID-19 to prevent severe clinical outcomes. [54][ non-primary source needed]
Monoclonal antibodies

Monoclonal antibodies that target the receptor-binding domain of the spike protein have been developed as COVID-19 treatments. As of July 8, 2021, three monoclonal antibody products had received Emergency Use Authorization in the United States: [56] bamlanivimab/etesevimab, [57] [58] casirivimab/imdevimab, [59] and sotrovimab. [60] Bamlanivimab/etesevimab was not recommended in the United States due to the increase in SARS-CoV-2 variants that are less susceptible to these antibodies. [56]
SARS-CoV-2 variants
Throughout the COVID-19 pandemic, the genome of SARS-CoV-2 viruses was sequenced many times, resulting in identification of thousands of distinct variants. [61] Many of these possess mutations that change the amino acid sequence of the spike protein. In a World Health Organization analysis from July 2020, the spike (S) gene was the second most frequently mutated in the genome, after ORF1ab (which encodes most of the virus' nonstructural proteins). [61] The evolution rate in the spike gene is higher than that observed in the genome overall. [62] Analyses of SARS-CoV-2 genomes suggests that some sites in the spike protein sequence, particularly in the receptor-binding domain, are of evolutionary importance [63] and are undergoing positive selection. [49] [64]
Spike protein mutations raise concern because they may affect infectivity or transmissibility, or facilitate immune escape. [49] The mutation D614 G has arisen independently in multiple viral lineages and become dominant among sequenced genomes; [65] [66] it may have advantages in infectivity and transmissibility [49] possibly due to increasing the density of spikes on the viral surface, [67] increasing the proportion of binding-competent conformations or improving stability, [68] but it does not affect vaccines. [69] The mutation N501Y is common to the Alpha, Beta, Gamma and Omicron Variants of SARS-CoV-2 and has contributed to enhanced infection and transmission, [70] reduced vaccine efficacy, [71] and the ability of SARS-CoV-2 to infect new rodent species. [72] N501Y increases the affinity of Spike for human ACE2 by around 10-fold, [73] which could underlie some of fitness advantages conferred by this mutation even though the relationship between affinity and infectivity is complex. [74] The mutation P681R alters the furin cleavage site, and has been responsible for increased infectivity, transmission and global impact of the SARS-CoV-2 Delta variant. [75] [76] Mutations at position E484, particularly E484 K, have been associated with immune escape and reduced antibody binding. [49] [62]
The SARS-CoV-2 Omicron variant is notable for having an unusually high number of mutations in the spike protein. [77] The SARS CoV-2 spike gene (S gene, S-gene) mutation 69–70del (Δ69-70) causes a TaqPath PCR test probe to not bind to its S gene target, leading to S gene target failure (SGTF) in SARS CoV-2 positive samples. This effect was used as a marker to monitor the propagation of the Alpha variant [78] [79] and the Omicron variant. [80]
Additional Key Role in Illness
In 2021, Circulation Research and Salk had a new study that proves COVID-19 can be also a vascular disease, not only respiratory disease. The scientists created an “pseudovirus”, surrounded by SARS-CoV-2 spike proteins but without any actual virus. And pseudovirus resulted in damaging lungs and arteries of animal models. It shows SARS-CoV-2 spike protein alone can cause vascular disease and could explain some covid-19 patients who suffered from strokes, or other vascular problems in other parts of human body at the same time. The team replicated the process by removing replicating capabilities of virus and showed the same damaging effect on vascular cells again. [81] [82]
Misinformation
During the COVID-19 pandemic, anti-vaccination misinformation about COVID-19 circulated on social media platforms related to the spike protein's role in COVID-19 vaccines. Spike proteins were said to be dangerously " cytotoxic" and mRNA vaccines containing them therefore in themselves dangerous. Spike proteins are not cytotoxic or dangerous. [83] [84] Even though studies has found that spike proteins are causing amyloid-disease associated blood coagulation and fibrinolytic disturbances, along with neurologic and cardiac problems.[ needs copy edit] [85] Spike proteins were also said to be "shed" by vaccinated people, in an erroneous allusion to the phenomenon of vaccine-induced viral shedding, which is a rare effect of live-virus vaccines unlike those used for COVID-19. "Shedding" of spike proteins is not possible. [86] [87]
Evolution, conservation and recombination
The class I fusion proteins, a group whose well-characterized examples include the coronavirus spike protein, influenza virus hemagglutinin, and HIV Gp41, are thought to be evolutionarily related. [7] [88] The S2 region of the spike protein responsible for membrane fusion is more highly conserved than the S1 region responsible for receptor interactions. [4] [5] [7] The S1 region appears to have undergone significant diversifying selection. [89]
Within the S1 region, the N-terminal domain (NTD) is more conserved than the C-terminal domain (CTD). [7] The NTD's galectin-like protein fold suggests a relationship with structurally similar cellular proteins from which it may have evolved through gene capture from the host. [7] It has been suggested that the CTD may have evolved from the NTD by gene duplication. [7] The surface-exposed position of the CTD, vulnerable to the host immune system, may place this region under high selective pressure. [7] Comparisons of the structures of different coronavirus CTDs suggests they may be under diversifying selection [90] and in some cases, distantly related coronaviruses that use the same cell-surface receptor may do so through convergent evolution. [14]
References
- ^ Solodovnikov, Alexey; Arkhipova, Valeria (29 July 2021). "Достоверно красиво: как мы сделали 3D-модель SARS-CoV-2" [Truly beautiful: how we made the SARS-CoV-2 3D model] (in Russian). N+1. Archived from the original on 30 July 2021. Retrieved 30 July 2021.
