| Bone scintigraphy | |
|---|---|
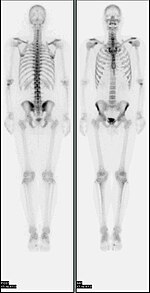 A nuclear medicine whole-body bone scan. The nuclear medicine whole-body bone scan is generally used in evaluations of various bone-related pathology, such as for bone pain, stress fracture, nonmalignant bone lesions, bone infections, or the spread of cancer to the bone. | |
| ICD-9-CM | 92.14 |
| OPS-301 code | 3-705 |
| MedlinePlus | 003833 |
A bone scan or bone scintigraphy /sɪnˈtɪɡrəfi/ is a nuclear medicine imaging technique of the bone. It can help diagnose a number of bone conditions, including cancer of the bone or metastasis, location of bone inflammation and fractures (that may not be visible in traditional X-ray images), and bone infection (osteomyelitis). [1]
Nuclear medicine provides functional imaging and allows visualisation of bone metabolism or bone remodeling, which most other imaging techniques (such as X-ray computed tomography, CT) cannot. [2] [3] Bone scintigraphy competes with positron emission tomography (PET) for imaging of abnormal metabolism in bones, but is considerably less expensive. [4] Bone scintigraphy has higher sensitivity but lower specificity than CT or MRI for diagnosis of scaphoid fractures following negative plain radiography. [5]
History

Some of the earliest investigations into skeletal metabolism were carried out by George de Hevesy in the 1930s, using phosphorus-32 and by Charles Pecher in the 1940s. [6] [7]
In the 1950s and 1960s calcium-45 was investigated, but as a beta emitter proved difficult to image. Imaging of positron and gamma emitters such as fluorine-18 and isotopes of strontium with rectilinear scanners was more useful. [8] [9] Use of technetium-99m (99mTc) labelled phosphates, diphosphonates or similar agents, as in the modern technique, was first proposed in 1971. [10] [11]
Principle
The most common radiopharmaceutical for bone scintigraphy is 99mTc with methylene diphosphonate (MDP). [12] Other bone radiopharmaceuticals include 99mTc with HDP, HMDP and DPD. [13] [14] MDP adsorbs onto the crystalline hydroxyapatite mineral of bone. [15] Mineralisation occurs at osteoblasts, representing sites of bone growth, where MDP (and other diphosphates) "bind to the hydroxyapatite crystals in proportion to local blood flow and osteoblastic activity and are therefore markers of bone turnover and bone perfusion". [16] [17]
The more active the bone turnover, the more radioactive material will be seen. Some tumors, fractures and infections show up as areas of increased uptake. [18]
Note that the technique depends on the osteoblastic activity during remodelling and repair processes following initial osteolytic activity. This leads to a limitation of the applicability of this imaging technique with diseases not featuring this osteoblastic (reactive) activity, for example with multiple myeloma. Scintigraphic images remain falsely negative for a long period of time and therefore have only limited diagnostic value. In these cases CT or MRI scans are preferred for diagnosis and staging.
Technique
In a typical bone scan technique, the patient is injected (usually into a vein in the arm or hand, occasionally the foot) with up to 740 MBq of technetium-99m-MDP and then scanned with a gamma camera, which captures planar anterior and posterior or single photon emission computed tomography (SPECT) images. [19] [14] In order to view small lesions SPECT imaging technique may be preferred over planar scintigraphy. [20]
In a single phase protocol (skeletal imaging alone), which will primarily highlight osteoblasts, images are usually acquired 2–5 hours after the injection (after four hours 50–60% of the activity will be fixed to bones). [19] [14] [21] A two or three phase protocol utilises additional scans at different points after the injection to obtain additional diagnostic information. A dynamic (i.e. multiple acquired frames) study immediately after the injection captures perfusion information. [21] [22] A second phase "blood pool" image following the perfusion (if carried out in a three phase technique) can help to diagnose inflammatory conditions or problems of blood supply. [23]
A typical effective dose obtained during a bone scan is 6.3 millisieverts (mSv). [24]
-
Person undergoing a bone scan on the skull
-
A patient undergoing a SPECT bone scan.
PET bone imaging
Although bone scintigraphy generally refers to gamma camera imaging of 99mTc radiopharmaceuticals, imaging with positron emission tomography (PET) scanners is also possible, using fluorine-18 sodium fluoride ([18F]NaF).