- ^ a b c d e f g Deng, X.; Baker, S.C. (2021). "Coronaviruses: Molecular Biology (Coronaviridae)". Encyclopedia of Virology: 198–207. doi: 10.1016/B978-0-12-814515-9.02550-9. ISBN 9780128145166.
- ^ a b c Masters, Paul S. (2006). "The Molecular Biology of Coronaviruses". Advances in Virus Research. 66: 193–292. doi: 10.1016/S0065-3527(06)66005-3. ISBN 9780120398690. PMC 7112330. PMID 16877062.
- ^ a b c d e f g h i j k l m n o p Wang, Yuhang; Grunewald, Matthew; Perlman, Stanley (2020). "Coronaviruses: An Updated Overview of Their Replication and Pathogenesis". Coronaviruses. Methods in Molecular Biology. Vol. 2203. pp. 1–29. doi: 10.1007/978-1-0716-0900-2_1. ISBN 978-1-0716-0899-9. PMC 7682345. PMID 32833200.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa Zhu, Chaogeng; He, Guiyun; Yin, Qinqin; Zeng, Lin; Ye, Xiangli; Shi, Yongzhong; Xu, Wei (14 June 2021). "Molecular biology of the SARs-CoV-2 spike protein: A review of current knowledge". Journal of Medical Virology. 93 (10): 5729–5741. doi: 10.1002/jmv.27132. PMC 8427004. PMID 34125455.
- ^ "Virology: Coronaviruses". Nature. 220 (5168): 650. November 1968. Bibcode: 1968Natur.220..650.. doi: 10.1038/220650b0. PMC 7086490.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae af ag Li, Fang (29 September 2016). "Structure, Function, and Evolution of Coronavirus Spike Proteins". Annual Review of Virology. 3 (1): 237–261. doi: 10.1146/annurev-virology-110615-042301. PMC 5457962. PMID 27578435.
- ^ a b c d e f g h i j k l m n Millet, Jean Kaoru; Whittaker, Gary R. (April 2018). "Physiological and molecular triggers for SARS-CoV membrane fusion and entry into host cells". Virology. 517: 3–8. doi: 10.1016/j.virol.2017.12.015. PMC 7112017. PMID 29275820.
- ^ a b c d e f g h i j k l m n V’kovski, Philip; Kratzel, Annika; Steiner, Silvio; Stalder, Hanspeter; Thiel, Volker (March 2021). "Coronavirus biology and replication: implications for SARS-CoV-2". Nature Reviews Microbiology. 19 (3): 155–170. doi: 10.1038/s41579-020-00468-6. PMC 7592455. PMID 33116300.
- ^ a b Flanagan, Katie L.; Best, Emma; Crawford, Nigel W.; Giles, Michelle; Koirala, Archana; Macartney, Kristine; Russell, Fiona; Teh, Benjamin W.; Wen, Sophie CH (2 October 2020). "Progress and Pitfalls in the Quest for Effective SARS-CoV-2 (COVID-19) Vaccines". Frontiers in Immunology. 11: 579250. doi: 10.3389/fimmu.2020.579250. hdl: 11343/251733. PMC 7566192. PMID 33123165.
- ^ a b Le, Tung Thanh; Cramer, Jakob P.; Chen, Robert; Mayhew, Stephen (October 2020). "Evolution of the COVID-19 vaccine development landscape". Nature Reviews Drug Discovery. 19 (10): 667–668. doi: 10.1038/d41573-020-00151-8. PMID 32887942. S2CID 221503034.
- ^ a b Kyriakidis, Nikolaos C.; López-Cortés, Andrés; González, Eduardo Vásconez; Grimaldos, Alejandra Barreto; Prado, Esteban Ortiz (December 2021). "SARS-CoV-2 vaccines strategies: a comprehensive review of phase 3 candidates". npj Vaccines. 6 (1): 28. doi: 10.1038/s41541-021-00292-w. PMC 7900244. PMID 33619260.
- ^ a b Wrapp, Daniel; Wang, Nianshuang; Corbett, Kizzmekia S.; Goldsmith, Jory A.; Hsieh, Ching-Lin; Abiona, Olubukola; Graham, Barney S.; McLellan, Jason S. (13 March 2020). "Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation". Science. 367 (6483): 1260–1263. Bibcode: 2020Sci...367.1260W. doi: 10.1126/science.abb2507. PMC 7164637. PMID 32075877.
- ^ a b Hulswit, R.J.G.; de Haan, C.A.M.; Bosch, B.-J. (2016). "Coronavirus Spike Protein and Tropism Changes". Advances in Virus Research. 96: 29–57. doi: 10.1016/bs.aivir.2016.08.004. ISBN 9780128047361. PMC 7112277. PMID 27712627.
- ^ https://doi.org/10.1126/science.abm3125
- ^ https://doi.org/10.1016/S0021-9258(18)63732-9
- ^ Zimmer, Carl (9 October 2020). "The Coronavirus Unveiled". The New York Times. Retrieved 12 August 2021.
- ^ a b Casalino, Lorenzo; Gaieb, Zied; Goldsmith, Jory A.; Hjorth, Christy K.; Dommer, Abigail C.; Harbison, Aoife M.; Fogarty, Carl A.; Barros, Emilia P.; Taylor, Bryn C.; McLellan, Jason S.; Fadda, Elisa; Amaro, Rommie E. (28 October 2020). "Beyond Shielding: The Roles of Glycans in the SARS-CoV-2 Spike Protein". ACS Central Science. 6 (10): 1722–1734. doi: 10.1021/acscentsci.0c01056. PMC 7523240. PMID 33140034.