For quantitative measurements, 99mTc-MDP has some advantages over [18F]NaF. MDP renal clearance is not affected by urine flow rate and simplified data analysis can be employed which assumes steady state conditions. It has negligible tracer uptake in red blood cells, therefore correction for plasma to whole blood ratios is not required unlike [18F]NaF. However, disadvantages include higher rates of protein binding (from 25% immediately after injection to 70% after 12 hours leading to the measurement of freely available MDP over time), and less diffusibility due to higher molecular weight than [18F]NaF, leading to lower capillary permeability. [25]
There are several advantages of the PET technique, which are common to PET imaging in general, including improved spatial resolution and more developed attenuation correction techniques. Patient experience is improved as imaging can be started much more quickly following radiopharmaceutical injection (30–45 minutes, compared to 2–3 hours for MDP/HDP). [26] [27] [18F]NaF PET is hampered by high demand for scanners, and limited tracer availability. [28] [29]
References
- ^ Bahk, Yong-Whee (2000). Combined scintigraphic and radiographic diagnosis of bone and joint diseases (2nd ed.). Berlin, Heidelberg: Springer. p. 3. ISBN 9783662041062.
- ^ Ćwikła, Jarosław B. (2013). "New imaging techniques in reumathology: MRI, scintigraphy and PET". Polish Journal of Radiology. 78 (3): 48–56. doi: 10.12659/PJR.889138. PMC 3789933. PMID 24115960.
- ^ Livieratos, Lefteris (2012). "Basic Principles of SPECT and PET Imaging". In Fogelman, Ignac; Gnanasegaran, Gopinath; van der Wall, Hans (eds.). Radionuclide and Hybrid Bone Imaging. Berlin: Springer. p. 345. doi: 10.1007/978-3-642-02400-9_12. ISBN 978-3-642-02399-6.
- ^ O'Sullivan, Gerard J (2015). "Imaging of bone metastasis: An update". World Journal of Radiology. 7 (8): 202–11. doi: 10.4329/wjr.v7.i8.202. PMC 4553252. PMID 26339464.
- ^ Mallee, WH; Wang, J; Poolman, RW; Kloen, P; Maas, M; de Vet, HC; Doornberg, JN (5 June 2015). "Computed tomography versus magnetic resonance imaging versus bone scintigraphy for clinically suspected scaphoid fractures in patients with negative plain radiographs". The Cochrane Database of Systematic Reviews. 2015 (6): CD010023. doi: 10.1002/14651858.CD010023.pub2. PMC 6464799. PMID 26045406.
- ^ Pecher, Charles (1941). "Biological Investigations with Radioactive Calcium and Strontium". Proceedings of the Society for Experimental Biology and Medicine. 46 (1): 86–91. doi: 10.3181/00379727-46-11899. ISSN 0037-9727. S2CID 88173163.
- ^ Carlson, Sten (8 July 2009). "A Glance At The History Of Nuclear Medicine". Acta Oncologica. 34 (8): 1095–1102. doi: 10.3109/02841869509127236. PMID 8608034.
- ^ Bridges, R. L.; Wiley, C. R.; Christian, J. C.; Strohm, A. P. (11 May 2007). "An Introduction to Na18F Bone Scintigraphy: Basic Principles, Advanced Imaging Concepts, and Case Examples". Journal of Nuclear Medicine Technology. 35 (2): 64–76. doi: 10.2967/jnmt.106.032870. PMID 17496010.
- ^ Fleming, William H.; McIlraith, James D.; Richard King, Capt. E. (October 1961). "Photoscanning of Bone Lesions Utilizing Strontium 85". Radiology. 77 (4): 635–636. doi: 10.1148/77.4.635. PMID 13893538.
- ^ Subramanian, G.; McAfee, J. G. (April 1971). "A New Complex of 99mTc for Skeletal Imaging". Radiology. 99 (1): 192–196. doi: 10.1148/99.1.192. PMID 5548678.
- ^ Fogelman, I (2013). "The Bone Scan—Historical Aspects". Bone Scanning in Clinical Practice. London: Springer. pp. 1–6. doi: 10.1007/978-1-4471-1407-9_1. ISBN 978-1-4471-1409-3.
- ^ Biersack, Hans-Jürgen; Freeman, Leonard M.; Zuckier, Lionel S.; Grünwald, Frank (2007). Clinical Nuclear Medicine. Berlin: Springer. p. 243. ISBN 9783540280255.
- ^ Weissman, Barbara N (2009). Imaging of Arthritis and Metabolic Bone Disease. Elsevier Health Sciences. p. 17. ISBN 978-0-323-04177-5.
- ^ a b c Van den Wyngaert, T.; Strobel, K.; Kampen, W. U.; Kuwert, T.; van der Bruggen, W.; Mohan, H. K.; Gnanasegaran, G.; Delgado-Bolton, R.; Weber, W. A.; Beheshti, M.; Langsteger, W.; Giammarile, F.; Mottaghy, F. M.; Paycha, F. (4 June 2016). "The EANM practice guidelines for bone scintigraphy". European Journal of Nuclear Medicine and Molecular Imaging. 43 (9): 1723–1738. doi: 10.1007/s00259-016-3415-4. PMC 4932135. PMID 27262701.
- ^ Chopra, A (24 August 2009). "99mTc-Methyl diphosphonate". Molecular Imaging and Contrast Agent Database. National Center for Biotechnology Information (US). PMID 20641923.