- ^ Shajahan, Asif; Supekar, Nitin T; Gleinich, Anne S; Azadi, Parastoo (9 December 2020). "Deducing the N- and O-glycosylation profile of the spike protein of novel coronavirus SARS-CoV-2". Glycobiology. 30 (12): 981–988. doi: 10.1093/glycob/cwaa042. PMC 7239183. PMID 32363391.
- ^ a b c d Ujike, Makoto; Taguchi, Fumihiro (3 April 2015). "Incorporation of Spike and Membrane Glycoproteins into Coronavirus Virions". Viruses. 7 (4): 1700–1725. doi: 10.3390/v7041700. PMC 4411675. PMID 25855243.
- ^ Walls, Alexandra C.; Park, Young-Jun; Tortorici, M. Alejandra; Wall, Abigail; McGuire, Andrew T.; Veesler, David (April 2020). "Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein". Cell. 181 (2): 281–292.e6. doi: 10.1016/j.cell.2020.02.058. PMC 7102599. PMID 32155444.
- ^ a b Klein, Steffen; Cortese, Mirko; Winter, Sophie L.; Wachsmuth-Melm, Moritz; Neufeldt, Christopher J.; Cerikan, Berati; Stanifer, Megan L.; Boulant, Steeve; Bartenschlager, Ralf; Chlanda, Petr (December 2020). "SARS-CoV-2 structure and replication characterized by in situ cryo-electron tomography". Nature Communications. 11 (1): 5885. Bibcode: 2020NatCo..11.5885K. doi: 10.1038/s41467-020-19619-7. PMC 7676268. PMID 33208793.
- ^ Cai, Yongfei; Zhang, Jun; Xiao, Tianshu; Peng, Hanqin; Sterling, Sarah M.; Walsh, Richard M.; Rawson, Shaun; Rits-Volloch, Sophia; Chen, Bing (25 September 2020). "Distinct conformational states of SARS-CoV-2 spike protein". Science. 369 (6511): 1586–1592. Bibcode: 2020Sci...369.1586C. doi: 10.1126/science.abd4251. PMC 7464562. PMID 32694201.
- ^ a b Boson, Bertrand; Legros, Vincent; Zhou, Bingjie; Siret, Eglantine; Mathieu, Cyrille; Cosset, François-Loïc; Lavillette, Dimitri; Denolly, Solène (January 2021). "The SARS-CoV-2 envelope and membrane proteins modulate maturation and retention of the spike protein, allowing assembly of virus-like particles". Journal of Biological Chemistry. 296: 100111. doi: 10.1074/jbc.RA120.016175. PMC 7833635. PMID 33229438.
- ^ Goodsell, David S.; Voigt, Maria; Zardecki, Christine; Burley, Stephen K. (6 August 2020). "Integrative illustration for coronavirus outreach". PLOS Biology. 18 (8): e3000815. doi: 10.1371/journal.pbio.3000815. PMC 7433897. PMID 32760062.
- ^ a b Neuman, Benjamin W.; Kiss, Gabriella; Kunding, Andreas H.; Bhella, David; Baksh, M. Fazil; Connelly, Stephen; Droese, Ben; Klaus, Joseph P.; Makino, Shinji; Sawicki, Stanley G.; Siddell, Stuart G.; Stamou, Dimitrios G.; Wilson, Ian A.; Kuhn, Peter; Buchmeier, Michael J. (April 2011). "A structural analysis of M protein in coronavirus assembly and morphology". Journal of Structural Biology. 174 (1): 11–22. doi: 10.1016/j.jsb.2010.11.021. PMC 4486061. PMID 21130884.
- ^ Ke, Zunlong; Oton, Joaquin; Qu, Kun; Cortese, Mirko; Zila, Vojtech; McKeane, Lesley; Nakane, Takanori; Zivanov, Jasenko; Neufeldt, Christopher J.; Cerikan, Berati; Lu, John M.; Peukes, Julia; Xiong, Xiaoli; Kräusslich, Hans-Georg; Scheres, Sjors H. W.; Bartenschlager, Ralf; Briggs, John A. G. (17 December 2020). "Structures and distributions of SARS-CoV-2 spike proteins on intact virions". Nature. 588 (7838): 498–502. Bibcode: 2020Natur.588..498K. doi: 10.1038/s41586-020-2665-2. PMC 7116492. PMID 32805734.
- ^ a b Lim, Yvonne; Ng, Yan; Tam, James; Liu, Ding (25 July 2016). "Human Coronaviruses: A Review of Virus–Host Interactions". Diseases. 4 (3): 26. doi: 10.3390/diseases4030026. PMC 5456285. PMID 28933406.
- ^ Varricchio, Romualdo; De Simone, Giovanna; Vita, Gian Marco; Nocera Cariola, Walter; Viscardi, Maurizio; Brandi, Sergio; Picazio, Gerardo; Zerbato, Verena; Koncan, Raffaella; Segat, Ludovica; Di Bella, Stefano; Fusco, Giovanna; Ascenzi, Paolo; di Masi, Alessandra (2024). "Human serum albumin binds spike protein and protects cells from SARS-CoV-2 infection by modulating the RAS pathway". Aspects of Molecular Medicine. 3: 100033. doi: 10.1016/j.amolm.2023.100033.
- ^ Yeager, Curtis L.; Ashmun, Richard A.; Williams, Richard K.; Cardellichio, Christine B.; Shapiro, Linda H.; Look, A. Thomas; Holmes, Kathryn V. (June 1992). "Human aminopeptidase N is a receptor for human coronavirus 229E". Nature. 357 (6377): 420–422. Bibcode: 1992Natur.357..420Y. doi: 10.1038/357420a0. PMC 7095410. PMID 1350662.
- ^ Hofmann, H.; Pyrc, K.; van der Hoek, L.; Geier, M.; Berkhout, B.; Pohlmann, S. (31 May 2005). "Human coronavirus NL63 employs the severe acute respiratory syndrome coronavirus receptor for cellular entry". Proceedings of the National Academy of Sciences. 102 (22): 7988–7993. Bibcode: 2005PNAS..102.7988H. doi: 10.1073/pnas.0409465102. PMC 1142358. PMID 15897467.
- ^ Huang, Xingchuan; Dong, Wenjuan; Milewska, Aleksandra; Golda, Anna; Qi, Yonghe; Zhu, Quan K.; Marasco, Wayne A.; Baric, Ralph S.; Sims, Amy C.; Pyrc, Krzysztof; Li, Wenhui; Sui, Jianhua (15 July 2015). "Human Coronavirus HKU1 Spike Protein Uses O -Acetylated Sialic Acid as an Attachment Receptor Determinant and Employs Hemagglutinin-Esterase Protein as a Receptor-Destroying Enzyme". Journal of Virology. 89 (14): 7202–7213. doi: 10.1128/JVI.00854-15. PMC 4473545. PMID 25926653.
- ^ Künkel, Frank; Herrler, Georg (July 1993). "Structural and Functional Analysis of the Surface Protein of Human Coronavirus OC43". Virology. 195 (1): 195–202. doi: 10.1006/viro.1993.1360. PMC 7130786. PMID 8317096.
- ^ Raj, V. Stalin; Mou, Huihui; Smits, Saskia L.; Dekkers, Dick H. W.; Müller, Marcel A.; Dijkman, Ronald; Muth, Doreen; Demmers, Jeroen A. A.; Zaki, Ali; Fouchier, Ron A. M.; Thiel, Volker; Drosten, Christian; Rottier, Peter J. M.; Osterhaus, Albert D. M. E.; Bosch, Berend Jan; Haagmans, Bart L. (March 2013). "Dipeptidyl peptidase 4 is a functional receptor for the emerging human coronavirus-EMC". Nature. 495 (7440): 251–254. Bibcode: 2013Natur.495..251R. doi: 10.1038/nature12005. PMC 7095326. PMID 23486063.
- ^ Li, Wenhui; Moore, Michael J.; Vasilieva, Natalya; Sui, Jianhua; Wong, Swee Kee; Berne, Michael A.; Somasundaran, Mohan; Sullivan, John L.; Luzuriaga, Katherine; Greenough, Thomas C.; Choe, Hyeryun; Farzan, Michael (November 2003). "Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus". Nature. 426 (6965): 450–454. Bibcode: 2003Natur.426..450L. doi: 10.1038/nature02145. PMC 7095016. PMID 14647384.
- ^ https://doi.org/10.1126/science.abm3125
- ^ a b Jackson CB, Farzan M, Chen B, Choe H (2022). "Mechanisms of SARS-CoV-2 entry into cells". Nature Reviews Molecular Cell Biology. 23 (1): 3–20. doi: 10.1038/s41580-021-00418-x. PMC 8491763. PMID 34611326.
- ^ Walls, Alexandra C.; Xiong, Xiaoli; Park, Young-Jun; Tortorici, M. Alejandra; Snijder, Joost; Quispe, Joel; Cameroni, Elisabetta; Gopal, Robin; Dai, Mian; Lanzavecchia, Antonio; Zambon, Maria; Rey, Félix A.; Corti, Davide; Veesler, David (February 2019). "Unexpected Receptor Functional Mimicry Elucidates Activation of Coronavirus Fusion". Cell. 176 (5): 1026–1039.e15. doi: 10.1016/j.cell.2018.12.028. PMC 6751136. PMID 30712865.
- ^ Fan, Xiaoyi; Cao, Duanfang; Kong, Lingfei; Zhang, Xinzheng (December 2020). "Cryo-EM analysis of the post-fusion structure of the SARS-CoV spike glycoprotein". Nature Communications. 11 (1): 3618. Bibcode: 2020NatCo..11.3618F. doi: 10.1038/s41467-020-17371-6. PMC 7367865. PMID 32681106.
- ^ a b c White, Judith M.; Whittaker, Gary R. (June 2016). "Fusion of Enveloped Viruses in Endosomes". Traffic. 17 (6): 593–614. doi: 10.1111/tra.12389. PMC 4866878. PMID 26935856.
- ^ Belouzard, Sandrine; Millet, Jean K.; Licitra, Beth N.; Whittaker, Gary R. (20 June 2012). "Mechanisms of Coronavirus Cell Entry Mediated by the Viral Spike Protein". Viruses. 4 (6): 1011–1033. doi: 10.3390/v4061011. PMC 3397359. PMID 22816037.
- ^ a b Buchrieser, Julian; Dufloo, Jérémy; Hubert, Mathieu; Monel, Blandine; Planas, Delphine; Rajah, Maaran Michael; Planchais, Cyril; Porrot, Françoise; Guivel-Benhassine, Florence; Van der Werf, Sylvie; Casartelli, Nicoletta; Mouquet, Hugo; Bruel, Timothée; Schwartz, Olivier (December 2020). "Syncytia formation by SARS-CoV-2-infected cells". The EMBO Journal. 39 (23): e106267. doi: 10.15252/embj.2020106267. PMC 7646020. PMID 33051876.