- ^ Brenner, Arnold I.; Koshy, June; Morey, Jose; Lin, Cheryl; DiPoce, Jason (January 2012). "The Bone Scan". Seminars in Nuclear Medicine. 42 (1): 11–26. doi: 10.1053/j.semnuclmed.2011.07.005. PMID 22117809.
- ^ Wong, K. K.; Piert, M. (12 March 2013). "Dynamic Bone Imaging with 99mTc-Labeled Diphosphonates and 18F-NaF: Mechanisms and Applications". Journal of Nuclear Medicine. 54 (4): 590–599. doi: 10.2967/jnumed.112.114298. PMID 23482667.
- ^ Verberne, SJ; Raijmakers, PG; Temmerman, OP (5 October 2016). "The Accuracy of Imaging Techniques in the Assessment of Periprosthetic Hip Infection: A Systematic Review and Meta-Analysis". The Journal of Bone and Joint Surgery. American Volume. 98 (19): 1638–1645. doi: 10.2106/jbjs.15.00898. PMID 27707850. S2CID 9202184. Archived from the original on 16 December 2016. Retrieved 20 November 2016.
- ^ a b Donohoe, Kevin J.; Brown, Manuel L.; Collier, B. David; Carretta, Robert F.; Henkin, Robert E.; O'Mara, Robert E.; Royal, Henry D. (20 June 2003). Procedure Guideline for Bone Scintigraphy (PDF) (Report). Society of Nuclear Medicine. 3.0.
- ^ Kane, Tom; Kulshrestha, Randeep; Notghi, Alp; Elias, Mark (2013). "Clinical Utility (Applications) of SPECT/CT". In Wyn Jones, David; Hogg, Peter; Seeram, Euclid (eds.). Practical SPECT/CT in nuclear medicine. London: Springer. p. 197. ISBN 9781447147039.
- ^ a b "Clinical Guideline for Bone Scintigraphy" (PDF). BNMS. July 2014.
- ^ Weissman, Barbara N. (2009). Imaging of arthritis and metabolic bone disease. Philadelphia, PA: Mosby/Elsevier. p. 18. ISBN 9780323041775.
- ^ Schauwecker, D S (January 1992). "The scintigraphic diagnosis of osteomyelitis". American Journal of Roentgenology. 158 (1): 9–18. doi: 10.2214/ajr.158.1.1727365. PMID 1727365.
- ^ Mettler, Fred A.; Huda, Walter; Yoshizumi, Terry T.; Mahesh, Mahadevappa (July 2008). "Effective Doses in Radiology and Diagnostic Nuclear Medicine: A Catalog". Radiology. 248 (1): 254–263. doi: 10.1148/radiol.2481071451. PMID 18566177.
- ^ Moore, A. E.B.; Blake, G. M.; Fogelman, I. (2008-02-20). "Quantitative Measurements of Bone Remodeling Using 99mTc-Methylene Diphosphonate Bone Scans and Blood Sampling". Journal of Nuclear Medicine. 49 (3): 375–382. doi: 10.2967/jnumed.107.048595. ISSN 0161-5505. PMID 18287266.
- ^ Segall, G.; Delbeke, D.; Stabin, M. G.; Even-Sapir, E.; Fair, J.; Sajdak, R.; Smith, G. T. (4 November 2010). "SNM Practice Guideline for Sodium 18F-Fluoride PET/CT Bone Scans 1.0". Journal of Nuclear Medicine. 51 (11): 1813–1820. doi: 10.2967/jnumed.110.082263. PMID 21051652.
- ^ Beheshti, M.; Mottaghy, F. M.; Payche, F.; Behrendt, F. F. F.; Van den Wyngaert, T.; Fogelman, I.; Strobel, K.; Celli, M.; Fanti, S.; Giammarile, F.; Krause, B.; Langsteger, W. (23 July 2015). "18F-NaF PET/CT: EANM procedure guidelines for bone imaging". European Journal of Nuclear Medicine and Molecular Imaging. 42 (11): 1767–1777. doi: 10.1007/s00259-015-3138-y. PMID 26201825.
- ^ Langsteger, Werner; Rezaee, Alireza; Pirich, Christian; Beheshti, Mohsen (November 2016). "18F-NaF-PET/CT and 99mTc-MDP Bone Scintigraphy in the Detection of Bone Metastases in Prostate Cancer". Seminars in Nuclear Medicine. 46 (6): 491–501. doi: 10.1053/j.semnuclmed.2016.07.003. PMID 27825429.
- ^ Beheshti, Mohsen (October 2018). "18F-Sodium Fluoride PET/CT and PET/MR Imaging of Bone and Joint Disorders". PET Clinics. 13 (4): 477–490. doi: 10.1016/j.cpet.2018.05.004. PMID 30219183. S2CID 52280057.
External links
- "Bone scans". WebMD. Retrieved July 9, 2008.