- ^ Zhang, Zhengrong; Zheng, You; Niu, Zubiao; Zhang, Bo; Wang, Chenxi; Yao, Xiaohong; Peng, Haoran; Franca, Del Nonno; Wang, Yunyun; Zhu, Yichao; Su, Yan; Tang, Meng; Jiang, Xiaoyi; Ren, He; He, Meifang; Wang, Yuqi; Gao, Lihua; Zhao, Ping; Shi, Hanping; Chen, Zhaolie; Wang, Xiaoning; Piacentini, Mauro; Bian, Xiuwu; Melino, Gerry; Liu, Liang; Huang, Hongyan; Sun, Qiang (20 April 2021). "SARS-CoV-2 spike protein dictates syncytium-mediated lymphocyte elimination". Cell Death & Differentiation. 28 (9): 2765–2777. doi: 10.1038/s41418-021-00782-3. PMC 8056997. PMID 33879858.
- ^ Braga, Luca; Ali, Hashim; Secco, Ilaria; Chiavacci, Elena; Neves, Guilherme; Goldhill, Daniel; Penn, Rebecca; Jimenez-Guardeño, Jose M.; Ortega-Prieto, Ana M.; Bussani, Rossana; Cannatà, Antonio; Rizzari, Giorgia; Collesi, Chiara; Schneider, Edoardo; Arosio, Daniele; Shah, Ajay M.; Barclay, Wendy S.; Malim, Michael H.; Burrone, Juan; Giacca, Mauro (3 June 2021). "Drugs that inhibit TMEM16 proteins block SARS-CoV-2 spike-induced syncytia". Nature. 594 (7861): 88–93. Bibcode: 2021Natur.594...88B. doi: 10.1038/s41586-021-03491-6. PMC 7611055. PMID 33827113.
- ^ Lin, Liangyu; Li, Qing; Wang, Ying; Shi, Yufang (June 2021). "Syncytia formation during SARS-CoV-2 lung infection: a disastrous unity to eliminate lymphocytes". Cell Death & Differentiation. 28 (6): 2019–2021. doi: 10.1038/s41418-021-00795-y. PMC 8114657. PMID 33981020.
- ^ a b c d Ho, Mitchell (April 2020). "Perspectives on the development of neutralizing antibodies against SARS-CoV-2". Antibody Therapeutics. 3 (2): 109–114. doi: 10.1093/abt/tbaa009. ISSN 2516-4236. PMC 7291920. PMID 32566896.
- ^ Yang, Lifei; Liu, Weihan; Yu, Xin; Wu, Meng; Reichert, Janice M.; Ho, Mitchell (July 2020). "COVID-19 antibody therapeutics tracker: a global online database of antibody therapeutics for the prevention and treatment of COVID-19". Antibody Therapeutics. 3 (3): 205–212. doi: 10.1093/abt/tbaa020. ISSN 2516-4236. PMC 7454247. PMID 33215063.
- ^ Premkumar, Lakshmanane; Segovia-Chumbez, Bruno; Jadi, Ramesh; Martinez, David R.; Raut, Rajendra; Markmann, Alena; Cornaby, Caleb; Bartelt, Luther; Weiss, Susan; Park, Yara; Edwards, Caitlin E.; Weimer, Eric; Scherer, Erin M.; Rouphael, Nadine; Edupuganti, Srilatha; Weiskopf, Daniela; Tse, Longping V.; Hou, Yixuan J.; Margolis, David; Sette, Alessandro; Collins, Matthew H.; Schmitz, John; Baric, Ralph S.; de Silva, Aravinda M. (11 June 2020). "The receptor binding domain of the viral spike protein is an immunodominant and highly specific target of antibodies in SARS-CoV-2 patients". Science Immunology. 5 (48): eabc8413. doi: 10.1126/sciimmunol.abc8413. PMC 7292505. PMID 32527802.
- ^ a b c d e Harvey, William T.; Carabelli, Alessandro M.; Jackson, Ben; Gupta, Ravindra K.; Thomson, Emma C.; Harrison, Ewan M.; Ludden, Catherine; Reeve, Richard; Rambaut, Andrew; Peacock, Sharon J.; Robertson, David L. (July 2021). "SARS-CoV-2 variants, spike mutations and immune escape". Nature Reviews Microbiology. 19 (7): 409–424. doi: 10.1038/s41579-021-00573-0. PMC 8167834. PMID 34075212.
- ^ Hong, Jessica; Kwon, Hyung Joon; Cachau, Raul; Chen, Catherine Z.; Butay, Kevin John; Duan, Zhijian; Li, Dan; Ren, Hua; Liang, Tianyuzhou; Zhu, Jianghai; Dandey, Venkata P.; Martin, Negin P.; Esposito, Dominic; Ortega-Rodriguez, Uriel; Xu, Miao (3 May 2022). "Dromedary camel nanobodies broadly neutralize SARS-CoV-2 variants". Proceedings of the National Academy of Sciences of the United States of America. 119 (18): e2201433119. Bibcode: 2022PNAS..11901433H. doi: 10.1073/pnas.2201433119. ISSN 1091-6490. PMC 9170159. PMID 35476528.
- ^ Buffington, Jesse; Duan, Zhijian; Kwon, Hyung Joon; Hong, Jessica; Li, Dan; Feng, Mingqian; Xie, Hang; Ho, Mitchell (June 2023). "Identification of nurse shark VNAR single-domain antibodies targeting the spike S2 subunit of SARS-CoV-2". FASEB Journal. 37 (6): e22973. doi: 10.1096/fj.202202099RR. ISSN 1530-6860. PMC 10715488. PMID 37191949. S2CID 258717083.
- ^ Fauci, Anthony S. (9 April 2021). "The story behind COVID-19 vaccines". Science. 372 (6538): 109. Bibcode: 2021Sci...372..109F. doi: 10.1126/science.abi8397. PMID 33833099. S2CID 233186026.
- ^ Koenig, Paul-Albert; Schmidt, Florian I. (17 June 2021). "Spike D614G — A Candidate Vaccine Antigen Against Covid-19". New England Journal of Medicine. 384 (24): 2349–2351. doi: 10.1056/NEJMcibr2106054. PMID 34133867.
-
^ Yonker, Lael M.; Swank, Zoe; Bartsch, Yannic C.; Burns, Madeleine D.; Kane, Abigail; Boribong, Brittany P.; Davis, Jameson P.; Loiselle, Maggie; Novak, Tanya; Senussi, Yasmeen; Cheng, Chi-An; Burgess, Eleanor; Edlow, Andrea G.; Chou, Janet; Dionne, Audrey; Balaguru, Duraisamy; Lahoud-Rahme, Manuella; Arditi, Moshe; Julg, Boris; Randolph, Adrienne G.;
Alter, Galit; Fasano, Alessio; Walt, David R. (4 January 2023).
"Circulating Spike Protein Detected in Post–COVID-19 mRNA Vaccine Myocarditis". Circulation. 147 (11): 867–876.
doi:
10.1161/CIRCULATIONAHA.122.061025.
PMC
10010667.
PMID
36597886.
S2CID
255475007.
Extensive antibody profiling and T-cell responses in the individuals who developed postvaccine myocarditis were essentially indistinguishable from those of vaccinated control subjects, [...] A notable finding was that markedly elevated levels of full-length spike protein (33.9±22.4 pg/mL), unbound by antibodies, were detected in the plasma of individuals with postvaccine myocarditis, [...] (unpaired t test; P<0.0001).
- ^ Hansen, Johanna; Baum, Alina; Pascal, Kristen E.; Russo, Vincenzo; Giordano, Stephanie; Wloga, Elzbieta; Fulton, Benjamin O.; Yan, Ying; Koon, Katrina; Patel, Krunal; Chung, Kyung Min; Hermann, Aynur; Ullman, Erica; Cruz, Jonathan; Rafique, Ashique; Huang, Tammy; Fairhurst, Jeanette; Libertiny, Christen; Malbec, Marine; Lee, Wen-yi; Welsh, Richard; Farr, Glen; Pennington, Seth; Deshpande, Dipali; Cheng, Jemmie; Watty, Anke; Bouffard, Pascal; Babb, Robert; Levenkova, Natasha; Chen, Calvin; Zhang, Bojie; Romero Hernandez, Annabel; Saotome, Kei; Zhou, Yi; Franklin, Matthew; Sivapalasingam, Sumathi; Lye, David Chien; Weston, Stuart; Logue, James; Haupt, Robert; Frieman, Matthew; Chen, Gang; Olson, William; Murphy, Andrew J.; Stahl, Neil; Yancopoulos, George D.; Kyratsous, Christos A. (21 August 2020). "Studies in humanized mice and convalescent humans yield a SARS-CoV-2 antibody cocktail". Science. 369 (6506): 1010–1014. Bibcode: 2020Sci...369.1010H. doi: 10.1126/science.abd0827. PMC 7299284. PMID 32540901.
- ^ a b "Therapeutic Management of Nonhospitalized Adults With COVID-19". Covid-19 Treatment Guidelines. National Institutes of Health. Archived from the original on 4 December 2021. Retrieved 11 August 2021.
- ^ "etesevimab". IUPHAR/BPS Guide to Pharmacology. Retrieved 10 February 2021.
- ^ "Lilly announces agreement with U.S. government to supply 300,000 vials of investigational neutralizing antibody bamlanivimab (LY-CoV555) in an effort to fight COVID-19". Eli Lilly and Company (Press release). 28 October 2020.
- ^ "Casirivimab injection, solution, concentrate Imdevimab injection, solution, concentrate REGEN-COV- casirivimab and imdevimab kit". DailyMed. Retrieved 18 March 2021.
- ^ "Sotrovimab injection, solution, concentrate". DailyMed. Retrieved 15 June 2021.
- ^ a b Koyama, Takahiko; Platt, Daniel; Parida, Laxmi (1 July 2020). "Variant analysis of SARS-CoV-2 genomes". Bulletin of the World Health Organization. 98 (7): 495–504. doi: 10.2471/BLT.20.253591. PMC 7375210. PMID 32742035.
- ^ a b Winger, Anna; Caspari, Thomas (27 May 2021). "The Spike of Concern—The Novel Variants of SARS-CoV-2". Viruses. 13 (6): 1002. doi: 10.3390/v13061002. PMC 8229995. PMID 34071984.
- ^ Saputri, Dianita S.; Li, Songling; van Eerden, Floris J.; Rozewicki, John; Xu, Zichang; Ismanto, Hendra S.; Davila, Ana; Teraguchi, Shunsuke; Katoh, Kazutaka; Standley, Daron M. (17 September 2020). "Flexible, Functional, and Familiar: Characteristics of SARS-CoV-2 Spike Protein Evolution". Frontiers in Microbiology. 11: 2112. doi: 10.3389/fmicb.2020.02112. PMC 7527407. PMID 33042039.
- ^ Cagliani, Rachele; Forni, Diego; Clerici, Mario; Sironi, Manuela (June 2020). "Computational Inference of Selection Underlying the Evolution of the Novel Coronavirus, Severe Acute Respiratory Syndrome Coronavirus 2". Journal of Virology. 94 (12): e00411-20. doi: 10.1128/JVI.00411-20. PMC 7307108. PMID 32238584.
- ^ Isabel, Sandra; Graña-Miraglia, Lucía; Gutierrez, Jahir M.; Bundalovic-Torma, Cedoljub; Groves, Helen E.; Isabel, Marc R.; Eshaghi, AliReza; Patel, Samir N.; Gubbay, Jonathan B.; Poutanen, Tomi; Guttman, David S.; Poutanen, Susan M. (December 2020). "Evolutionary and structural analyses of SARS-CoV-2 D614G spike protein mutation now documented worldwide". Scientific Reports. 10 (1): 14031. Bibcode: 2020NatSR..1014031I. doi: 10.1038/s41598-020-70827-z. PMC 7441380. PMID 32820179.
- ^ Korber, Bette; Fischer, Will M.; Gnanakaran, Sandrasegaram; Yoon, Hyejin; Theiler, James; Abfalterer, Werner; Hengartner, Nick; Giorgi, Elena E.; Bhattacharya, Tanmoy; Foley, Brian; Hastie, Kathryn M.; Parker, Matthew D.; Partridge, David G.; Evans, Cariad M.; Freeman, Timothy M.; de Silva, Thushan I.; McDanal, Charlene; Perez, Lautaro G.; Tang, Haili; Moon-Walker, Alex; Whelan, Sean P.; LaBranche, Celia C.; Saphire, Erica O.; Montefiori, David C.; Angyal, Adrienne; Brown, Rebecca L.; Carrilero, Laura; Green, Luke R.; Groves, Danielle C.; Johnson, Katie J.; Keeley, Alexander J.; Lindsey, Benjamin B.; Parsons, Paul J.; Raza, Mohammad; Rowland-Jones, Sarah; Smith, Nikki; Tucker, Rachel M.; Wang, Dennis; Wyles, Matthew D. (August 2020). "Tracking Changes in SARS-CoV-2 Spike: Evidence that D614G Increases Infectivity of the COVID-19 Virus". Cell. 182 (4): 812–827.e19. doi: 10.1016/j.cell.2020.06.043. PMC 7332439. PMID 32697968.
- ^ Zhang, Lizhou; Jackson, Cody B.; Mou, Huihui; Ojha, Amrita; Peng, Haiyong; Quinlan, Brian D.; Rangarajan, Erumbi S.; Pan, Andi; Vanderheiden, Abigail; Suthar, Mehul S.; Li, Wenhui; Izard, Tina; Rader, Christoph; Farzan, Michael; Choe, Hyeryun (December 2020). "SARS-CoV-2 spike-protein D614G mutation increases virion spike density and infectivity". Nature Communications. 11 (1): 6013. Bibcode: 2020NatCo..11.6013Z. doi: 10.1038/s41467-020-19808-4. PMC 7693302. PMID 33243994.
- ^ Jackson, Cody B.; Zhang, Lizhou; Farzan, Michael; Choe, Hyeryun (January 2021). "Functional importance of the D614G mutation in the SARS-CoV-2 spike protein". Biochemical and Biophysical Research Communications. 538: 108–115. doi: 10.1016/j.bbrc.2020.11.026. PMC 7664360. PMID 33220921.
- ^ McAuley, Alexander J. (October 2020). "Experimental and in silico evidence suggests vaccines are unlikely to be affected by D614G mutation in SARS-CoV-2 spike protein". npj Vaccines. 5: 96. doi: 10.1038/s41541-020-00246-8. PMC 7546614. PMID 33083031.
- ^ Liu, Yang (November 2021). "The N501Y Spike substitution enhances SARS-CoV-2 infection and transmission". Nature. 602 (7896): 294–299. doi: 10.1038/s41586-021-04245-0. PMC 8900207. PMID 34818667. S2CID 244647259.
- ^ Abdool Karim, S.S. (2021). "New SARS-CoV-2 variants — clinical, public health, and vaccine implications". New England Journal of Medicine. 384 (19): 1866–1868. doi: 10.1056/NEJMc2100362. PMC 8008749. PMID 33761203.
- ^ Kuiper, Michael (2021). "But Mouse, you are not alone: On some severe acute respiratory syndrome coronavirus 2 variants infecting mice". ILAR Journal. 62 (1–2): 48–59. doi: 10.1093/ilar/ilab031. PMC 9236659. PMID 35022734.
- ^ Barton, Michael I; MacGowan, Stuart A; Kutuzov, Mikhail A; Dushek, Omer; Barton, Geoffrey John; van der Merwe, P Anton (26 August 2021). Fouchier, Ron AM; Van der Meer, Jos W; Fouchier, Ron AM (eds.). "Effects of common mutations in the SARS-CoV-2 Spike RBD and its ligand, the human ACE2 receptor on binding affinity and kinetics". eLife. 10: e70658. doi: 10.7554/eLife.70658. ISSN 2050-084X. PMC 8480977. PMID 34435953.
- ^ MacGowan, Stuart A.; Barton, Michael I.; Kutuzov, Mikhail; Dushek, Omer; Merwe, P. Anton van der; Barton, Geoffrey J. (2 March 2022). "Missense variants in human ACE2 strongly affect binding to SARS-CoV-2 Spike providing a mechanism for ACE2 mediated genetic risk in Covid-19: A case study in affinity predictions of interface variants". PLOS Computational Biology. 18 (3): e1009922. Bibcode: 2022PLSCB..18E9922M. doi: 10.1371/journal.pcbi.1009922. ISSN 1553-7358. PMC 8920257. PMID 35235558.
- ^ Callaway, Ewen (2021). "The mutation that helps Delta spread like wildfire". Nature. 596 (7873): 472–473. Bibcode: 2021Natur.596..472C. doi: 10.1038/d41586-021-02275-2. PMID 34417582. S2CID 237254466.
- ^ Peacock, T.P. (2021). "The SARS-CoV-2 variants associated with infections in India, B.1.617, show enhanced Spike cleavage by furin" (PDF). bioRxiv. doi: 10.1101/2021.05.28.446163. S2CID 235249387.
- ^ "Classification of Omicron (B.1.1.529): SARS-CoV-2 Variant of Concern". World Health Organization. 26 November 2021. Archived from the original on 26 November 2021. Retrieved 26 November 2021.
- ^ Brown KA, Gubbay J, Hopkins J, Patel S, Buchan SA, Daneman N, Goneau LW (25 May 2021). "S-Gene Target Failure as a Marker of Variant B.1.1.7 Among SARS-CoV-2 Isolates in the Greater Toronto Area, December 2020 to March 2021". JAMA. 325 (20): 2115–2116. doi: 10.1001/jama.2021.5607. ISSN 0098-7484. PMC 8033504. PMID 33830171.
- ^ Methods for the detection and identification of SARS-CoV-2 variants (Technical report). Stockholm and Copenhagen: European Centre for Disease Prevention and Control/World Health Organization Regional Office for Europe. 3 March 2021. Diagnostic screening assays of known VOCs.
- ^ SARS-CoV-2 variants of concern and variants under investigation in England Variant of concern: Omicron, VOC21NOV-01 (B.1.1.529), technical briefing 30 (PDF) (Briefing). Public Health England. 3 December 2021. GOV-10547. Archived (PDF) from the original on 11 December 2021. Retrieved 15 December 2021.
- ^ "The novel coronavirus' spike protein plays additional key role in illness". Salk researchers. 30 April 2021. Archived from the original on 1 December 2022.
- ^ Lei, Yuyang; Zhang, Jiao; Schiavon, Cara R.; He, Ming; Chen, Lili; Shen, Hui; Zhang, Yichi; Yin, Qian; Cho, Yoshitake; Andrade, Leonardo; Shadel, Gerald S.; Hepokoski, Mark; Lei, Ting; Wang, Hongliang; Zhang, Jin; Yuan, Jason X.-J.; Malhotra, Atul; Manor, Uri; Wang, Shengpeng; Yuan, Zu-Yi; Shyy, John Y-J. (31 March 2021). "SARS-CoV-2 Spike Protein Impairs Endothelial Function via Downregulation of ACE 2". Circulation Research. 128 (9). AHA Journals: 1323–1326. doi: 10.1161/CIRCRESAHA.121.318902. PMC 8091897. PMID 33784827. S2CID 232430540.
- ^ "COVID-19 vaccines are not 'cytotoxic'" (Fact check). Reuters. 18 June 2021.
- ^ Gorski DH (24 May 2021). "The 'deadly' coronavirus spike protein (according to antivaxxers)". Science-Based Medicine.
- ^ Nyström, Sofie; Hammarström, Per (2022). "Amyloidogenesis of SARS-CoV-2 Spike Protein". Journal of the American Chemical Society. 144 (20): 8945–8950. doi: 10.1021/jacs.2c03925. PMC 9136918. PMID 35579205.
- ^ McCarthy B (5 May 2021). "Debunking the anti-vaccine hoax about 'vaccine shedding'". PolitiFact. Retrieved 11 May 2021.
- ^ Fiore K (29 April 2021). "The Latest Anti-Vax Myth: 'Vaccine Shedding'". MedPage Today. Retrieved 11 May 2021.
- ^ Vance, Tyler D.R.; Lee, Jeffrey E. (July 2020). "Virus and eukaryote fusogen superfamilies". Current Biology. 30 (13): R750–R754. Bibcode: 2020CBio...30.R750V. doi: 10.1016/j.cub.2020.05.029. PMC 7336913. PMID 32634411.
- ^ Li, F. (1 March 2012). "Evidence for a Common Evolutionary Origin of Coronavirus Spike Protein Receptor-Binding Subunits". Journal of Virology. 86 (5): 2856–2858. doi: 10.1128/jvi.06882-11. PMC 3302248. PMID 22205743.
- ^ Shang, Jian; Zheng, Yuan; Yang, Yang; Liu, Chang; Geng, Qibin; Luo, Chuming; Zhang, Wei; Li, Fang (23 April 2018). "Cryo-EM structure of infectious bronchitis coronavirus spike protein reveals structural and functional evolution of coronavirus spike proteins". PLOS Pathogens. 14 (4): e1007009. doi: 10.1371/journal.ppat.1007009. PMC 5933801. PMID 29684066.
External links
- Scudellari, Megan (28 July 2021). "How the coronavirus infects cells — and why Delta is so dangerous". Nature. Retrieved 15 August 2021.
- Iwasa, Janet; Meyer, Miriah; Lex, Alexander; Rogers, Jen; Liu, Ann (Hui); Riggi, Margot. "Building a visual consensus model of the SARS-CoV-2 life cycle". Animation Lab. University of Utah. Retrieved 15 August 2021.
